Int J Stem Cells.
2016 Nov;9(2):221-229. 10.15283/ijsc16024.
Assessment of the Role of Noni (Morinda citrifolia) Juice for Inducing Osteoblast Differentiation in Isolated Rat Bone Marrow Derived Mesenchymal Stem Cells
- Affiliations
-
- 1Dental Sciences, Bharat University, Madha Dental College and Hospital, Dr.MGR Medical University, Chennai, Tamilnadu, India.
- 2School of BioSciences and Technology, Vellore Institute of Technology, VIT University, Vellore, Tamilnadu, India. tamizhselvi.r@vit.ac.in
- KMID: 2362516
- DOI: http://doi.org/10.15283/ijsc16024
Abstract
- BACKGROUND AND OBJECTIVES
Morinda citrifolia (Noni), an important traditional medicinal plant still used in patients with bone fractures or dislocation to promote connective tissue repair and to reduce inflammation. However, the effects of Noni on bone metabolism and whether it influences the osteogenic differentiation is yet to be clarified. In this study, we investigated the effect of Morinda citrifolia (Noni) juice on the proliferation rate of rat bone marrow derived mesenchymal stem cells (BMSC) and the osteoblastic differentiation as shown by alkaline phosphatase (ALP), Runt-related transcription factor 2 (Runx2) and osteocalcin (OCN) mRNA expression in vitro.
METHODS AND RESULTS
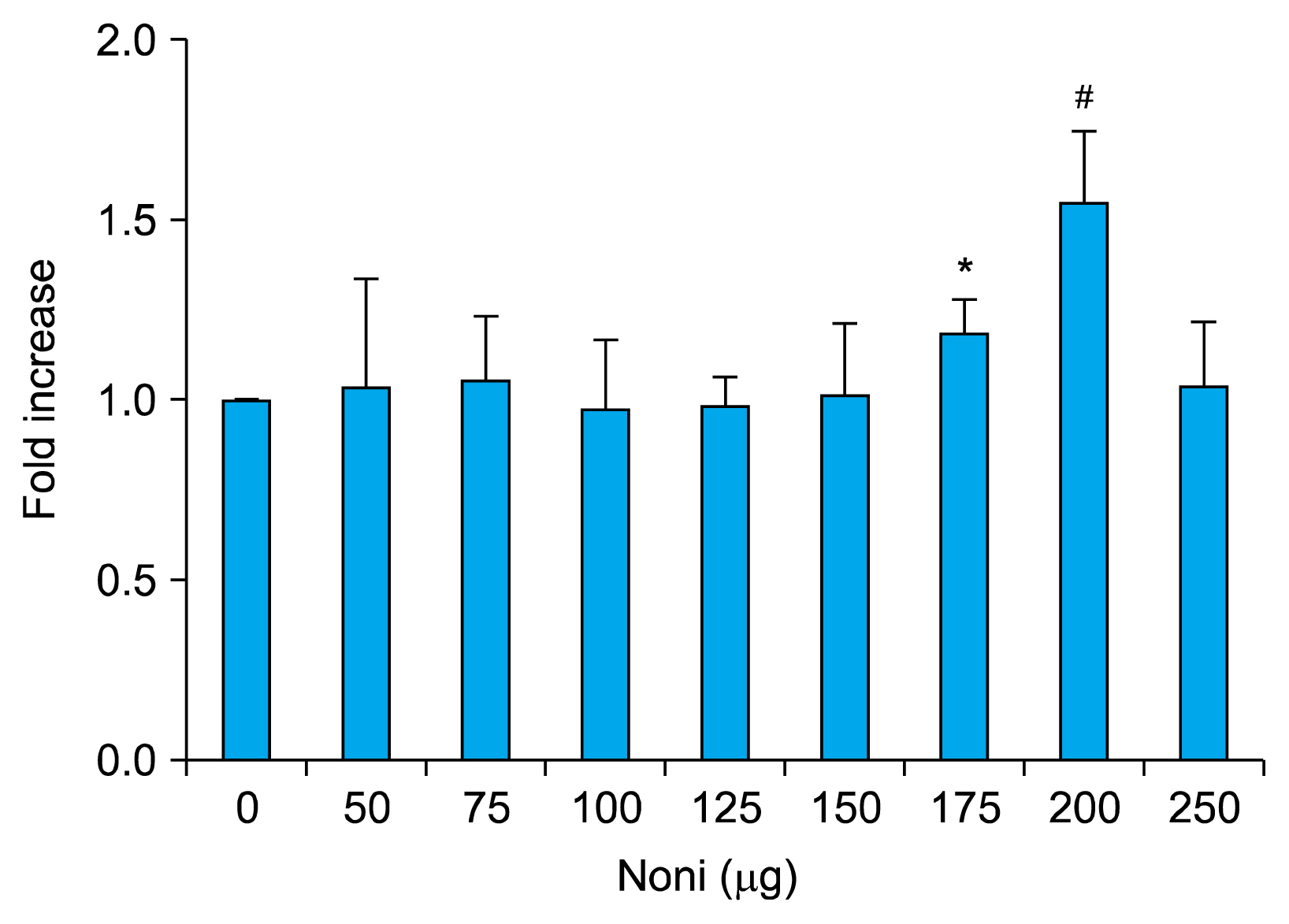
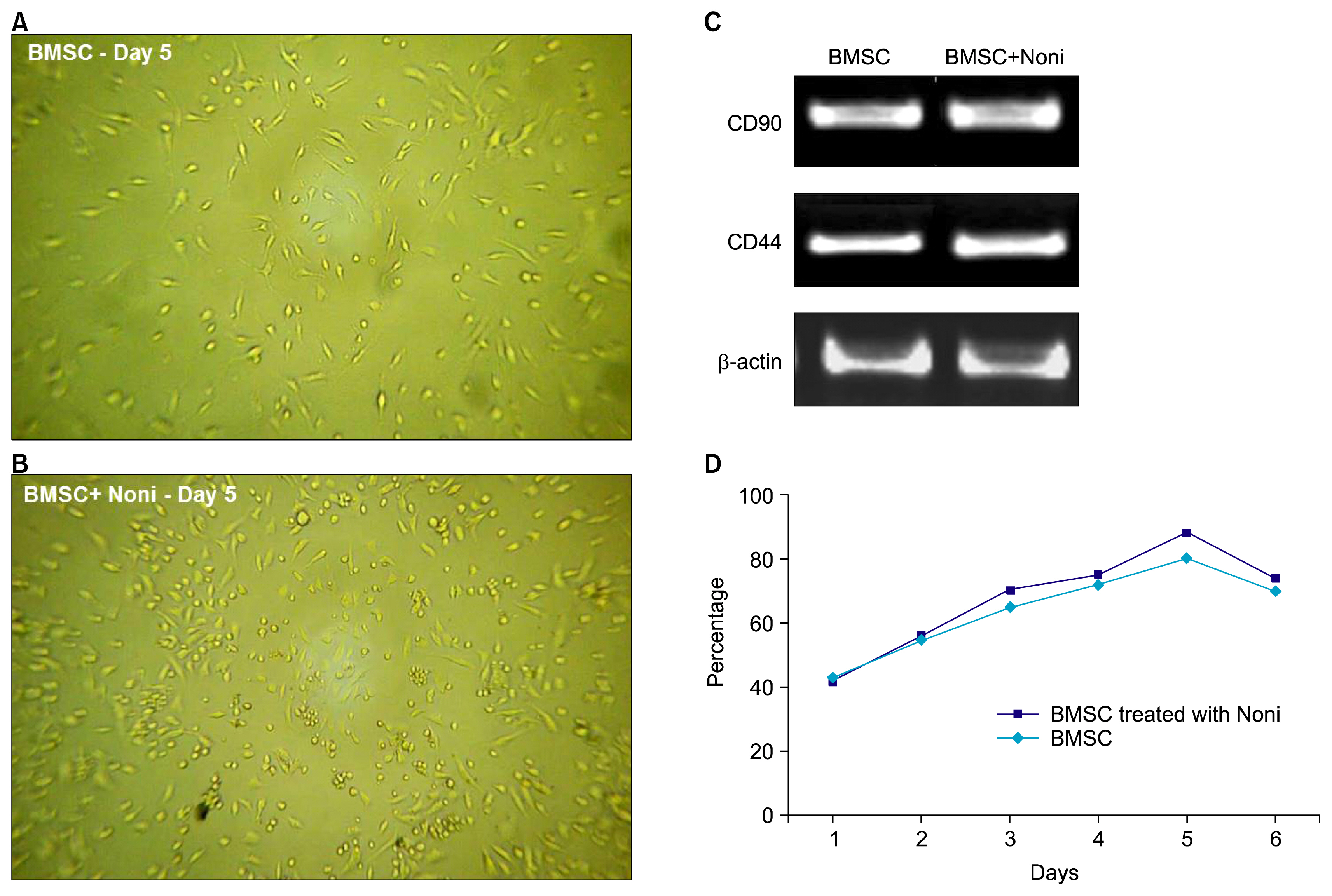
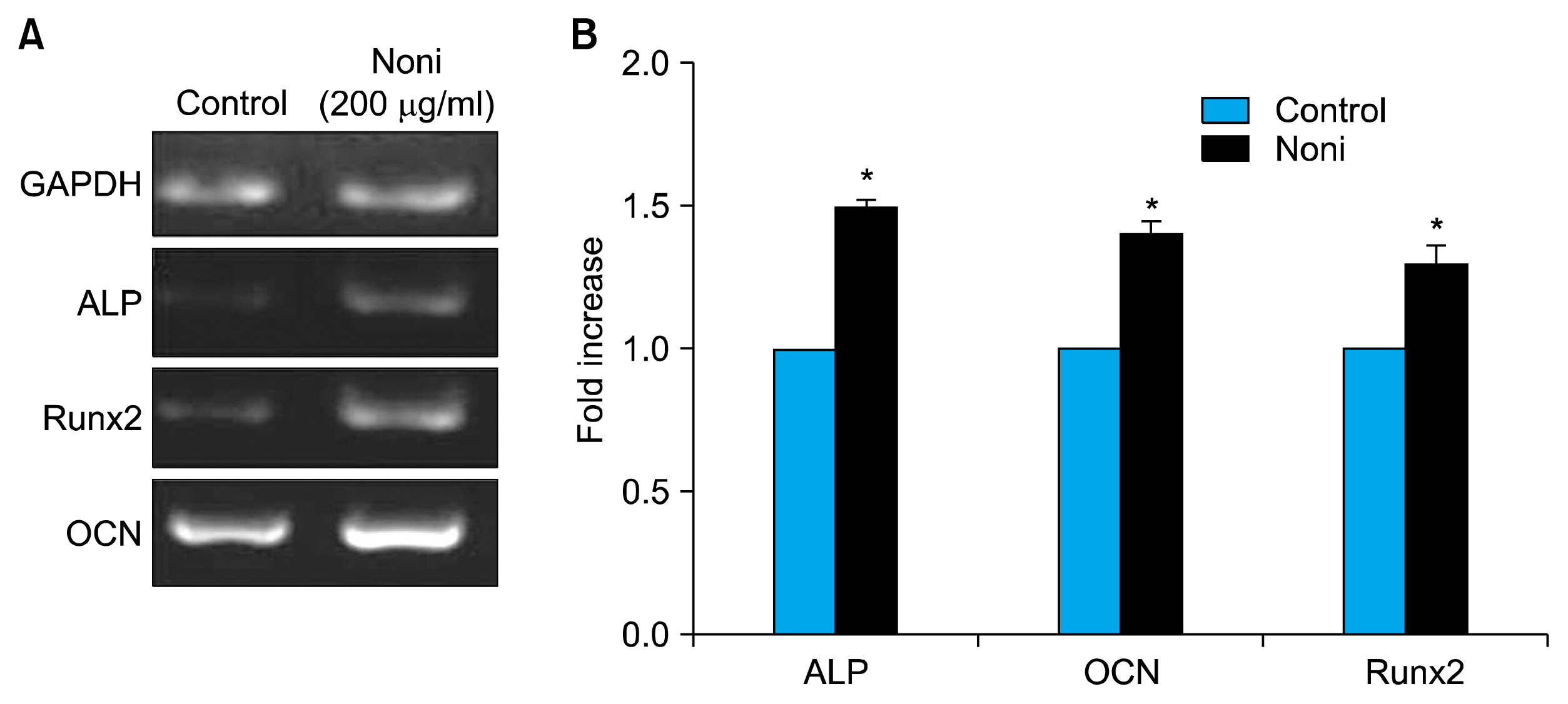
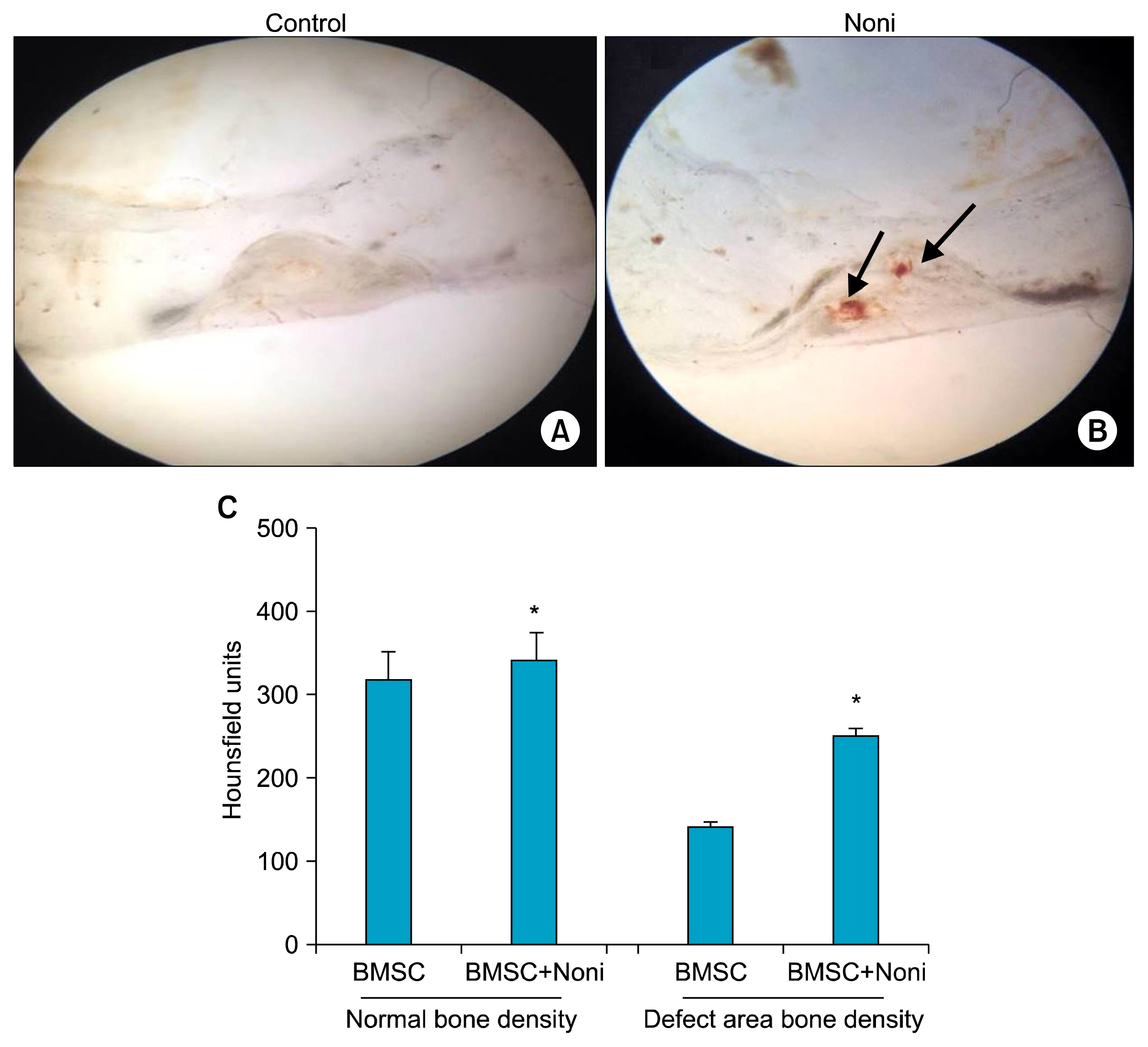
Treatment with 200 μg/ml Noni juice enhanced the proliferation rate of the BMSC and also upregulated the osteogenic differentiation marker genes ALP and OCN, and Runx2 measured by RTPCR. Consistent with these results collagen scaffolds implanted in vivo, which were loaded with BMSC pre-exposed to Noni, showed increased bone density measured by computed tomography and histological analysis revealed neo-angiogenesis for bone formation.
CONCLUSIONS
These results suggest that Noni stimulates osteoblastogenesis and can be used as adjuvant natural medicine for bone diseases such as osteoporosis.
Keyword
MeSH Terms
-
Alkaline Phosphatase
Animals
Bone Density
Bone Diseases
Bone Marrow*
Collagen
Connective Tissue
Dislocations
Fractures, Bone
Humans
In Vitro Techniques
Inflammation
Mesenchymal Stromal Cells*
Metabolism
Morinda*
Osteoblasts*
Osteocalcin
Osteogenesis
Osteoporosis
Plants, Medicinal
Rats*
RNA, Messenger
Transcription Factors
Alkaline Phosphatase
Collagen
Osteocalcin
RNA, Messenger
Transcription Factors
Figure
Reference
-
References
1. The United Nations (UN). 2012. Available from: http://www.un.org/en/. Accessed July 2012.2. The World Health Organization (WHO). 2012. Available from: http://www.who.int/en/. Accessed July 2012.3. Woolf AD, Pfleger B. Burden of major musculoskeletal conditions. Bull World Health Organ. 2003; 81:646–656.4. Scheven BA, Visser JW, Nijweide PJ. In vitro osteoclast generation from different bone marrow fractions, including a highly enriched haematopoietic stem cell population. Nature. 1986; 321:79–81. DOI: 10.1038/321079a0. PMID: 3754620.
Article5. Long F. Building strong bones: molecular regulation of the osteoblast lineage. Nat Rev Mol Cell Biol. 2011; 13:27–38. DOI: 10.1038/nrm3254. PMID: 22189423.
Article6. Deng S, West BJ, Palu ‘K, Jensen CJ. Determination and comparative analysis of major iridoids in different parts and cultivation sources of Morinda citrifolia. Phytochem Anal. 2011; 22:26–30. DOI: 10.1002/pca.1246.
Article7. Paredes R, Arriagada G, Cruzat F, Olate J, Van Wijnen A, Lian J, Stein G, Stein J, Montecino M. The Runx2 transcription factor plays a key role in the 1alpha,25-dihydroxy Vitamin D3-dependent upregulation of the rat osteocalcin (OC) gene expression in osteoblastic cells. J Steroid Biochem Mol Biol. 2004; 89–90:269–271. DOI: 10.1016/j.jsbmb.2004.03.076.8. Johnsen SA, Subramaniam M, Ruesink TJ, Spelsberg TC. Opposing roles of osterix and TGF beta inducible early gene in osteoblast differentiation downstream of Cbfa1/Runx2. J Bone Miner Res. 2002; 17:1016–1019.9. Marom R, Shur I, Solomon R, Benayahu D. Characterization of adhesion and differentiation markers of osteogenic marrow stromal cells. J Cell Physiol. 2005; 202:41–48. DOI: 10.1002/jcp.20109.
Article10. Aubin JE. Regulation of osteoblast formation and function. Rev Endocr Metab Disord. 2001; 2:81–94. DOI: 10.1023/A:1010011209064. PMID: 11704982.11. Bushnell OA, Fukuda M, Makinodian T. The antibacterial properties of some plants found in Hawaii. Pac Sci. 1950; 4:167–183.12. Pawlus AD, Su BN, Keller WJ, Kinghorn AD. An anthraquinone with potent quinone reductase-inducing activity and other constituents of the fruits of Morinda citrifolia (noni). J Nat Prod. 2005; 68:1720–1722. DOI: 10.1021/np050383k. PMID: 16378361.
Article13. Akihisa T, Matsumoto K, Tokuda H, Yasukawa K, Seino K, Nakamoto K, Kuninaga H, Suzuki T, Kimura Y. Anti-inflammatory and potential cancer chemopreventive constituents of the fruits of Morinda citrifolia (Noni). J Nat Prod. 2007; 70:754–757. DOI: 10.1021/np068065o. PMID: 17480098.
Article14. Alitheen NB, Manaf AA, Yeap SK, Shuhaimi M, Nordin L, Mashitoh AR. Immunomodulatory effects of damnacanthal isolated from roots of Morinda elliptica. Pharm Biol. 2010; 48:446–452. DOI: 10.3109/13880200903168031. PMID: 20645725.
Article15. Gupta RK, Patel AK. Do the health claims made for Morinda citrifolia (Noni) harmonize with current scientific knowledge and evaluation of its biological effects. Asian Pac J Cancer Prev. 2013; 14:4495–4499. DOI: 10.7314/APJCP.2013.14.8.4495. PMID: 24083691.
Article16. Wong YH, Abdul Kadir H. Induction of mitochondria-mediated apoptosis in Ca Ski human cervical cancer cells triggered by mollic acid arabinoside isolated from leea indica. Evid Based Complement Alternat Med. 2012; DOI: 10.1155/2012/684740.17. Gupta RK, Banerjee A, Pathak S, Sharma C, Singh N. Induction of mitochondrial-mediated apoptosis by Morinda citrifolia (Noni) in human cervical cancer cells. Asian Pac J Cancer Prev. 2013; 14:237–242. DOI: 10.7314/APJCP.2013.14.1.237. PMID: 23534730.
Article18. West BJ, Deng S, Jensen CJ. Nutrient and phytochemical analyses of processed noni puree. Food Res Intern. 2011; 44:2295–2301. DOI: 10.1016/j.foodres.2010.09.038.
Article19. Geiger F, Lorenz H, Xu W, Szalay K, Kasten P, Claes L, Augat P, Richter W. VEGF producing bone marrow stromal cells (BMSC) enhance vascularization and resorption of a natural coral bone substitute. Bone. 2007; 41:516–522. DOI: 10.1016/j.bone.2007.06.018. PMID: 17693148.
Article20. Kasten P, Luginbühl R, van Griensven M, Barkhausen T, Krettek C, Bohner M, Bosch U. Comparison of human bone marrow stromal cells seeded on calcium-deficient hydroxyapatite, beta-tricalcium phosphate and demineralized bone matrix. Biomaterials. 2003; 24:2593–2603. DOI: 10.1016/S0142-9612(03)00062-0. PMID: 12726713.
Article21. Bhargavan B, Gautam AK, Singh D, Kumar A, Chaurasia S, Tyagi AM, Yadav DK, Mishra JS, Singh AB, Sanyal S, Goel A, Maurya R, Chattopadhyay N. Methoxylated isoflavones, cajanin and isoformononetin, have non-estrogenic bone forming effect via differential mitogen activated protein kinase (MAPK) signaling. J Cell Biochem. 2009; 108:388–399. DOI: 10.1002/jcb.22264. PMID: 19598169.
Article22. Bramorski A, Cherem AR, Marmentini CP, Torresani J, Mezadri T, Costa AAS. Total polyphenol content and antioxidant activity of commercial Noni (Morinda citrifolia L.) juice and its components. Braz J Pharm Sci. 2010; 46:651–656. DOI: 10.1590/S1984-82502010000400006.
Article23. Srivastava S, Bankar R, Roy P. Assessment of the role of flavonoids for inducing osteoblast differentiation in isolated mouse bone marrow derived mesenchymal stem cells. Phytomedicine. 2013; 20:683–690. DOI: 10.1016/j.phymed.2013.03.001. PMID: 23570998.
Article24. Deschaseaux F, Sensébé L, Heymann D. Mechanisms of bone repair and regeneration. Trends Mol Med. 2009; 15:417–429. DOI: 10.1016/j.molmed.2009.07.002. PMID: 19740701.
Article25. Ducy P, Karsenty G. Two distinct osteoblast-specific cis-acting elements control expression of a mouse osteocalcin gene. Mol Cell Biol. 1995; 15:1858–1869. DOI: 10.1128/MCB.15.4.1858. PMID: 7891679. PMCID: 230411.
Article26. Mohd Zin Z, Abdul Hamid A, Osman A, Saari N, Misran A. Isolation and identification of antioxidative compound from fruit of mengkudu (Morinda citrifolia L). Int J Food Prop. 2007; 10:363–373. DOI: 10.1080/10942910601052723.
Article27. Chen CH, Ho ML, Chang JK, Hung SH, Wang GJ. Green tea catechin enhances osteogenesis in a bone marrow mesenchymal stem cell line. Osteoporos Int. 2005; 16:2039–2045. DOI: 10.1007/s00198-005-1995-0. PMID: 16170444.
Article28. Parenteau-Bareil R, Gauvin R, Berthod F. Collagen-based biomaterials for tissue engineering applications. Materials. 2010; 3:1863–1887. DOI: 10.3390/ma3031863.
Article29. Rodrigues M, Turner O, Stolz D, Griffith LG, Wells A. Production of reactive oxygen species by multipotent stromal cells/mesenchymal stem cells upon exposure to fas ligand. Cell Transplant. 2012; 21:2171–2187. DOI: 10.3727/096368912X639035. PMID: 22526333. PMCID: 3876879.
Article30. Dussossoy E, Brat P, Bony E, Boudard F, Poucheret P, Mertz C, Giaimis J, Michel A. Characterization, anti-oxidative and anti-inflammatory effects of Costa Rican noni juice (Morinda citrifolia L.). J Ethnopharmacol. 2011; 133:108–115. DOI: 10.1016/j.jep.2010.08.063.
Article31. Serafini MR, Santos RC, Guimarães AG, Dos Santos JP, da Conceicão Santos AD, Alves IA, Gelain DP, de Lima Nogueira PC, Quintans-Júnior LJ, Bonjardim LR, de Souza Araújo AA. Morinda citrifolia Linn leaf extract possesses antioxidant activities and reduces nociceptive behavior and leukocyte migration. J Med Food. 2011; 14:1159–1166. DOI: 10.1089/jmf.2010.0254. PMID: 21548805.
Article32. Wang MY, West BJ, Jensen CJ, Nowicki D, Su C, Palu AK, Anderson G. Morinda citrifolia (Noni): a literature review and recent advances in Noni research. Acta Pharmacol Sin. 2002; 23:1127–1141. PMID: 12466051.33. Potterat O, Hamburger M. Morinda citrifolia (Noni) fruit--phytochemistry, pharmacology, safety. Planta Med. 2007; 73:191–199. DOI: 10.1055/s-2007-967115. PMID: 17286240.
Article34. Deng ZL, Sharff KA, Tang N, Song WX, Luo J, Luo X, Chen J, Bennett E, Reid R, Manning D, Xue A, Montag AG, Luu HH, Haydon RC, He TC. Regulation of osteogenic differentiation during skeletal development. Front Biosci. 2008; 13:2001–2021. DOI: 10.2741/2819.35. Wang MY, Lutfiyya MN, Weidenbacher-Hoper V, Anderson G, Su CX, West BJ. Antioxidant activity of noni juice in heavy smokers. Chem Cent J. 2009; 3:13–17. DOI: 10.1186/1752-153X-3-13. PMID: 19807926. PMCID: 2765950.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Chemical Constituents of Fermented Noni (Morinda citrifolia) Juice Exudates and Their Biological Activity
- Effects of bFGF and PDGF-BB on osteoblast differentiation of bone marrow-derived mesenchymal stem cell in rat
- Concise Review: Differentiation of Human Adult Stem Cells Into Hepatocyte-like Cells In vitro
- Differential Potential of Stem Cells Following Their Origin: Subacromial Bursa, Bone Marrow, Umbilical Cord Blood
- Isolation of Mesenchymal Stem Cells from the Mononuclear Cells Remaining in the Bone Marrow Processing Kit