Allergy Asthma Immunol Res.
2020 May;12(3):523-536. 10.4168/aair.2020.12.3.523.
Oxidative Stress Modulates the Expression Pattern of Peroxiredoxin-6 in Peripheral Blood Mononuclear Cells of Asthmatic Patients and Bronchial Epithelial Cells
- Affiliations
-
- 1Division of Allergy and Clinical Immunology, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. yscho@amc.seoul.kr
- 2Division of Pulmonary, Allergy and Critical Care Medicine, Department of Internal Medicine, Konkuk University Medical Center, Seoul, Korea.
- KMID: 2471234
- DOI: http://doi.org/10.4168/aair.2020.12.3.523
Abstract
- PURPOSE
Reduction-oxidation reaction homeostasis is vital for regulating inflammatory conditions and its dysregulation may affect the pathogenesis of chronic airway inflammatory diseases such as asthma. Peroxiredoxin-6, an important intracellular anti-oxidant molecule, is reported to be highly expressed in the airways and lungs. The aim of this study was to analyze the expression pattern of peroxiredoxin-6 in the peripheral blood mononuclear cells (PBMCs) of asthmatic patients and in bronchial epithelial cells (BECs).
METHODS
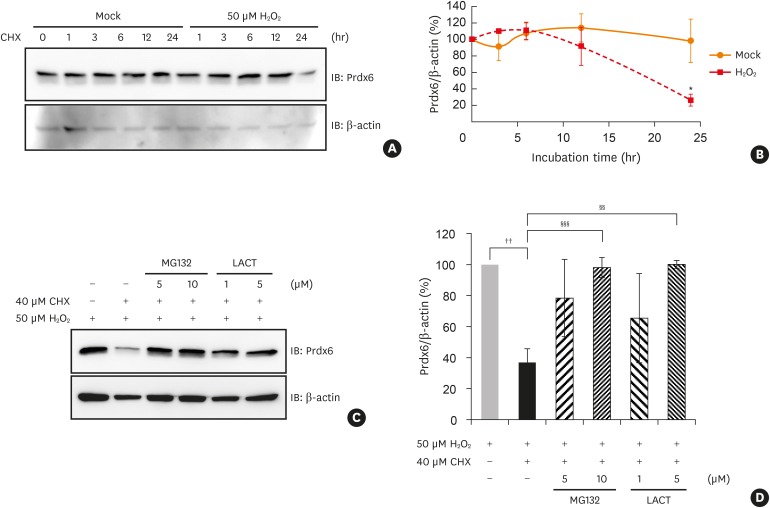
The expression levels and modifications of peroxiredoxin-6 were evaluated in PBMCs from 22 asthmatic patients. Phosphorylated and acetylated peroxiredoxin-6 in hydrogen peroxide-treated human BECs was detected using immunoprecipitation analysis. The expression level of peroxiredoxin-6 was also investigated in BECs treated with hydrogen peroxide. Cycloheximide and proteasome inhibitors were used to determine whether peroxiredoxin-6 is degraded by proteasomes.
RESULTS
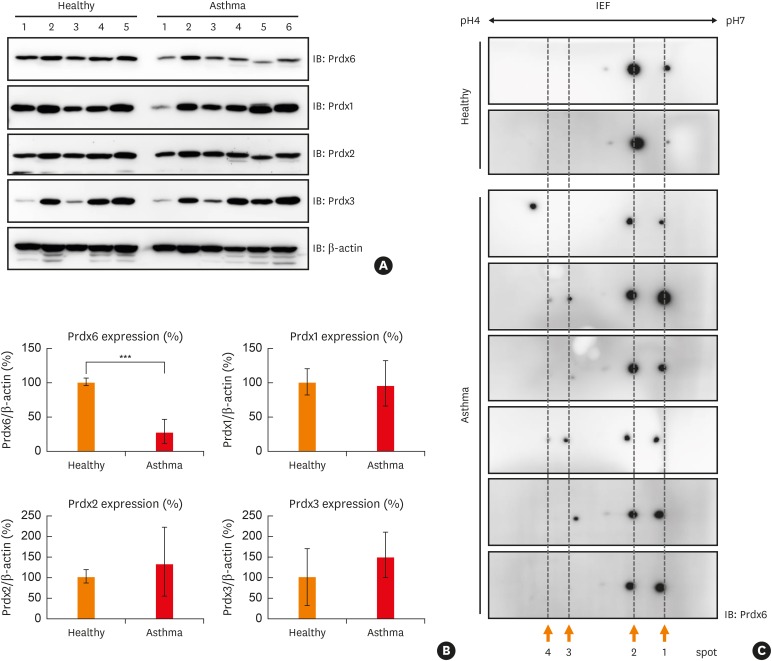
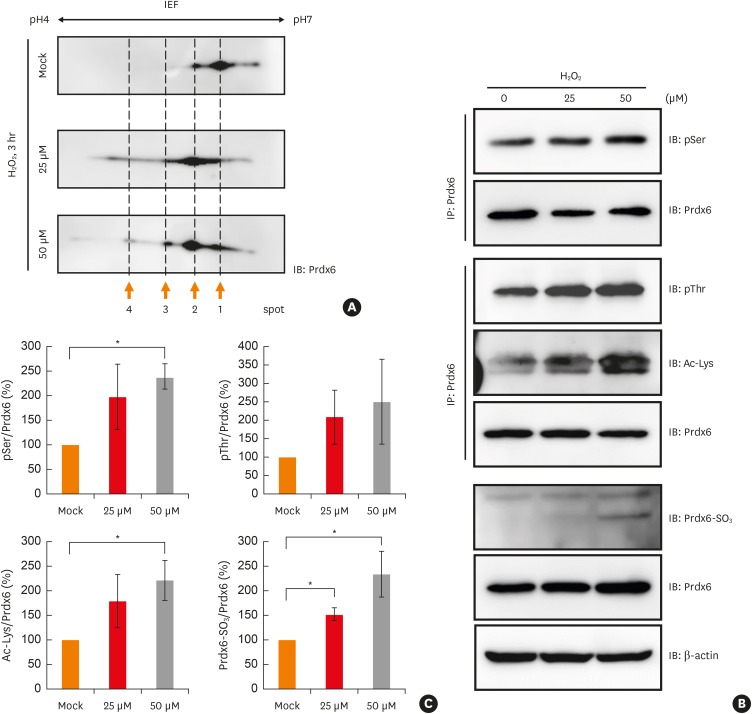
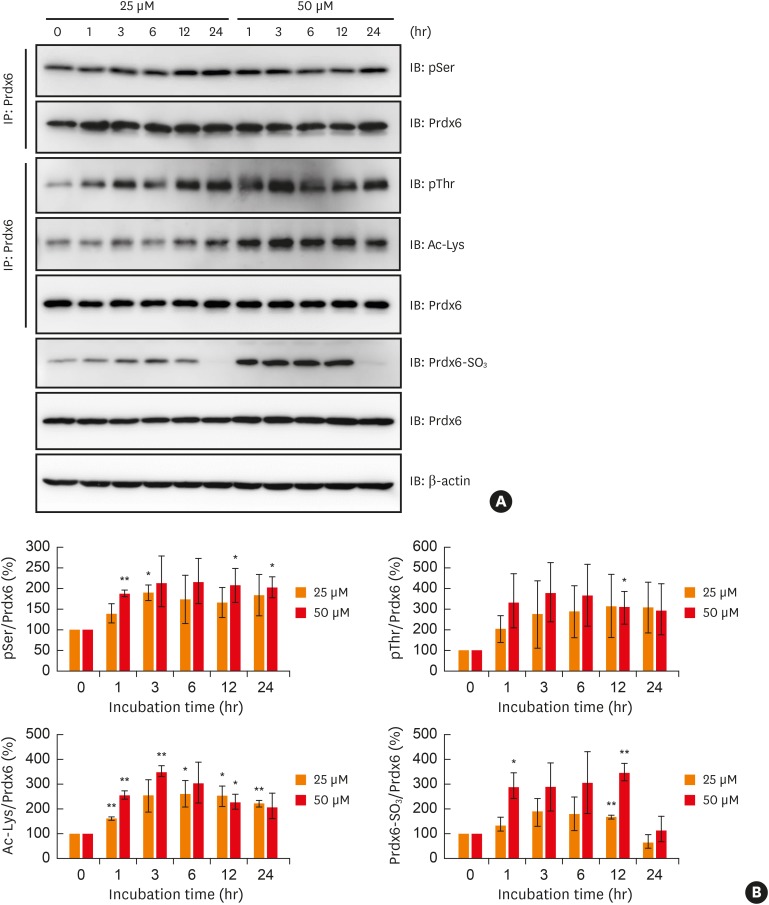
Peroxiredoxin-6 expression was significantly reduced in the PBMCs of asthmatic patients compared to control subjects. Distinct modification patterns for peroxiredoxin-6 were observed in the PBMCs of asthmatic patients using 2-dimensional-electrophoresis. The levels of phosphorylated serine and acetylated lysine in peroxiredoxin-6 were significantly increased in the BECs following hydrogen peroxide treatment. The level of peroxiredoxin-6 expression was reduced in hydrogen peroxide-stimulated BECs, presumably due to proteasomes.
CONCLUSIONS
The expression of peroxiredoxin-6, which is down-regulated in the immune cells of asthmatic patients and BECs, can be modified by oxidative stress. This phenomenon may have an effect on asthmatic airway inflammation.
Keyword
MeSH Terms
Figure
Reference
-
1. Cho YS, Moon HB. The role of oxidative stress in the pathogenesis of asthma. Allergy Asthma Immunol Res. 2010; 2:183–187. PMID: 20592917.
Article2. Brown DI, Griendling KK. Nox proteins in signal transduction. Free Radic Biol Med. 2009; 47:1239–1253. PMID: 19628035.
Article3. Li N, Nel AE. Role of the Nrf2-mediated signaling pathway as a negative regulator of inflammation: implications for the impact of particulate pollutants on asthma. Antioxid Redox Signal. 2006; 8:88–98. PMID: 16487041.
Article4. Bullone M, Lavoie JP. The contribution of oxidative stress and inflamm-aging in human and equine asthma. Int J Mol Sci. 2017; 18:E2612. PMID: 29206130.
Article5. Takeyama K, Dabbagh K, Shim JJ, Dao-Pick T, Ueki IF, Nadel JA. Oxidative stress causes mucin synthesis via transactivation of epidermal growth factor receptor: role of neutrophils. J Immunol. 2000; 164:1546–1552. PMID: 10640773.
Article6. Fitzpatrick AM, Teague WG, Holguin F, Yeh M, Brown LA. Severe Asthma Research Program. Airway glutathione homeostasis is altered in children with severe asthma: evidence for oxidant stress. J Allergy Clin Immunol. 2009; 123:146–152.e8. PMID: 19130935.
Article7. Stone JR, Yang S. Hydrogen peroxide: a signaling messenger. Antioxid Redox Signal. 2006; 8:243–270. PMID: 16677071.
Article8. Trachootham D, Lu W, Ogasawara MA, Nilsa RD, Huang P. Redox regulation of cell survival. Antioxid Redox Signal. 2008; 10:1343–1374. PMID: 18522489.
Article9. Veal EA, Day AM, Morgan BA. Hydrogen peroxide sensing and signaling. Mol Cell. 2007; 26:1–14. PMID: 17434122.
Article10. Robinson MW, Hutchinson AT, Dalton JP, Donnelly S. Peroxiredoxin: a central player in immune modulation. Parasite Immunol. 2010; 32:305–313. PMID: 20500659.
Article11. Wang Y, Manevich Y, Feinstein SI, Fisher AB. Adenovirus-mediated transfer of the 1-cys peroxiredoxin gene to mouse lung protects against hyperoxic injury. Am J Physiol Lung Cell Mol Physiol. 2004; 286:L1188–93. PMID: 15136296.
Article12. Grimsrud PA, Xie H, Griffin TJ, Bernlohr DA. Oxidative stress and covalent modification of protein with bioactive aldehydes. J Biol Chem. 2008; 283:21837–21841. PMID: 18445586.
Article13. Riquier S, Breton J, Abbas K, Cornu D, Bouton C, Drapier JC. Peroxiredoxin post-translational modifications by redox messengers. Redox Biol. 2014; 2:777–785. PMID: 25009779.
Article14. Pajares M, Jiménez-Moreno N, Dias IH, Debelec B, Vucetic M, Fladmark KE, et al. Redox control of protein degradation. Redox Biol. 2015; 6:409–420. PMID: 26381917.
Article15. Davies KJ. Degradation of oxidized proteins by the 20S proteasome. Biochimie. 2001; 83:301–310. PMID: 11295490.
Article16. Spange S, Wagner T, Heinzel T, Krämer OH. Acetylation of non-histone proteins modulates cellular signalling at multiple levels. Int J Biochem Cell Biol. 2009; 41:185–198. PMID: 18804549.
Article17. Song IK, Lee JJ, Cho JH, Jeong J, Shin DH, Lee KJ. Degradation of redox-sensitive proteins including peroxiredoxins and DJ-1 is promoted by oxidation-induced conformational changes and ubiquitination. Sci Rep. 2016; 6:34432. PMID: 27703196.
Article18. Kwon HS, Bae YJ, Moon KA, Lee YS, Lee T, Lee KY, et al. Hyperoxidized peroxiredoxins in peripheral blood mononuclear cells of asthma patients is associated with asthma severity. Life Sci. 2012; 90:502–508. PMID: 22285837.
Article19. Sahiner UM, Birben E, Erzurum S, Sackesen C, Kalayci O. Oxidative stress in asthma. World Allergy Organ J. 2011; 4:151–158. PMID: 23268432.
Article20. Zuo L, Otenbaker NP, Rose BA, Salisbury KS. Molecular mechanisms of reactive oxygen species-related pulmonary inflammation and asthma. Mol Immunol. 2013; 56:57–63. PMID: 23665383.
Article21. Dozor AJ. The role of oxidative stress in the pathogenesis and treatment of asthma. Ann N Y Acad Sci. 2010; 1203:133–137. PMID: 20716295.
Article22. Ahmad A, Shameem M, Husain Q. Relation of oxidant-antioxidant imbalance with disease progression in patients with asthma. Ann Thorac Med. 2012; 7:226–232. PMID: 23189100.
Article23. Comhair SA, Erzurum SC. Redox control of asthma: molecular mechanisms and therapeutic opportunities. Antioxid Redox Signal. 2010; 12:93–124. PMID: 19634987.
Article24. Rahman I, Biswas SK, Kode A. Oxidant and antioxidant balance in the airways and airway diseases. Eur J Pharmacol. 2006; 533:222–239. PMID: 16500642.
Article25. Sackesen C, Ercan H, Dizdar E, Soyer O, Gumus P, Tosun BN, et al. A comprehensive evaluation of the enzymatic and nonenzymatic antioxidant systems in childhood asthma. J Allergy Clin Immunol. 2008; 122:78–85. PMID: 18485467.
Article26. Moon EY, Kang JS, Han SH, Yang KH, Pyo S, Lee MY, et al. Differential role of peroxiredoxin II (PrxII) on the expression of toll-like receptor 4 (TLR4) and B-cell activating factor (BAFF) in ovalbumin (OVA)-induced mouse asthma. Int Immunopharmacol. 2008; 8:935–944. PMID: 18442801.
Article27. Eismann T, Huber N, Shin T, Kuboki S, Galloway E, Wyder M, et al. Peroxiredoxin-6 protects against mitochondrial dysfunction and liver injury during ischemia-reperfusion in mice. Am J Physiol Gastrointest Liver Physiol. 2009; 296:G266–74. PMID: 19033532.
Article28. Wang Y, Feinstein SI, Fisher AB. Peroxiredoxin 6 as an antioxidant enzyme: protection of lung alveolar epithelial type II cells from H2O2-induced oxidative stress. J Cell Biochem. 2008; 104:1274–1285. PMID: 18260127.29. Rhee SG, Woo HA, Kil IS, Bae SH. Peroxiredoxin functions as a peroxidase and a regulator and sensor of local peroxides. J Biol Chem. 2012; 287:4403–4410. PMID: 22147704.
Article30. Jeong J, Kim Y, Seong JK, Lee KJ. Comprehensive identification of novel post-translational modifications in cellular peroxiredoxin 6. Proteomics. 2012; 12:1452–1462. PMID: 22589192.
Article31. Jung T, Höhn A, Grune T. The proteasome and the degradation of oxidized proteins: Part II - protein oxidation and proteasomal degradation. Redox Biol. 2014; 2:99–104. PMID: 25460724.
Article32. Rhee SG, Yang KS, Kang SW, Woo HA, Chang TS. Controlled elimination of intracellular H2O2: regulation of peroxiredoxin, catalase, and glutathione peroxidase via post-translational modification. Antioxid Redox Signal. 2005; 7:619–626. PMID: 15890005.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effect of Oxidative Stress on the Expression of Gelatinases in Cultured RPE
- Peroxiredoxin 2 Inhibits Lipopolysaccharide Induced Mucin Expression and Reactive Oxygen Species Production in Human Airway Epithelial Cells
- Effects of Antioxidant on Oxidative Stress and Autophagy in Bronchial Epithelial Cells Exposed to Particulate Matter and Cigarette Smoke Extract
- Exposure to Toluene Diisocyanate (TDI) Induces IL-8 Production from Bronchial Epithelial Cells: Effect of Pro-inflammatory Cytokines
- Modulation of Peroxiredoxin I Expression by UVB Irradiation in Human Keratinocytes: H2O2-mediated Modulation of Peroxiredoxin I