Korean Circ J.
2009 Feb;39(2):57-65. 10.4070/kcj.2009.39.2.57.
Angiopoietin-1 Protects Endothelial Cells From Hypoxia-Induced Apoptosis via Inhibition of Phosphatase and Tensin Homologue Deleted From Chromosome Ten
- Affiliations
-
- 1National Research Laboratory for Cardiovascular Stem Cells and IRICT, Seoul National University Hospital, Seoul, Korea. hyosoo@snu.ac.kr
- 2Department of Internal Medicine and Cardiovascular Center, Seoul National University Hospital, Seoul, Korea.
- 3Ischemic/Hypoxic Disease Institute, Seoul National University College of Medicine, Seoul, Korea.
- 4Cardiovascular Center, Seoul National University Bundang Hospital, Seongnam, Korea.
- 5Biomedical Research Center, Korea Advanced Institute of Science and Technology, Daejeon, Korea.
- KMID: 2297950
- DOI: http://doi.org/10.4070/kcj.2009.39.2.57
Abstract
- BACKGROUND AND OBJECTIVES
Angiopoietin-1 (Ang1) is a regulator of blood vessel growth and maturation, and prevents radiation-induced or serum deprivation-induced apoptosis. Phosphatase and tensin homologue deleted from chromosome ten (PTEN), a well-known tumor suppressor, regulates cell cycle arrest and apoptosis. Hypoxia induces apoptosis by increasing the expression of PTEN. We hypothesized that Ang1 may regulate PTEN expression and, thus, reduce endothelial apoptosis under hypoxia in vitro and in vivo. Materials and
METHODS
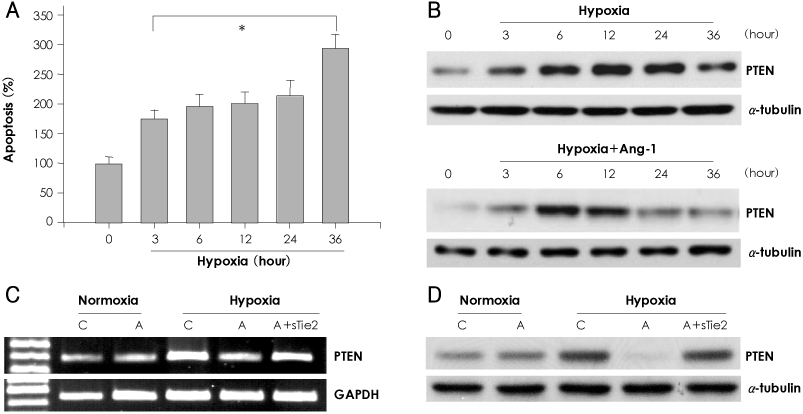
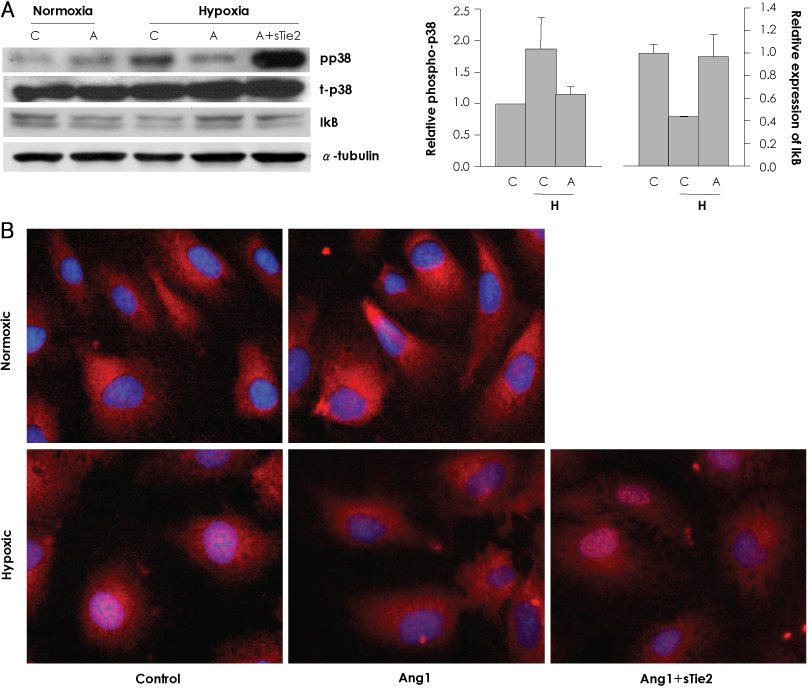
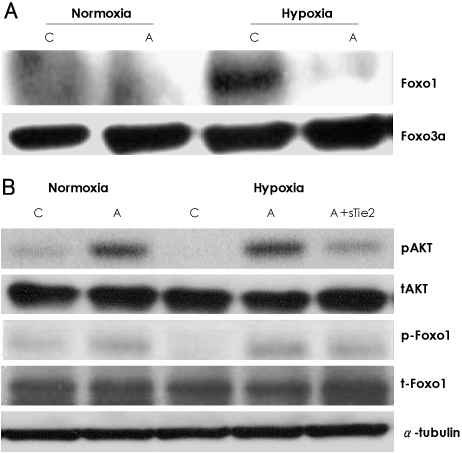
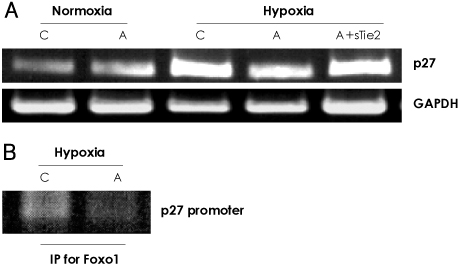
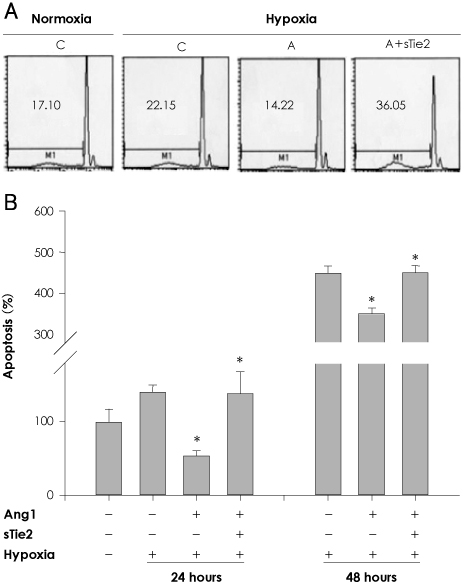
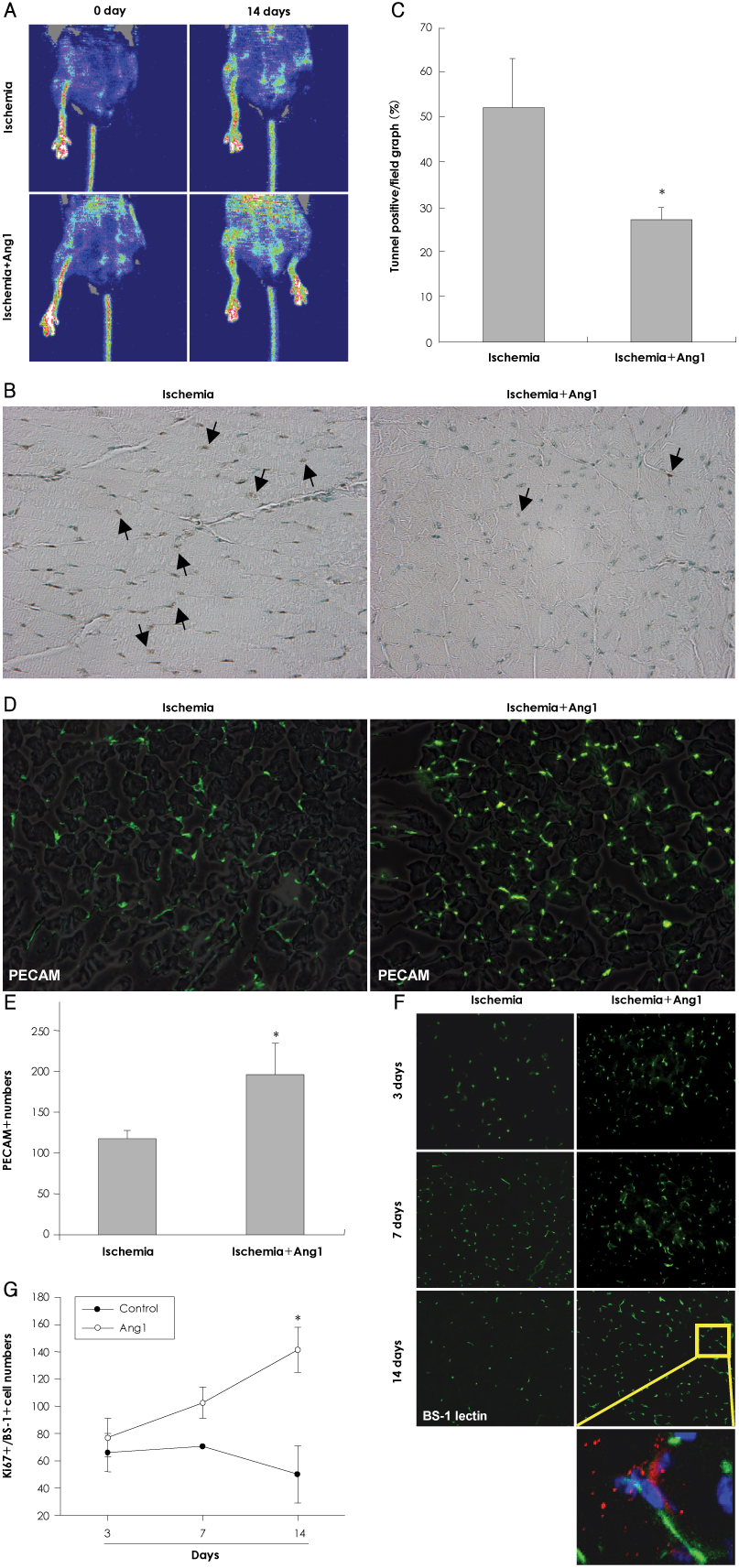
In vitro, human umbilical vein endothelial cells (HUVECs) were treated with Ang1, and signaling pathways were investigated. In vivo, eight-week-old C57BL/6 mice were used for a hind limb ischemia model. Ang1 or normal saline was intramusculary injected. Blood flow was evaluated by a laser Doppler perfusion analyzer and tissue histology. RESULTS: The expression of PTEN was markedly upregulated in HUVECs after hypoxic stimulation, whereas Ang1 suppressed PTEN expression. Tie2-Fc, a soluble form of Tie2 (sTie2) that blocks Ang1, reversed the Ang1 effect on PTEN reduction under hypoxia. Ang1 inhibited the nuclear translocation of nuclear transcription factor-kB (NF-kB), a binding factor for the PTEN promoter and Foxo1. Hypoxia-induced p27 expression and apoptosis were also suppressed by Ang1. In the mouse hind limb ischemia model, we observed a high capillary density, numerous proliferating cells and diminished cell death in skeletal muscle tissue in the Ang1 injected group.
CONCLUSION
Ang1 enhanced endothelial cell survival by reducing apoptosis via PTEN down-regulation in HUVECs under hypoxia. Local injection of Ang1 significantly reduced apoptotic cells in vivo, and prevented limb loss for ischemic hind limb mice. Thus, Ang1 may be an effective therapeutic for protection from ischemic-endothelial cell injury.
Keyword
MeSH Terms
-
Angiopoietin-1
Animals
Anoxia
Apoptosis
Blood Vessels
Capillaries
Cell Cycle
Cell Cycle Checkpoints
Cell Death
Down-Regulation
Endothelial Cells
Extremities
Glycosaminoglycans
Human Umbilical Vein Endothelial Cells
Ischemia
Mice
Microfilament Proteins
Muscle, Skeletal
Perfusion
Angiopoietin-1
Glycosaminoglycans
Microfilament Proteins
Figure
Reference
-
1. Di Cristofano A, Pandolfi PP. The multiple roles of PTEN in tumor suppression. Cell. 2000. 100:387–390.2. Li DM, Sun H. PTEN/MMAC1/TEP1 suppresses the tumorigenicity and induces G1 cell cycle arrest in human glioblastoma cells. Proc Natl Acad Sci U S A. 1998. 95:15406–15411.3. Haas-Kogan D, Shalev N, Wong M, Mills G, Yount G, Stokoe D. Protein kinase B (PKB/Akt) activity is elevated in glioblastoma cells due to mutation of the tumor suppressor PTEN/MMAC. Curr Biol. 1998. 8:1195–1198.4. Yan X, Fraser M, Qiu Q, Tsang BK. Over-expression of PTEN sensitizes human ovarian cancer cells to cisplatin-induced apoptosis in a p53-dependent manner. Gynecol Oncol. 2006. 102:348–355.5. Delgado-Esteban M, Martin-Zanca D, Andres-Martin L, Almeida A, Bolaños JP. Inhibition of PTEN by peroxynitrite activates the phosphoinositide-3-kinase/Akt neuroprotective signaling pathway. J Neurochem. 2007. 102:194–205.6. Jain RK. Molecular regulation of vessel maturation. Nat Med. 2003. 9:685–693.7. Yancopoulos GD, Davis S, Gale NW, Rudge JS, Wiegand SJ, Holash J. Vascular-specific growth factors and blood vessel formation. Nature. 2000. 407:242–248.8. Kim HJ, Kim DK. Angiogenic gene therapy in patients with ischemic cardiovascular diseases. Korean Circ J. 2003. 33:7–14.9. Lee SW, Kim WJ, Choi YK, et al. SSeCKS regulates angiogenesis and tight junction formation in blood-brain barrier. Nat Med. 2003. 9:900–906.10. Nag S. The blood-brain barrier and cerebral angiogenesis: lessons from the cold-injury model. Trends Mol Med. 2002. 8:38–44.11. Cho CH, Kammerer RA, Lee HJ, et al. Designed angiopoietin-1 variant, COMP-Ang1, protects against radiation-induced endothelial cell apoptosis. Proc Natl Acad Sci U S A. 2004. 101:5553–5558.12. Dallabrida SM, Ismail N, Oberle JR, Himes BE, Rupnick MA. Angiopoietin-1 promotes cardiac and skeletal myocyte survival throuth integrins. Circ Res. 2005. 96:e8–e24.13. Park SJ, Kim DH, Seo JB, et al. Thalidomide as a potent inhibitor of neointimal hyperplasia after balloon injury in rat carotid artery. Korean Circ J. 2004. 34:346–355.14. Freedman SJ, Sun ZY, Kung AL, France DS, Wagner G, Eck MJ. Structural basis for negative regulation of hypoxia-inducible factor-1 alpha by CITED2. Nat Struct Biol. 2003. 10:504–512.15. Hwang KK, Kim HS, Seo JU, et al. Apoptosis in the human neonatal vascular remodeling. Korean Circ J. 2001. 31:681–700.16. Choi YK, Kim JH, Kim WJ, et al. AKAP12 regulates human blood-retinal barrier formation by downregulation of hypoxiainducible factor-1 alpha. J Neurosci. 2007. 27:4472–4481.17. Chandrasekar B, Valente AJ, Freeman GL, Mahimainathan L, Mummidi S. Interleukin-18 induces human cardiac endothelial cell death via a novel signaling pathway involving NF-kappaB-dependent PTEN activation. Biochem Biophys Res Commun. 2006. 339:956–963.18. Ryan S, McNicholas WT, Taylor CT. A critical role for p38 map kinase in NF-kappaB signaling during intermittent hypoxia/reoxygenation. Biochem Biophys Res Commun. 2007. 355:728–733.19. DiDonato JA, Hayakawa M, Rothwarf DM, Zandi E, Karin M. A cytokine-responsive IkappaB kinase that activates the transcription factor NF-kappaB. Nature. 1997. 388:548–554.20. Nakamura N, Ramaswamy S, Vazquez F, Signoretti S, Loda M, Sellers WR. Forkhead transcription factors are critical effectors of cell death and cell cycle arrest downstream of PTEN. Mol Cell Biol. 2000. 20:8969–8982.21. Harfouche R, Hasséssian HM, Guo Y, et al. Mechanisms which mediate the antiapoptotic effects of angiopoietin-1 on endothelial cells. Microvasc Res. 2002. 64:135–147.22. Morris JB, Kenney B, Huynh H, Woodcock EA. Regulation of the proapoptotic factor FOXO1 (FKHR) in cardiomyocytes by growth factors and alpha1-adrenergic agonists. Endocrinology. 2005. 146:4370–4376.23. Lee HY, Chung JW, Youn SW, et al. Forkhead transcription factor FOXO3a is a negative regulator of angiogenic immediate early gene CYR61, leading to inhibition of vascular smooth muscle cell proliferation and neointimal hyperplasia. Circ Res. 2007. 100:372–380.24. Medema RH, Kops GJ, Bos JL, Burgering BM. AFX-like Forkhead transcription factors mediate cell-cycle regulation by Ras and PKB through p27kip1. Nature. 2000. 404:782–787.25. Takahashi K, Ito Y, Morikawa M, et al. Adenoviral-delivered angiopoietin-1 reduces the infarction and attenuates the progression of cardiac dysfunction in the rat model of acute myocardial infarction. Mol Ther. 2003. 8:584–592.26. Jung H, Gurunluoglu R, Scharpf J, Siemionow M. Adenovirus-mediated angiopoietin-1 gene therapy enhances skin flap survival. Microsurgery. 2003. 23:374–380.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Over-expression of PTEN Involved in Troglitazone-induced Apoptosis in Human Osteosarcoma Cells
- Hypoxia/reoxygenation Regulates Expression of Tumor Suppressor PTEN in Rheumatoid Synovial Fibroblasts
- The Role of Phosphatase and Tensin Homolog Deleted on Chromosome Ten (PTEN) in Lung Cancer
- The Antiapoptotic Effect of Vascular Endothelial Growth Factor and Angiopoietin-1 on Glomerular Endothelial Cells in Adriamycin or Cisplatin-induced Apoptosis
- Inhibition of Store-Operated Calcium Entry Protects Endothelial Progenitor Cells from Hâ‚‚Oâ‚‚-Induced Apoptosis