J Periodontal Implant Sci.
2013 Aug;43(4):191-197. 10.5051/jpis.2013.43.4.191.
Effect of quercetin on the production of nitric oxide in murine macrophages stimulated with lipopolysaccharide from Prevotella intermedia
- Affiliations
-
- 1Department of Periodontology, Institute of Translational Dental Sciences, Pusan National University School of Dentistry, Yangsan, Korea. sungjokim@pusan.ac.kr
- KMID: 2027815
- DOI: http://doi.org/10.5051/jpis.2013.43.4.191
Abstract
- PURPOSE
Nitric oxide (NO) is a short-lived bioactive molecule that is known to play an important role in the pathogenesis of periodontal disease. In the current study, we investigated the effect of the flavonoid quercetin on the production of NO in murine macrophages activated with lipopolysaccharide (LPS) from Prevotella intermedia, a pathogen related to inflammatory periodontal disease, and tried to elucidate the underlying mechanisms of action.
METHODS
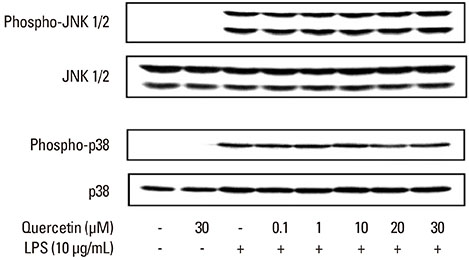
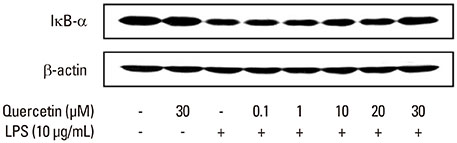
LPS was isolated from P. intermedia ATCC 25611 cells by the standard hot phenol-water method. The concentration of NO in cell culture supernatants was determined by measuring the accumulation of nitrite. Inducible NO synthase (iNOS) and heme oxygenase-1 (HO-1) protein expression, phosphorylation of c-Jun N-terminal kinase (JNK) and p38, inhibitory kappaB (IkappaB)-alpha degradation, and signal transducer and activator of transcription 1 (STAT1) phosphorylation were analyzed via immunoblotting.
RESULTS
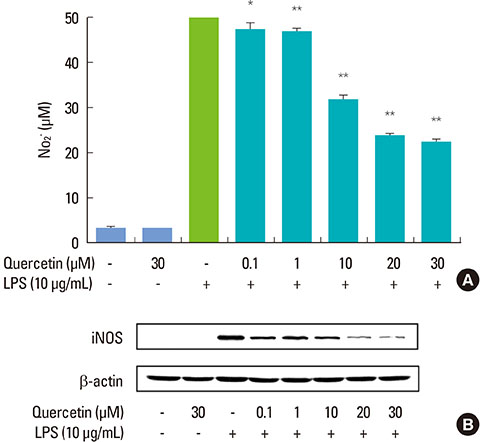
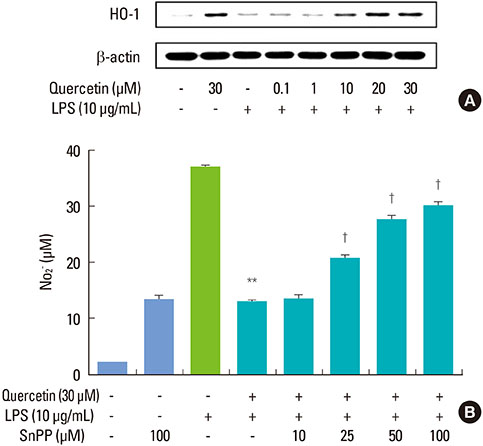
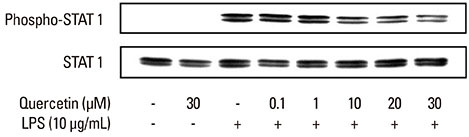
Quercetin significantly attenuated iNOS-derived NO production in RAW246.7 cells activated by P. intermedia LPS. In addition, quercetin induced HO-1 protein expression in cells activated with P. intermedia LPS. Tin protoporphyrin IX (SnPP), a competitive inhibitor of HO-1, abolished the inhibitory effect of quercetin on LPS-induced NO production. Quercetin did not affect the phosphorylation of JNK and p38 induced by P. intermedia LPS. The degradation of IkappaB-alpha induced by P. intermedia LPS was inhibited when the cells were treated with quercetin. Quercetin also inhibited LPS-induced STAT1 signaling.
CONCLUSIONS
Quercetin significantly inhibits iNOS-derived NO production in murine macrophages activated by P. intermedia LPS via anti-inflammatory HO-1 induction and inhibition of the nuclear factor-kappaB and STAT1 signaling pathways. Our study suggests that quercetin may contribute to the modulation of host-destructive responses mediated by NO and appears to have potential as a novel therapeutic agent for treating inflammatory periodontal disease.
MeSH Terms
-
Cell Culture Techniques
Heme Oxygenase-1
I-kappa B Proteins
JNK Mitogen-Activated Protein Kinases
Lipopolysaccharides
Macrophages
Metalloporphyrins
Nitric Oxide
Nitric Oxide Synthase
Periodontal Diseases
Phosphorylation
Prevotella
Prevotella intermedia
Protoporphyrins
Quercetin
STAT1 Transcription Factor
Tin
Heme Oxygenase-1
I-kappa B Proteins
JNK Mitogen-Activated Protein Kinases
Lipopolysaccharides
Metalloporphyrins
Nitric Oxide
Nitric Oxide Synthase
Protoporphyrins
Quercetin
STAT1 Transcription Factor
Tin
Figure
Reference
-
1. Williams RC. Periodontal disease. N Engl J Med. 1990; 322:373–382.
Article2. Teng YT, Taylor GW, Scannapieco F, Kinane DF, Curtis M, Beck JD, et al. Periodontal health and systemic disorders. J Can Dent Assoc. 2002; 68:188–192.3. Seymour GJ, Ford PJ, Cullinan MP, Leishman S, Yamazaki K. Relationship between periodontal infections and systemic disease. Clin Microbiol Infect. 2007; 13:Suppl 4. 3–10.
Article4. Tanner AC, Haffer C, Bratthall GT, Visconti RA, Socransky SS. A study of the bacteria associated with advancing periodontitis in man. J Clin Periodontol. 1979; 6:278–307.
Article5. Slots J, Bragd L, Wikstrom M, Dahlen G. The occurrence of Actinobacillus actinomycetemcomitans, Bacteroides gingivalis and Bacteroides intermedius in destructive periodontal disease in adults. J Clin Periodontol. 1986; 13:570–577.
Article6. Chung CP, Nisengard RJ, Slots J, Genco RJ. Bacterial IgG and IgM antibody titers in acute necrotizing ulcerative gingivitis. J Periodontol. 1983; 54:557–562.
Article7. Kornman KS, Loesche WJ. The subgingival microbial flora during pregnancy. J Periodontal Res. 1980; 15:111–122.
Article8. Hamada S, Takada H, Ogawa T, Fujiwara T, Mihara J. Lipopolysaccharides of oral anaerobes associated with chronic inflammation: chemical and immunomodulating properties. Int Rev Immunol. 1990; 6:247–261.
Article9. Kirikae T, Nitta T, Kirikae F, Suda Y, Kusumoto S, Qureshi N, et al. Lipopolysaccharides (LPS) of oral black-pigmented bacteria induce tumor necrosis factor production by LPS-refractory C3H/HeJ macrophages in a way different from that of Salmonella LPS. Infect Immun. 1999; 67:1736–1742.
Article10. Hashimoto M, Asai Y, Tamai R, Jinno T, Umatani K, Ogawa T. Chemical structure and immunobiological activity of lipid A from Prevotella intermedia ATCC 25611 lipopolysaccharide. FEBS Lett. 2003; 543:98–102.
Article11. Preshaw PM. Host response modulation in periodontics. Periodontol 2000. 2008; 48:92–110.
Article12. Reddy MS, Geurs NC, Gunsolley JC. Periodontal host modulation with antiproteinase, anti-inflammatory, and bone-sparing agents: a systematic review. Ann Periodontol. 2003; 8:12–37.
Article13. Sosroseno W, Barid I, Herminajeng E, Susilowati H. Nitric oxide production by a murine macrophage cell line (RAW264.7) stimulated with lipopolysaccharide from Actinobacillus actinomycetemcomitans. Oral Microbiol Immunol. 2002; 17:72–78.
Article14. Kim SJ, Ha MS, Choi EY, Choi JI, Choi IS. Prevotella intermedia lipopolysaccharide stimulates release of nitric oxide by inducing expression of inducible nitric oxide synthase. J Periodontal Res. 2004; 39:424–431.
Article15. Kim SJ, Ha MS, Choi EY, Choi JI, Choi IS. Nitric oxide production and inducible nitric oxide synthase expression induced by Prevotella nigrescens lipopolysaccharide. FEMS Immunol Med Microbiol. 2005; 01. 1. 43:51–58.
Article16. Kim SJ, Choi EY, Kim EG, Shin SH, Lee JY, Choi JI, et al. Prevotella intermedia lipopolysaccharide stimulates release of tumor necrosis factor-alpha through mitogen-activated protein kinase signaling pathways in monocyte-derived macrophages. FEMS Immunol Med Microbiol. 2007; 51:407–413.
Article17. Moncada S, Palmer RM, Higgs EA. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev. 1991; 43:109–142.18. Matejka M, Partyka L, Ulm C, Solar P, Sinzinger H. Nitric oxide synthesis is increased in periodontal disease. J Periodontal Res. 1998; 33:517–518.
Article19. Kendall HK, Haase HR, Li H, Xiao Y, Bartold PM. Nitric oxide synthase type-II is synthesized by human gingival tissue and cultured human gingival fibroblasts. J Periodontal Res. 2000; 35:194–200.
Article20. Lappin DF, Kjeldsen M, Sander L, Kinane DF. Inducible nitric oxide synthase expression in periodontitis. J Periodontal Res. 2000; 35:369–373.
Article21. Hirose M, Ishihara K, Saito A, Nakagawa T, Yamada S, Okuda K. Expression of cytokines and inducible nitric oxide synthase in inflamed gingival tissue. J Periodontol. 2001; 72:590–597.
Article22. Batista AC, Silva TA, Chun JH, Lara VS. Nitric oxide synthesis and severity of human periodontal disease. Oral Dis. 2002; 8:254–260.
Article23. Harwood M, Danielewska-Nikiel B, Borzelleca JF, Flamm GW, Williams GM, Lines TC. A critical review of the data related to the safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxic/carcinogenic properties. Food Chem Toxicol. 2007; 45:2179–2205.
Article24. Davis JM, Murphy EA, McClellan JL, Carmichael MD, Gangemi JD. Quercetin reduces susceptibility to influenza infection following stressful exercise. Am J Physiol Regul Integr Comp Physiol. 2008; 295:R505–R509.
Article25. Utesch D, Feige K, Dasenbrock J, Broschard TH, Harwood M, Danielewska-Nikiel B, et al. Evaluation of the potential in vivo genotoxicity of quercetin. Mutat Res. 2008; 654:38–44.
Article26. Choi EY, Jin JY, Lee JY, Choi JI, Choi IS, Kim SJ. Melatonin inhibits Prevotella intermedia lipopolysaccharide-induced production of nitric oxide and interleukin-6 in murine macrophages by suppressing NF-κB and STAT1 activity. J Pineal Res. 2011; 50:197–206.27. Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem. 1982; 126:131–138.
Article28. Otterbein LE, Soares MP, Yamashita K, Bach FH. Heme oxygenase-1: unleashing the protective properties of heme. Trends Immunol. 2003; 24:449–455.
Article29. Ryter SW, Alam J, Choi AM. Heme oxygenase-1/carbon monoxide: from basic science to therapeutic applications. Physiol Rev. 2006; 86:583–650.
Article30. Morse D, Choi AM. Heme oxygenase-1: the "emerging molecule" has arrived. Am J Respir Cell Mol Biol. 2002; 27:8–16.31. Poss KD, Tonegawa S. Reduced stress defense in heme oxygenase 1-deficient cells. Proc Natl Acad Sci U S A. 1997; 94:10925–10930.32. Otterbein LE, Bach FH, Alam J, Soares M, Tao Lu H, Wysk M, et al. Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nat Med. 2000; 6:422–428.
Article33. Morse D, Pischke SE, Zhou Z, Davis RJ, Flavell RA, Loop T, et al. Suppression of inflammatory cytokine production by carbon monoxide involves the JNK pathway and AP-1. J Biol Chem. 2003; 278:36993–36998.
Article34. Sarady-Andrews JK, Liu F, Gallo D, Nakao A, Overhaus M, Ollinger R, et al. Biliverdin administration protects against endotoxin-induced acute lung injury in rats. Am J Physiol Lung Cell Mol Physiol. 2005; 289:L1131–L1137.
Article35. Choi IS, Choi EY, Jin JY, Park HR, Choi JI, Kim SJ. Kaempferol inhibits P. intermedia lipopolysaccharide-induced production of nitric oxide through translational regulation in murine macrophages: critical role of heme oxygenase-1-mediated ROS reduction. J Periodontol. 2013; 84:545–555.
Article36. Baeuerle PA, Henkel T. Function and activation of NF-κB in the immune system. Annu Rev Immunol. 1994; 12:141–179.37. Karin M, Ben-Neriah Y. Phosphorylation meets ubiquitination: the control of NF-κB activity. Annu Rev Immunol. 2000; 18:621–663.
Article38. Li Q, Verma IM. NF-κB regulation in the immune system. Nat Rev Immunol. 2002; 2:725–734.
Article39. Gao JJ, Filla MB, Fultz MJ, Vogel SN, Russell SW, Murphy WJ. Autocrine/paracrine IFN-αβ mediates the lipopolysaccharide-induced activation of transcription factor Stat1α in mouse macrophages: pivotal role of Stat1alpha in induction of the inducible nitric oxide synthase gene. J Immunol. 1998; 161:4803–4810.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Chemical and Immunobiological Characterization of Lipopolysaccharides from Prevotella intermedia and Prevotella nigrescens
- Suppression of nitric oxide and interleukin-6 production by methanol extract of Sophorae Flos in macrophage cells
- The effect of high concentration of glucose on the production of proinflammatory cytokines and nitric oxide induced by lipopolysaccharides from periodontopathic bacteria
- Phosphatidylinositol-3-kinase Inhibitor Enhances Nitric Oxide Synthesis and Apoptosis in LPS-Stimulated Macrophages
- Interleukin-8 production and interleukin-8 mRNA expression induced by lipopolysaccharides from Prevotella intermedia and Prevotella nigrescens in monocyte-derived macrophages