Allergy Asthma Immunol Res.
2011 Jul;3(3):186-193. 10.4168/aair.2011.3.3.186.
Increased Expression and Role of Thymic Stromal Lymphopoietin in Nasal Polyposis
- Affiliations
-
- 1Department of Otolaryngology, Nippon Medical School, Tokyo, Japan. pawankar.ruby@gmail.com
- KMID: 1803324
- DOI: http://doi.org/10.4168/aair.2011.3.3.186
Abstract
- PURPOSE
Nasal polyposis is a chronic inflammatory disease of the upper airways often associated with asthma and characterized by markedly increased numbers of eosinophils, Th2 type lymphocytes, fibroblasts, goblet cells and mast cells. Previous studies have shown elevated levels of thymic stromal lymphopoietin (TSLP) in atopic diseases like asthma, atopic dermatitis and mainly in animal models of allergic rhinitis (AR). Here, we investigated the expression of TSLP in nasal polyps from atopics and non-atopics in comparison with the nasal mucosa and its potential role in nasal polyposis.
METHODS
Messenger RNA expression for TSLP, thymus and activation-regulated chemokine (TARC) and macrophage derived chemokine (MDC) in nasal polyps and nasal mucosa of atopics and non-atopics was analyzed by real time PCR. Immunoreactivity for TSLP in nasal polyps and in the nasal mucosa of patients with AR and non-allergic rhinitis (NAR) was analyzed by immunohistochemistry. Eosinophil counts was analyzed by Wright-Giemsa staining and nasal polyp tissue IgE, by ELISA.
RESULTS
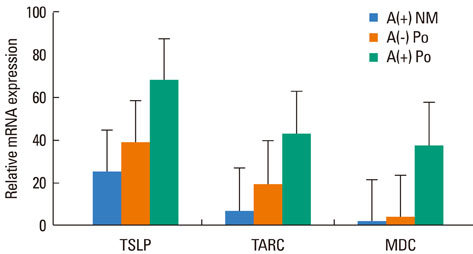
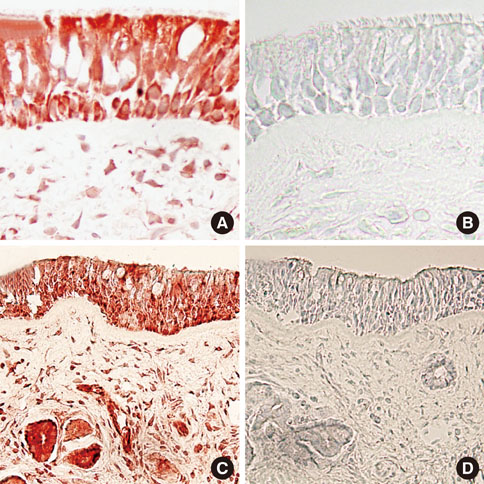
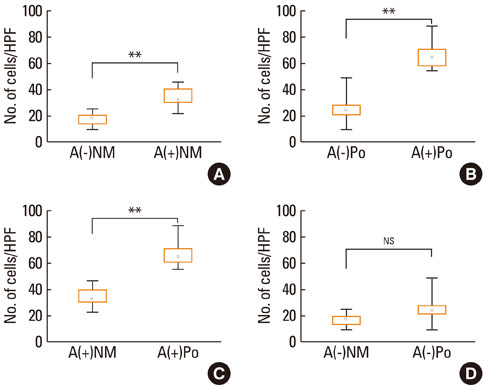
Messenger RNA expression for TSLP,TARC and MDC was markedly higher in nasal polyps as compared to the allergic nasal mucosa. Immunoreactivity for TSLP was detected in epithelial cells, endothelial cells, fibroblasts and inflammatory cells of the nasal mucosa and nasal polyps. The number of TSLP+ cells was significantly greater in the nasal mucosa of AR than NAR patients. The number of TSLP+ cells in nasal polyps from atopics was significantly greater than that of non-atopics and that in the allergic nasal mucosa. The number of TSLP+ cells correlated well with the number of eosinophils and the levels of IgE in nasal polyps.
CONCLUSIONS
The high expression of TSLP in nasal polyps and its strong correlation to eosinophils and IgE suggest a potential role for TSLP in the pathogenesis of nasal polyps by regulating the Th2 type and eosinophilic inflammation.
Keyword
MeSH Terms
-
Asthma
Chemokine CCL17
Chemokine CCL22
Cytokines
Dermatitis, Atopic
Endothelial Cells
Eosinophils
Epithelial Cells
Fibroblasts
Goblet Cells
Humans
Immunoglobulin E
Immunohistochemistry
Inflammation
Lymphocytes
Mast Cells
Models, Animal
Nasal Mucosa
Nasal Polyps
Real-Time Polymerase Chain Reaction
Rhinitis
Rhinitis, Allergic, Perennial
RNA, Messenger
Chemokine CCL17
Chemokine CCL22
Cytokines
Immunoglobulin E
RNA, Messenger
Figure
Cited by 1 articles
-
Overview on the pathomechanisms of allergic rhinitis
Ruby Pawankar, Sachiko Mori, Chika Ozu, Satoko Kimura
Asia Pac Allergy. 2011;1(3):157-167. doi: 10.5415/apallergy.2011.1.3.157.
Reference
-
1. Hamilos DL, Leung DY, Wood R, Cunningham L, Bean DK, Yasruel Z, Schotman E, Hamid Q. Evidence for distinct cytokine expression in allergic versus nonallergic chronic sinusitis. J Allergy Clin Immunol. 1995. 96:537–544.2. Pawankar R. Nasal polyposis: an update: editorial review. Curr Opin Allergy Clin Immunol. 2003. 3:1–6.3. Pawankar R, Lee KH, Nonaka M, Takizawa R. Role of mast cells and basophils in chronic rhinosinusitis. Clin Allergy Immunol. 2007. 20:93–101.4. Fan GK, Wang H, Takenaka H. Eosinophil infiltration and activation in nasal polyposis. Acta Otolaryngol. 2007. 127:521–526.5. Nonaka M, Ogihara N, Fukumoto A, Sakanushi A, Kusama K, Pawankar R, Yagi T. Combined stimulation with Poly(I:C), TNF-alpha and Th2 cytokines induces TARC production by human fibroblasts from the nose, bronchioles and lungs. Int Arch Allergy Immunol. 2010. 152:327–341.6. Nonaka M, Fukumoto A, Ogihara N, Sakanushi A, Pawankar R, Yagi T. Synergistic induction of thymic stromal lymphopoietin by tumor necrosis factor alpha and Th2 cytokine in nasal polyp fibroblasts. Am J Rhinol Allergy. 2010. 24:e14–e18.7. Toru H, Pawankar R, Ra C, Yata J, Nakahata T. Human mast cells produce IL-13 by high-affinity IgE receptor cross-linking: enhanced IL-13 production by IL-4-primed human mast cells. J Allergy Clin Immunol. 1998. 102:491–502.8. Quentmeier H, Drexler HG, Fleckenstein D, Zaborski M, Armstrong A, Sims JE, Lyman SD. Cloning of human thymic stromal lymphopoietin (TSLP) and signaling mechanisms leading to proliferation. Leukemia. 2001. 15:1286–1292.9. Sims JE, Williams DE, Morrissey PJ, Garka K, Foxworthe D, Price V, Friend SL, Farr A, Bedell MA, Jenkins NA, Copeland NG, Grabstein K, Paxton RJ. Molecular cloning and biological characterization of a novel murine lymphoid growth factor. J Exp Med. 2000. 192:671–680.10. Park LS, Martin U, Garka K, Gliniak B, Di Santo JP, Muller W, Largaespada DA, Copeland NG, Jenkins NA, Farr AG, Ziegler SF, Morrissey PJ, Paxton R, Sims JE. Cloning of the murine thymic stromal lymphopoietin (TSLP) receptor: Formation of a functional heteromeric complex requires interleukin 7 receptor. J Exp Med. 2000. 192:659–670.11. Huston DP, Liu YJ. Thymic stromal lymphopoietin:a potential therapeutic target for allergy and asthma. Curr Allergy Asthma Rep. 2006. 6:372–376.12. Holgate ST. The epithelium takes centre stage in asthma and atopic dermatitis. Trends Immunol. 2007. 28:248–251.13. Zhang K, Shan L, Rahman MS, Unruh H, Halayko AJ, Gounni AS. Constitutive and inducible thymic stromal lymphopoietin expression in human airway smooth muscle cells: role in chronic obstructive pulmonary disease. Am J Physiol Lung Cell Mol Physiol. 2007. 293:L375–L382.14. Liu YJ. Thymic stromal lymphopoietin and OX40 ligand pathway in the initiation of dendritic cell-mediated allergic inflammation. J Allergy Clin Immunol. 2007. 120:238–244.15. Reche PA, Soumelis V, Gorman DM, Clifford T, Liu M, Travis M, Zurawski SM, Johnston J, Liu YJ, Spits H, de Waal Malefyt R, Kastelein RA, Bazan JF. Human thymic stromal lymphopoietin preferentially stimulates myeloid cells. J Immunol. 2001. 167:336–343.16. Isaksen DE, Baumann H, Trobridge PA, Farr AG, Levin SD, Ziegler SF. Requirement for stat5 in thymic stromal lymphopoietin-mediated signal transduction. J Immunol. 1999. 163:5971–5977.17. Semlali A, Jacques E, Koussih L, Gounni AS, Chakir J. Thymic stromal lymphopoietin-induced human asthmatic airway epithelial cell proliferation through an IL-13-dependent pathway. J Allergy Clin Immunol. 2010. 125:844–850.18. Soumelis V, Reche PA, Kanzler H, Yuan W, Edward G, Homey B, Gilliet M, Ho S, Antonenko S, Lauerma A, Smith K, Gorman D, Zurawski S, Abrams J, Menon S, McClanahan T, de Waal-Malefyt Rd R, Bazan F, Kastelein RA, Liu YJ. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat Immunol. 2002. 3:673–680.19. Ying S, O'Connor B, Ratoff J, Meng Q, Fang C, Cousins D, Zhang G, Gu S, Gao Z, Shamji B, Edwards MJ, Lee TH, Corrigan CJ. Expression and cellular provenance of thymic stromal lymphopoietin and chemokines in patients with severe asthma and chronic obstructive pulmonary disease. J Immunol. 2008. 181:2790–2798.20. Zhu DD, Zhu XW, Jiang XD, Dong Z. Thymic stromal lymphopoietin expression is increased in nasal epithelial cells of patients with mugwort pollen sensitive-seasonal allergic rhinitis. Chin Med J (Engl). 2009. 122:2303–2307.21. Xu M, Guo Y. Expression of thymic stromal lymphopoietin in nasal mucosa of a mouse model with allergic rhinitis. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2009. 23:794–795. 79922. Fukumoto A, Nonaka M, Ogihara N, Pawankar R. Induction of TARC production by lipopolysaccharide and interleukin-4 in nasal fibroblasts. Int Arch Allergy Immunol. 2008. 145:291–297.23. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2ΔΔCT method. Methods. 2001. 25:402–408.24. Carmona R, Macías D, Guadix JA, Portillo V, Pérez-Pomares JM, Muñoz-Chápuli R. A simple technique of image analysis for specific nuclear immunolocalization of proteins. J Microsc. 2007. 225:96–99.25. Allakhverdi Z, Comeau MR, Jessup HK, Yoon BR, Brewer A, Chartier S, Paquette N, Ziegler SF, Sarfati M, Delespesse G. Thymic stromal lymphopoietin is released by human epithelial cells in response to microbes, trauma, or inflammation and potently activates mast cells. J Exp Med. 2007. 204:253–258.26. Fang C, Siew LQ, Corrigan CJ, Ying S. The role of thymic stromal lymphopoietin in allergic inflammation and chronic obstructive pulmonary disease. Arch Immunol Ther Exp (Warsz). 2010. 58:81–90.27. Matsuda A, Ebihara N, Yokoi N, Kawasaki S, Tanioka H, Inatomi T, de Waal Malefyt R, Hamuro J, Kinoshita S, Murakami A. Functional role of thymic stromal lymphopoietin in chronic allergic keratoconjunctivitis. Invest Ophthalmol Vis Sci. 2010. 51:151–155.28. Miyata M, Hatsushika K, Ando T, Shimokawa N, Ohnuma Y, Katoh R, Suto H, Ogawa H, Masuyama K, Nakao A. Mast cell regulation of epithelial TSLP expression plays an important role in the development of allergic rhinitis. Eur J Immunol. 2008. 38:1487–1492.29. Takizawa R, Pawankar R, Yamagishi S, Takenaka H, Yagi T. Increased expression of HLA-DR and CD86 in nasal epithelial cells in allergic rhinitics: antigen presentation to T cells and up-regulation by diesel exhaust particles. Clin Exp Allergy. 2007. 37:420–433.30. Al-Shami A, Spolski R, Kelly J, Keane-Myers A, Leonard WJ. A role for TSLP in the development of inflammation in an asthma model. J Exp Med. 2005. 202:829–839.31. Pawankar R, Yamagishi S, Takizawa R, Yagi T. Mast cell-IgE-and mast cell-structural cell interactions in allergic airway disease. Curr Drug Targets Inflamm Allergy. 2003. 2:303–312.32. Van Zele T, Gevaert P, Watelet JB, Claeys G, Holtappels G, Claeys C, van Cauwenberge P, Bachert C. Staphylococcus aureus colonization and IgE antibody formation to enterotoxins is increased in nasal polyposis. J Allergy Clin Immunol. 2004. 114:981–983.33. Suh KS, Park HS, Nahm DH, Kim YK, Lee YM, Park K. Role of IgG, IgA, and IgE antibodies in nasal polyp tissue: their relationships with eosinophilic infiltration and degranulation. J Korean Med Sci. 2002. 17:375–380.34. Pawankar R, Okuda M, Yssel H, Okumura K, Ra C. Nasal mast cells in perennial allergic rhinitics exhibit increased expression of the Fc epsilonRI, CD40L, IL-4, and IL-13, and can induce IgE synthesis in B cells. J Clin Invest. 1997. 99:1492–1499.35. Sheahan P, Ahn CN, Harvey RJ, Wise SK, Mulligan RM, Lathers DM, Schlosser RJ. Local IgE production in nonatopic nasal polyposis. J Otolaryngol Head Neck Surg. 2010. 39:45–51.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Alternaria Induces Production of Thymic Stromal Lymphopoietin in Nasal Fibroblasts Through Toll-like Receptor 2
- Epigenetic Regulation of Nasal Polyp Formation
- Blockade of thymic stromal lymphopoietin and CRTH2 attenuates airway inflammation in a murine model of allergic asthma
- The Role of Thymic Stromal Lymphopoietin (TSLP) in Glomerulonephritis
- Panonychus citri Can Induce T-helper Type 2 Immune Responses via the Release of Thymic Stromal Lymphopoietin and IL-4