J Nutr Health.
2018 Oct;51(5):379-385. 10.4163/jnh.2018.51.5.379.
Cellular zinc deficiency inhibits the mineralized nodule formation and downregulates bone-specific gene expression in osteoblastic MC3T3-E1 cells
- Affiliations
-
- 1Department of Food Science and Nutrition, Andong National University, Andong, Gyeongbuk 36729, South Korea. iskwun@andong.ac.kr
- KMID: 2427761
- DOI: http://doi.org/10.4163/jnh.2018.51.5.379
Abstract
- PURPOSE
Zinc (Zn) is an essential trace element for bone mineralization and osteoblast function. We examined the effects of Zn deficiency on osteoblast differentiation and mineralization in MC3T3-E1 cells.
METHODS
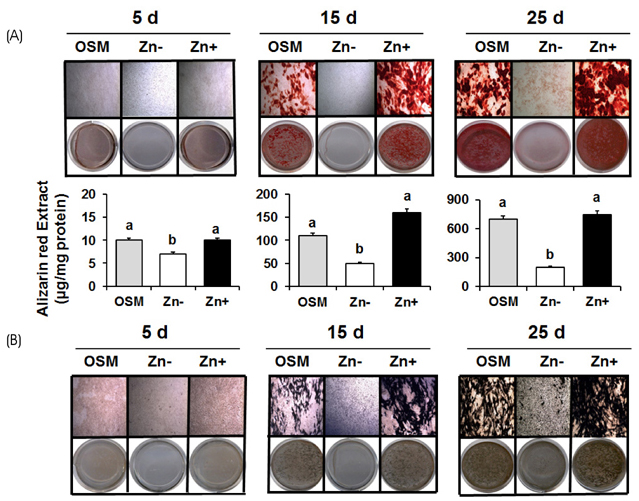
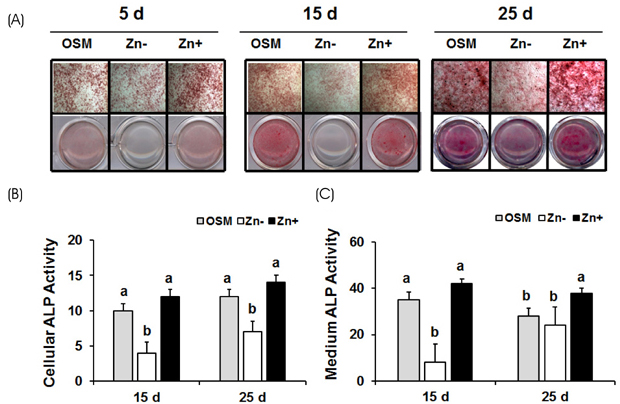
Osteoblastic MC3T3-E1 cells were cultured at concentration of 1 to 15 µM ZnCl2 (Zn− or Zn+) for 5, 15 and 25 days up to the calcification period. Extracellular matrix mineralization was detected by staining Ca and P deposits using Alizarin Red and von Kossa stain respectively, and alkaline phosphatase (ALP) activity was detected by ALP staining and colorimetric method.
RESULTS
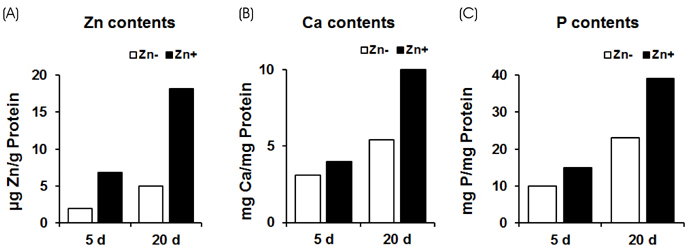
Extracellular matrix mineralization was decreased in Zn deficiency over 5, 15, and 25 days. Similarly, staining of ALP activity as the sign of an osteoblast differentiation, was also decreased by Zn deficiency over the same period. Interestingly, the gene expression of bone-related markers (ALP, PTHR; parathyroid hormone receptor, OPN; osteopontin, OC; osteocalcin and COLI; collagen type I), and bone-specific transcription factor Runx2 were downregulated by Zn deficiency for 5 or 15 days, however, this was restored at 25 days.
CONCLUSION
Our data suggests that Zn deficiency inhibits osteoblast differentiation by retarding bone marker gene expression and also inhibits bone mineralization by decreasing Ca/P deposition as well as ALP activity.
Keyword
MeSH Terms
-
Alkaline Phosphatase
Calcification, Physiologic
Collagen
Extracellular Matrix
Gene Expression*
Methods
Miners*
Osteoblasts*
Osteocalcin
Osteopontin
Receptor, Parathyroid Hormone, Type 1
Transcription Factors
Zinc*
Alkaline Phosphatase
Collagen
Osteocalcin
Osteopontin
Receptor, Parathyroid Hormone, Type 1
Transcription Factors
Zinc
Figure
Reference
-
1. Banerjee C, Javed A, Choi JY, Green J, Rosen V, van Wijnen AJ, Stein JL, Lian JB, Stein GS. Differential regulation of the two principal Runx2/Cbfa1 n-terminal isoforms in response to bone morphogenetic protein-2 during development of the osteoblast phenotype. Endocrinology. 2001; 142(9):4026–4039.
Article2. Nakashima K, Zhou X, Kunkel G, Zhang Z, Deng JM, Behringer RR, de Crombrugghe B. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell. 2002; 108(1):17–29.
Article3. Prasad AS. Zinc: an overview. Nutrition. 1995; 11:1 Suppl. 93–99.4. Leek JC, Vogler JB, Gershwin ME, Golub MS, Hurley LS, Hendrickx AG. Studies of marginal zinc deprivation in rhesus monkeys. V. Fetal and infant skeletal effects. Am J Clin Nutr. 1984; 40(6):1203–1212.
Article5. Yamaguchi M. Role of nutritional zinc in the prevention of osteoporosis. Mol Cell Biochem. 2010; 338(1-2):241–254.
Article6. Chowanadisai W, Kelleher SL, Lönnerdal B. Maternal zinc deficiency reduces NMDA receptor expression in neonatal rat brain, which persists into early adulthood. J Neurochem. 2005; 94(2):510–519.
Article7. Zhao N, Wang X, Zhang Y, Gu Q, Huang F, Zheng W, Li Z. Gestational zinc deficiency impairs humoral and cellular immune responses to hepatitis B vaccination in offspring mice. PLoS One. 2013; 8(9):e73461.
Article8. Salgueiro MJ, Zubillaga MB, Lysionek AE, Caro RA, Weill R, Boccio JR. The role of zinc in the growth and development of children. Nutrition. 2002; 18(6):510–519.
Article9. Angus RM, Sambrook PN, Pocock NA, Eisman JA. Dietary intake and bone mineral density. Bone Miner. 1988; 4(3):265–277.10. Kim JT, Baek SH, Lee SH, Park EK, Kim EC, Kwun IS, Shin HI. Zinc-deficient diet decreases fetal long bone growth through decreased bone matrix formation in mice. J Med Food. 2009; 12(1):118–123.
Article11. Yamaguchi M, Matsui T. Stimulatory effect of zinc-chelating dipeptide on deoxyribonucleic acid synthesis in osteoblastic MC3T3-E1 cells. Peptides. 1996; 17(7):1207–1211.
Article12. Alcantara EH, Lomeda RA, Feldmann J, Nixon GF, Beattie JH, Kwun IS. Zinc deprivation inhibits extracellular matrix calcification through decreased synthesis of matrix proteins in osteoblasts. Mol Nutr Food Res. 2011; 55(10):1552–1560.
Article13. Cho YE, Lomeda RA, Ryu SH, Sohn HY, Shin HI, Beattie JH, Kwun IS. Zinc deficiency negatively affects alkaline phosphatase and the concentration of Ca, Mg and P in rats. Nutr Res Pract. 2007; 1(2):113–119.
Article14. Cho YE, Kwun IS. Zinc upregulates bone-specific transcription factor Runx2 expression via BMP-2 signaling and Smad-1 phosphorylation in osteoblasts. J Nutr Health. 2018; 51(1):23–30.
Article15. Seo HJ, Cho YE, Kim T, Shin HI, Kwun IS. Zinc may increase bone formation through stimulating cell proliferation, alkaline phosphatase activity and collagen synthesis in osteoblastic MC3T3-E1 cells. Nutr Res Pract. 2010; 4(5):356–361.
Article16. Kwun IS, Cho YE, Lomeda RA, Shin HI, Choi JY, Kang YH, Beattie JH. Zinc deficiency suppresses matrix mineralization and retards osteogenesis transiently with catch-up possibly through Runx 2 modulation. Bone. 2010; 46(3):732–741.
Article17. Anderson HC. Matrix vesicles and calcification. Curr Rheumatol Rep. 2003; 5(3):222–226.
Article18. Anderson HC, Sipe JB, Hessle L, Dhanyamraju R, Atti E, Camacho NP, Millán JL. Impaired calcification around matrix vesicles of growth plate and bone in alkaline phosphatase-deficient mice. Am J Pathol. 2004; 164(3):841–847.
Article19. Golub EE, Harrison G, Taylor AG, Camper S, Shapiro IM. The role of alkaline phosphatase in cartilage mineralization. Bone Miner. 1992; 17(2):273–278.
Article20. Orimo H. The mechanism of mineralization and the role of alkaline phosphatase in health and disease. J Nippon Med Sch. 2010; 77(1):4–12.
Article21. Anderson HC. Molecular biology of matrix vesicles. Clin Orthop Relat Res. 1995; (314):266–280.
Article22. Ducy P, Zhang R, Geoffroy V, Ridall AL, Karsenty G. Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation. Cell. 1997; 89(5):747–754.
Article23. Karsenty G. Transcriptional control of skeletogenesis. Annu Rev Genomics Hum Genet. 2008; 9(1):183–196.
Article24. Franceschi RT, Iyer BS. Relationship between collagen synthesis and expression of the osteoblast phenotype in MC3T3-E1 cells. J Bone Miner Res. 1992; 7(2):235–246.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Zinc modulation of osterix in MC3T3-E1 cells
- Effects of Olibanum Extracts on the Activity and Differentiation of MC3T3-E1 Cells
- Zinc may increase bone formation through stimulating cell proliferation, alkaline phosphatase activity and collagen synthesis in osteoblastic MC3T3-E1 cells
- Glycyrrhiza uralensis (licorice) extracts increase cell proliferation and bone marker enzyme alkaline phosphatase activity in osteoblastic MC3T3-E1 cells
- Zinc upregulates bone-specific transcription factor Runx2 expression via BMP-2 signaling and Smad-1 phosphorylation in osteoblasts