J Nutr Health.
2018 Feb;51(1):23-30. 10.4163/jnh.2018.51.1.23.
Zinc upregulates bone-specific transcription factor Runx2 expression via BMP-2 signaling and Smad-1 phosphorylation in osteoblasts
- Affiliations
-
- 1Department of Food Science and Nutrition, Andong National University, 1375 Kyungdong Road, Andong, Kyungbook 36633, South Korea. iskwun@andong.ac.kr
- KMID: 2410322
- DOI: http://doi.org/10.4163/jnh.2018.51.1.23
Abstract
- PURPOSE
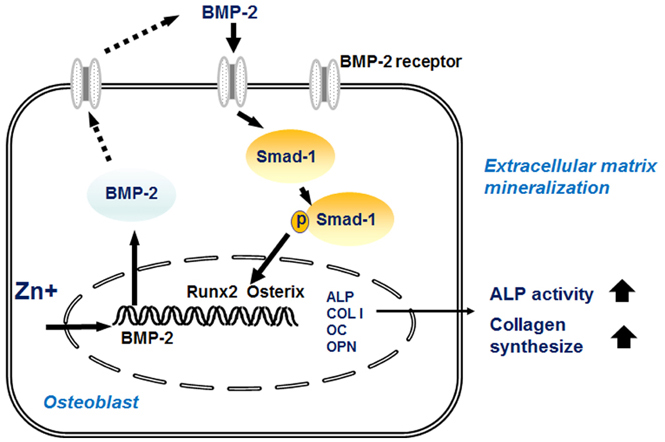
Runx2 (runt-related transcription factor 2), a bone-specific transcription factor, is a key regulator of osteoblast differentiation and its expression is induced by the activation of BMP-2 signaling. This study examined whether zinc modulates BMP-2 signaling and therefore stimulates Runx2 and osteoblast differentiation gene expression.
METHODS
Two osteoblastic MC3T3-E1 cell lines (subclones 4 as a high osteoblast differentiation and subclone 24 as a low osteoblastic differentiation) were cultured in an osteogenic medium (OSM) as the normal control, Zn− (1 µM Zn) or Zn+ (15 µM Zn) for 24 h. The genes and proteins for BMP-2 signaling (BMP-2, Smad-1/p-Smad-1), transcription factors (Runx2, osterix), and osteoblast differentiation marker proteins were assessed.
RESULTS
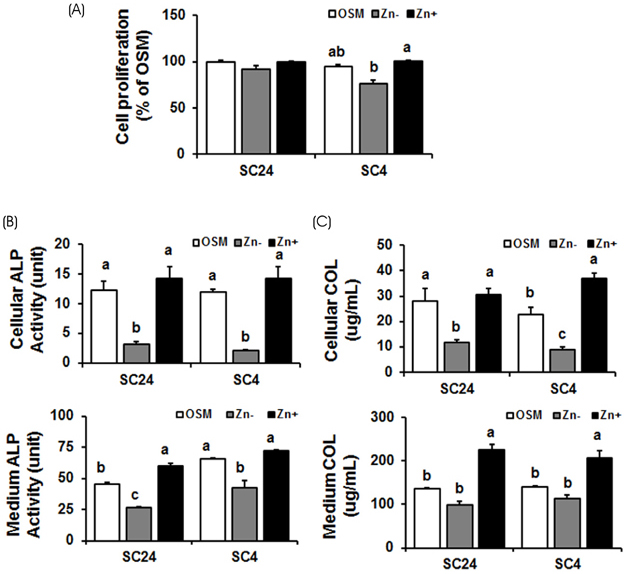
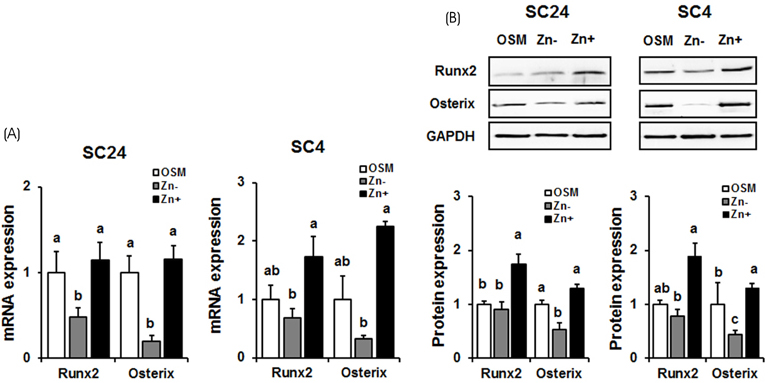
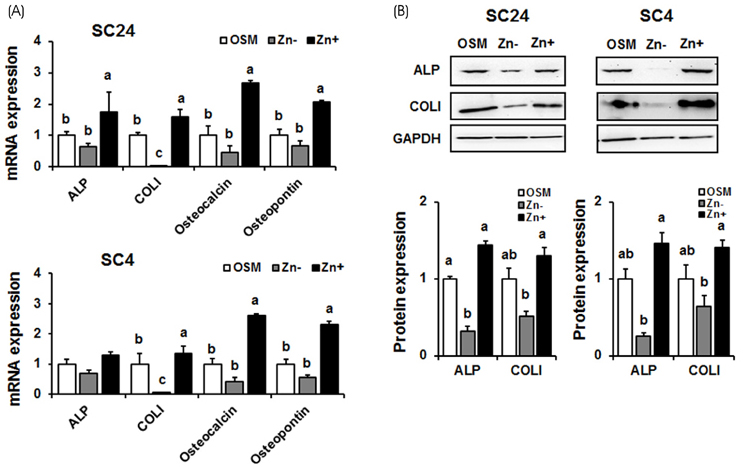
In both cell lines, BMP-2 mRAN and protein expression and extracellular BMP-2 secretion all decreased in Zn−. The expression of Smad-1 (downstream regulator of BMP-2 signaling) and p-Smad-1 (phosphorylated Smad-1) also downregulated in Zn−. Furthermore, the expression of the bone-specific transcription factors, Runx2 and osterix, decreased in Zn−, which might be due to the decreased BMP-2 expression and Smad-1 activation (p-Smad-1) by Zn−, because Runx2 and osterix both are downstream in BMP-2 signaling. Bone marker gene expression, such as alkaline phosphatase (ALP), collagen type I (COLI), osteocalcin, and osteopontin were also downregulated in Zn−.
CONCLUSION
The results suggest that a zinc deficiency in osteoblasts suppresses the BMP-2 signaling pathway via the suppression of Smad-1 activation, and this suppressed BMP-2 signaling can cause poor osteoblast differentiation.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Cellular zinc deficiency inhibits the mineralized nodule formation and downregulates bone-specific gene expression in osteoblastic MC3T3-E1 cells
Young-Eun Cho, In-Sook Kwun
J Nutr Health. 2018;51(5):379-385. doi: 10.4163/jnh.2018.51.5.379.Glycyrrhiza uralensis (licorice) extracts increase cell proliferation and bone marker enzyme alkaline phosphatase activity in osteoblastic MC3T3-E1 cells∗
Young-Eun Cho, In-Sook Kwun
J Nutr Health. 2018;51(4):316-322. doi: 10.4163/jnh.2018.51.4.316.
Reference
-
1. Coleman JE. Zinc proteins: enzymes, storage proteins, transcription factors, and replication proteins. Annu Rev Biochem. 1992; 61:897–946.
Article2. Maret W. Zinc in cellular regulation: the nature and significance of “Zinc Signals”. Int J Mol Sci. 2017; 18(11):2285–2296.
Article3. Tuerk MJ, Fazel N. Zinc deficiency. Curr Opin Gastroenterol. 2009; 25(2):136–143.
Article4. Chabosseau P, Rutter GA. Zinc and diabetes. Arch Biochem Biophys. 2016; 611:79–85.
Article5. Kim JH, Jeon J, Shin M, Won Y, Lee M, Kwak JS, Lee G, Rhee J, Ryu JH, Chun CH, Chun JS. Regulation of the catabolic cascade in osteoarthritis by the Zinc-ZIP8-MTF1 Axis. Cell. 2014; 156(4):730–743.
Article6. da Cunha Ferreira RM, Marquiegui IM, Elizaga IV. Teratogenicity of zinc deficiency in the rat: study of the fetal skeleton. Teratology. 1989; 39(2):181–194.
Article7. Hsieh HS, Navia JM. Zinc deficiency and bone formation in guinea pig alveolar implants. J Nutr. 1980; 110(8):1581–1588.
Article8. Hurley LS. Teratogenic aspects of manganese, zinc, and copper nutrition. Physiol Rev. 1981; 61(2):249–295.
Article9. Leek JC, Vogler JB, Gershwin ME, Golub MS, Hurley LS, Hendrickx AG. Studies of marginal zinc deprivation in rhesus monkeys. V. Fetal and infant skeletal effects. Am J Clin Nutr. 1984; 40(6):1203–1212.10. Oner G, Bhaumick B, Bala RM. Effect of zinc deficiency on serum somatomedin levels and skeletal growth in young rats. Endocrinology. 1984; 114(5):1860–1863.
Article11. Holloway WR, Collier FM, Herbst RE, Hodge JM, Nicholson GC. Osteoblast-mediated effects of zinc on isolated rat osteoclasts: inhibition of bone resorption and enhancement of osteoclast number. Bone. 1996; 19(2):137–142.
Article12. Togari A, Arakawa S, Arai M, Matsumoto S. Alteration of in vitro bone metabolism and tooth formation by zinc. Gen Pharmacol. 1993; 24(5):1133–1140.
Article13. Alcantara EH, Lomeda RA, Feldmann J, Nixon GF, Beattie JH, Kwun IS. Zinc deprivation inhibits extracellular matrix calcification through decreased synthesis of matrix proteins in osteoblasts. Mol Nutr Food Res. 2011; 55(10):1552–1560.
Article14. Cho YE, Lomeda RA, Ryu SH, Lee JH, Beattie JH, Kwun IS. Cellular Zn depletion by metal ion chelators (TPEN, DTPA and chelex resin) and its application to osteoblastic MC3T3-E1 cells. Nutr Res Pract. 2007; 1(1):29–35.
Article15. Cho YE, Lomeda RA, Ryu SH, Sohn HY, Shin HI, Beattie JH, Kwun IS. Zinc deficiency negatively affects alkaline phosphatase and the concentration of Ca, Mg and P in rats. Nutr Res Pract. 2007; 1(2):113–119.
Article16. Kim JT, Baek SH, Lee SH, Park EK, Kim EC, Kwun IS, Shin HI. Zinc-deficient diet decreases fetal long bone growth through decreased bone matrix formation in mice. J Med Food. 2009; 12(1):118–123.
Article17. Kwun IS, Cho YE, Lomeda RA, Shin HI, Choi JY, Kang YH, Beattie JH. Zinc deficiency suppresses matrix mineralization and retards osteogenesis transiently with catch-up possibly through Runx 2 modulation. Bone. 2010; 46(3):732–741.
Article18. Seo HJ, Cho YE, Kim T, Shin HI, Kwun IS. Zinc may increase bone formation through stimulating cell proliferation, alkaline phosphatase activity and collagen synthesis in osteoblastic MC3T3-E1 cells. Nutr Res Pract. 2010; 4(5):356–361.
Article19. Yamaguchi M, Goto M, Uchiyama S, Nakagawa T. Effect of zinc on gene expression in osteoblastic MC3T3-E1 cells: enhancement of Runx2, OPG, and regucalcin mRNA expressions. Mol Cell Biochem. 2008; 312(1-2):157–166.
Article20. Ducy P, Zhang R, Geoffroy V, Ridall AL, Karsenty G. Osf2/Cbfa1: a transcriptional activator of osteoblast differentiation. Cell. 1997; 89(5):747–754.
Article21. Lee MH, Kim YJ, Kim HJ, Park HD, Kang AR, Kyung HM, Sung JH, Wozney JM, Kim HJ, Ryoo HM. BMP-2-induced Runx2 expression is mediated by Dlx5, and TGF-beta 1 opposes the BMP-2-induced osteoblast differentiation by suppression of Dlx5 expression. J Biol Chem. 2003; 278(36):34387–34394.22. Lee MH, Kwon TG, Park HS, Wozney JM, Ryoo HM. BMP-2-induced Osterix expression is mediated by Dlx5 but is independent of Runx2. Biochem Biophys Res Commun. 2003; 309(3):689–694.
Article23. Ryoo HM, Lee MH, Kim YJ. Critical molecular switches involved in BMP-2-induced osteogenic differentiation of mesenchymal cells. Gene. 2006; 366(1):51–57.
Article24. Suzuki T, Katsumata S, Matsuzaki H, Suzuki K. A short-term zinc-deficient diet decreases bone formation through down-regulated BMP2 in rat bone. Biosci Biotechnol Biochem. 2016; 80(7):1433–1435.
Article25. Murshed M, Harmey D, Millan JS, McKee MD, Karsenty G. Unique coexpression in osteblasts of boadly expressed genes accounts for the apatial restrictionof ECM minralization to bone. Genes Dev. 2005; 19(9):1093–1104.26. Katagiri T, Takahashi N. Regulatory mechanisms of osteoblast and osteoclast differentiation. Oral Dis. 2002; 8(3):147–159.
Article27. Komori T. Regulation of osteoblast differentiation by Runx2. Adv Exp Med Biol. 2010; 658:43–49.
Article28. Golub EE, Harrison G, Taylor AG, Camper S, Shapiro IM. The role of alkaline phosphatase in cartilage mineralization. Bone Miner. 1992; 17(2):273–278.
Article29. Orimo H. The mechanism of mineralization and the role of alkaline phosphatase in health and disease. J Nippon Med Sch. 2010; 77(1):4–12.
Article30. Yamaguchi M. Role of nutritional zinc in the prevention of osteoporosis. Mol Cell Biochem. 2010; 338(1-2):241–254.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Function of runx2 and osterix in osteogenesis and teeth
- Bone Morphogenetic Protein-2 Desensitizes MC3T3-E1 Osteoblastic Cells to Estrogen Through Transcriptional Downregulation of Estrogen Receptor 1
- The synergistic regulatory effect of Runx2 and MEF transcription factors on osteoblast differentiation markers
- Melanin extract from Gallus gallus domesticus promotes proliferation and differentiation of osteoblastic MG-63 cells via bone morphogenetic protein-2 signaling
- Temporal and Spatial Expression of Bone Morphogenetic Protein -2 and -4 mRNA in Distraction Osteogenesis and Fracture Healing