Nutr Res Pract.
2016 Dec;10(6):575-582. 10.4162/nrp.2016.10.6.575.
High maysin corn silk extract reduces body weight and fat deposition in C57BL/6J mice fed high-fat diets
- Affiliations
-
- 1Department of Food Science and Nutrition, Dankook University, 152, Juljeon-ro, Suji-gu, Yonin-si, Gyeonggi 16890, Korea. wkkim@dankook.ac.kr
- 2Crop Foundation Division National Institute of Crop Science, Jeonbuk 55365, Korea.
- 3Department of Food Engineering, Dankook University, Chungnam 31116, Korea.
- KMID: 2359613
- DOI: http://doi.org/10.4162/nrp.2016.10.6.575
Abstract
- BACKGROUNG/OBJECTIVES: The study was performed to investigate the effects and mechanisms of action of high maysin corn silk extract on body weight and fat deposition in experimental animals.
MATERIALS/METHODS
A total of 30 male C57BL/6J mice, 4-weeks-old, were purchased and divided into three groups by weight using a randomized block design. The normal-fat (NF) group received 7% fat (diet weight basis), the high-fat (HF) group received 25% fat and 0.5% cholesterol, and the high-fat corn silk (HFCS) group received high-fat diet and high maysin corn silk extract at 100 mg/kg body weight through daily oral administration. Body weight and body fat were measured, and mRNA expression levels of proteins involved in adipocyte differentiation, fat accumulation, fat synthesis, lipolysis, and fat oxidation in adipose tissue and the liver were measured.
RESULTS
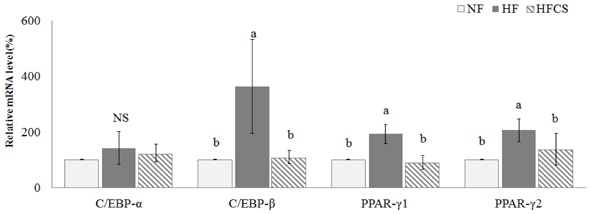
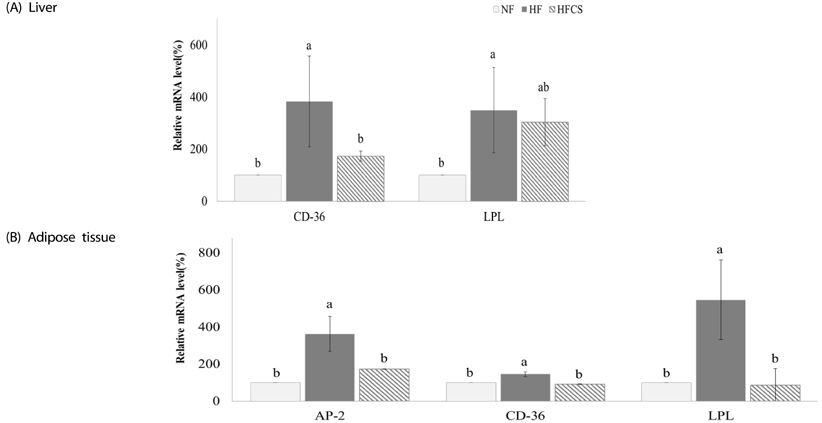
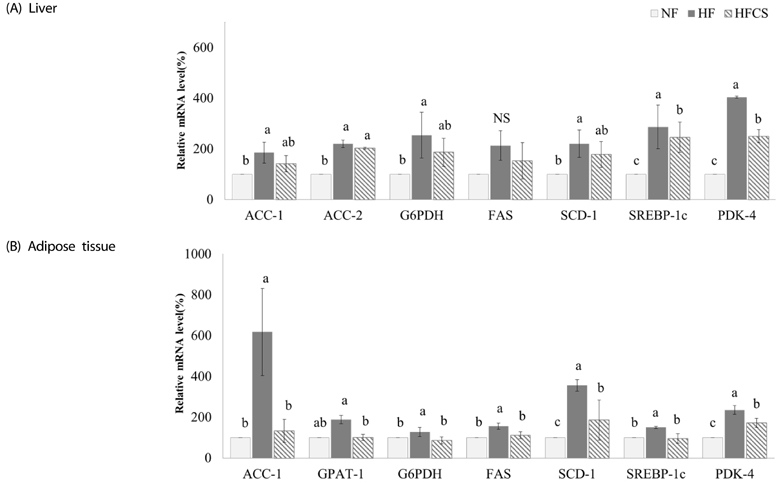
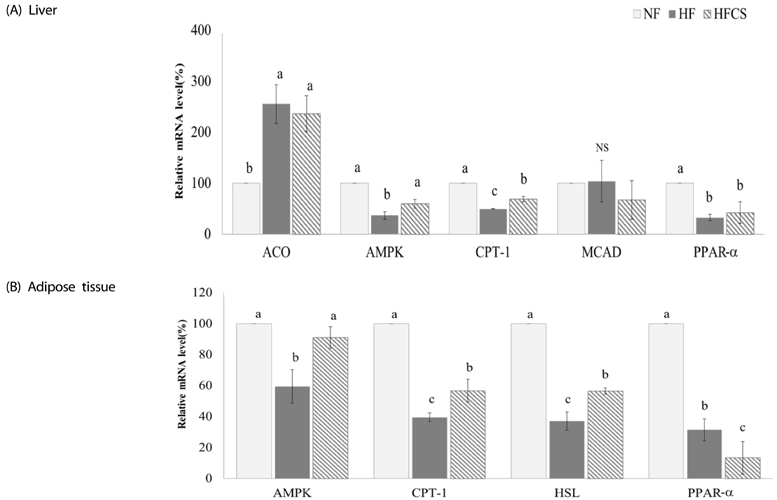
After experimental diet intake for 8 weeks, body weight was significantly lower in the HFCS group compared to the HF group (P < 0.05), and kidney fat and epididymal fat pad weights were significantly lower in the HFCS group compared to the HF group (P < 0.05). In the HFCS group, CCAAT/enhancer binding protein-β, peroxisome proliferator-activated receptor-γ1 (PPAR-γ1), and PPAR-γ2 mRNA expression levels were significantly reduced (P < 0.05) in the epididymal fat pad, whereas cluster of differentiation 36, lipoprotein lipase, acetyl-CoA carboxylase-1, sterol regulatory element binding protein-1c, pyruvate dehydrogenase kinase, isozyme-4, glucose-6-phosphate dehydrogenase, and stearoyl-CoA desaturase-1 mRNA expression levels were significantly decreased in liver and adipose tissues (P < 0.05). In the HFCS group, mRNA expression levels of AMP-activated protein kinase, hormone-sensitive lipase, and carnitine palmitoyltransferase-1 were elevated (P < 0.05).
CONCLUSIONS
It can be concluded that high maysin corn silk extract inhibits expression of genes involved in adipocyte differentiation, fat accumulation, and fat synthesis as well as promotes expression of genes involved in lipolysis and fat oxidation, further inhibiting body fat accumulation and body weight elevation in experimental animals.
Keyword
MeSH Terms
-
Acetyl Coenzyme A
Adipocytes
Adipose Tissue
Administration, Oral
AMP-Activated Protein Kinases
Animals
Body Weight*
Carnitine
Cholesterol
Diet
Diet, High-Fat*
Glucosephosphate Dehydrogenase
Humans
Kidney
Lipolysis
Lipoprotein Lipase
Liver
Male
Mice*
Oxidoreductases
Peroxisomes
Phosphotransferases
Pyruvic Acid
RNA, Messenger
Silk*
Sterol Esterase
Weights and Measures
Zea mays*
AMP-Activated Protein Kinases
Acetyl Coenzyme A
Carnitine
Cholesterol
Glucosephosphate Dehydrogenase
Lipoprotein Lipase
Oxidoreductases
Phosphotransferases
Pyruvic Acid
RNA, Messenger
Silk
Sterol Esterase
Figure
Cited by 1 articles
-
Corn silk extract improves benign prostatic hyperplasia in experimental rat model
So Ra Kim, Ae Wha Ha, Hyun Ji Choi, Sun Lim Kim, Hyeon Jung Kang, Myung Hwan Kim, Woo Kyoung Kim
Nutr Res Pract. 2017;11(5):373-380. doi: 10.4162/nrp.2017.11.5.373.
Reference
-
1. Velazquez DV, Xavier HS, Batista JE, de Castro-Chaves C. Zea mays L. extracts modify glomerular function and potassium urinary excretion in conscious rats. Phytomedicine. 2005; 12:363–369.
Article2. Maksimović Z, Malencić D, Kovacević N. Polyphenol contents and antioxidant activity of Maydis stigma extracts. Bioresour Technol. 2005; 96:873–877.
Article3. Hasanudin K, Hashim P, Mustafa S. Corn silk (Stigma maydis) in healthcare: a phytochemical and pharmacological review. Molecules. 2012; 17:9697–9715.
Article4. Grases F, March JG, Ramis M, Costa-Bauzá A. The influence of Zea mays on urinary risk factors for kidney stones in rats. Phytother Res. 1993; 7:146–149.
Article5. Barnes J, Anderson LA, Phillipson JD. Herbal Medicines: a Guide for Health-care Professionals. 2nd ed. London: Pharmaceutical Press;1996.6. George GO, Idu FK. Corn silk aqueous extracts and intraocular pressure of systemic and non-systemic hypertensive subjects. Clin Exp Optom. 2015; 98:138–149.
Article7. Du J, Xu QT, Gao XH. Effects of stigma maydis polysaccharide on gastrointestinal movement. Zhongguo Zhong Yao Za Zhi. 2007; 32:1203–1206.8. Yang J, Li X, Xue Y, Wang N, Liu W. Anti-hepatoma activity and mechanism of corn silk polysaccharides in H22 tumor-bearing mice. Int J Biol Macromol. 2014; 64:276–280.
Article9. Maksimović ZA, Kovacević N. Preliminary assay on the antioxidative activity of Maydis stigma extracts. Fitoterapia. 2003; 74:144–147.
Article10. Zeringue HJ Jr. Identification and effects of maize silk volatiles on cultures of Aspergillus flavus. J Agric Food Chem. 2000; 48:921–925.
Article11. Habtemariam S. Extract of corn silk (stigma of Zea mays) inhibits the tumour necrosis factor-alpha- and bacterial lipopolysaccharide-induced cell adhesion and ICAM-1 expression. Planta Med. 1998; 64:314–318.
Article12. Wang GQ, Xu T, Bu XM, Liu BY. Anti-inflammation effects of corn silk in a rat model of carrageenin-induced pleurisy. Inflammation. 2012; 35:822–827.
Article13. Guo J, Liu T, Han L, Liu Y. The effects of corn silk on glycaemic metabolism. Nutr Metab (Lond). 2009; 6:47.
Article14. Guo BZ, Zhang ZJ, Butrón A, Widstrom NW, Snook ME, Lynch RE, Plaisted D. Lost P1 allele in sh2 sweet corn: quantitative effects of p1 and a1 genes on concentrations of maysin, apimaysin, methoxymaysin, and chlorogenic acid in maize silk. J Econ Entomol. 2004; 97:2117–2126.
Article15. Lee EA, Byrne PF, McMullen MD, Snook ME, Wiseman BR, Widstrom NW, Coe EH. Genetic mechanisms underlying apimaysin and maysin synthesis and corn earworm antibiosis in maize (Zea mays L.). Genetics. 1998; 149:1997–2006.
Article16. Snook ME, Widstrom NW, Gueldner RC. Reversed-phase high-performance liquid chromatographic procedure for the determination of maysin in corn silks. J Chromatogr A. 1989; 477:439–447.
Article17. Kim SL, Snook ME, Lee JO. Radical scavenging activity and cytotoxicity of maysin(C-glycosylflavone) isolated from silks of Zea mays L. Korean J Crop Sci. 2003; 48:392–396.18. Choi DJ, Kim SL, Choi JW, Park YI. Neuroprotective effects of corn silk maysin via inhibition of H2O2-induced apoptotic cell death in SK-N-MC cells. Life Sci. 2014; 109:57–64.
Article19. Lee J, Kim SL, Lee S, Chung MJ, Park YI. Immunostimulating activity of maysin isolated from corn silk in murine RAW 264.7 macrophages. BMB Rep. 2014; 47:382–387.
Article20. World Health Organization. Obesity and overweight [Internet]. Geneva: World Health Organization;cited 2016 April 20. Available from: http://www.who.int/mediacentre/factsheets/fs311/en/.21. Ministry of Health and Welfare, Korea Centers for Disease Control and Prevention. Korea Health Statistics 2014: Korea National Health and Nutrition Examination Survey (KNHANES VI-2). Sejong: Korea Centers for Disease Control and Prevention;2015.22. Bray GA. Medical consequences of obesity. J Clin Endocrinol Metab. 2004; 89:2583–2589.
Article23. Peeters A, Barendregt JJ, Willekens F, Mackenbach JP, Al Mamun A, Bonneux L. NEDCOM, the Netherlands Epidemiology and Demography Compression of Morbidity Research Group. Obesity in adulthood and its consequences for life expectancy: a life-table analysis. Ann Intern Med. 2003; 138:24–32.
Article24. Wang C, Chan JS, Ren L, Yan JH. Obesity reduces cognitive and motor functions across the lifespan. Neural Plast. 2016; 2016:2473081.
Article25. Liu Q, Yuan B, Lo KA, Patterson HC, Sun Y, Lodish HF. Adiponectin regulates expression of hepatic genes critical for glucose and lipid metabolism. Proc Natl Acad Sci U S A. 2012; 109:14568–14573.
Article26. Nishimura J, Masaki T, Arakawa M, Seike M, Yoshimatsu H. Isoleucine prevents the accumulation of tissue triglycerides and upregulates the expression of PPARalpha and uncoupling protein in diet-induced obese mice. J Nutr. 2010; 140:496–500.
Article27. Thampy KG. Formation of malonyl coenzyme A in rat heart. Identification and purification of an isozyme of A carboxylase from rat heart. J Biol Chem. 1989; 264:17631–17634.28. Kim HJ, Miyazaki M, Man WC, Ntambi JM. Sterol regulatory element-binding proteins (SREBPs) as regulators of lipid metabolism: polyunsaturated fatty acids oppose cholesterol-mediated induction of SREBP-1 maturation. Ann N Y Acad Sci. 2002; 967:34–42.
Article29. Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, Wu M, Ventre J, Doebber T, Fujii N, Musi N, Hirshman MF, Goodyear LJ, Moller DE. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest. 2001; 108:1167–1174.
Article30. Min OJ, Sharma BR, Park CM, Rhyu DY. Effect of myadis stigma water extract on adipogenesis and blood glucose in 3T3-L1 adipocytes and db/db mice. Korean J Pharmacogn. 2011; 42:201–208.31. Reeves PG. Components of the AIN-93 diets as improvements in the AIN-76A diet. J Nutr. 1997; 127:838S–841S.
Article32. Kim SL, Kim MJ, Lee YY, Jung GH, Son BY, Lee JS, Kwon YU, Park YI. Isolation and identification of flavonoids from corn silk. Korean J Crop Sci. 2014; 59:435–444.
Article33. Kim SL, Jung TW. Maysin and other flavonoid contents in corn silks. Korean J Breed. 2001; 33:338–343.34. Ha AW, Kim WK, Kim JH, Kang NE. The supplementation effects of peanut sprout on reduction of abdominal fat and health indices in overweight and obese women. Nutr Res Pract. 2015; 9:249–255.
Article35. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001; 25:402–408.
Article36. Li Y, Qin G, Liu J, Mao L, Zhang Z, Shang J. Adipose tissue regulates hepatic cholesterol metabolism via adiponectin. Life Sci. 2014; 118:27–33.
Article37. Kadowaki T, Yamauchi T, Kubota N, Hara K, Ueki K, Tobe K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J Clin Invest. 2006; 116:1784–1792.
Article38. Yamauchi T, Kamon J, Waki H, Imai Y, Shimozawa N, Hioki K, Uchida S, Ito Y, Takakuwa K, Matsui J, Takata M, Eto K, Terauchi Y, Komeda K, Tsunoda M, Murakami K, Ohnishi Y, Naitoh T, Yamamura K, Ueyama Y, Froguel P, Kimura S, Nagai R, Kadowaki T. Globular adiponectin protected ob/ob mice from diabetes and ApoE-deficient mice from atherosclerosis. J Biol Chem. 2003; 278:2461–2468.
Article39. Floyd ZE, Wang ZQ, Kilroy G, Cefalu WT. Modulation of peroxisome proliferator-activated receptor gamma stability and transcriptional activity in adipocytes by resveratrol. Metabolism. 2008; 57:S32–S38.40. Larter CZ, Yeh MM, Van Rooyen DM, Teoh NC, Brooling J, Hou JY, Williams J, Clyne M, Nolan CJ, Farrell GC. Roles of adipose restriction and metabolic factors in progression of steatosis to steatohepatitis in obese, diabetic mice. J Gastroenterol Hepatol. 2009; 24:1658–1668.
Article41. Lee MS, Kim IH, Kim CT, Kim Y. Reduction of body weight by dietary garlic is associated with an increase in uncoupling protein mRNA expression and activation of AMP-activated protein kinase in diet-induced obese mice. J Nutr. 2011; 141:1947–1953.
Article42. O'Neill HM, Holloway GP, Steinberg GR. AMPK regulation of fatty acid metabolism and mitochondrial biogenesis: implications for obesity. Mol Cell Endocrinol. 2013; 366:135–151.43. Berger JJ, Barnard RJ. Effect of diet on fat cell size and hormone-sensitive lipase activity. J Appl Physiol (1985). 1999; 87:227–232.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Corn silk extract improves cholesterol metabolism in C57BL/6J mouse fed high-fat diets
- Effects of Liquid Culture of Coriolus Versicolor on Lipid Metabolism and Enzyme Activities in Rats Fed High Fat Diet
- Grape seed extract (Vitis vinifera) partially reverses high fat diet-induced obesity in C57BL/6J mice
- Effects of Liquid Culture of Agaricus blazei Murill on Lipid Metabolism and Enzyme Activities in Rats Fed High Fat Diet
- Effects of corn gluten hydrolyzates, branched chain amino acids, and leucine on body weight reduction in obese rats induced by a high fat diet