Korean J Hematol.
2005 Jun;40(2):75-81. 10.5045/kjh.2005.40.2.75.
Apoptosis Induction by Curcumin in Human Myelogenous Leukemia Cell Lines
- Affiliations
-
- 1Department of Internal Medicine, Konkuk University College of Medicine, Seoul, Korea.
- 2Cancer Research Institute, Seoul, Korea.
- 3Clinical Research Institute, Seoul National University Hospital, Seoul, Korea.
- 4Department of Internal Medicine, College of Medicine, Seoul National University, Seoul, Korea. ssysmc@snu.ac.kr
- 5Cancer Research Center, Samsung Biomedical Research Institute, Seoul, Korea.
- KMID: 2252341
- DOI: http://doi.org/10.5045/kjh.2005.40.2.75
Abstract
- BACKGROUND
Curcumin, a naturally occurring biologically active compound extracted from rhizomes of Curcuma species, has been shown to possess potent anti-inflammatory, anti-tumor and anti-oxidative properties. The effects and possible mechanism of this agent were investigated on 2 human myelogenous leukemic cell lines.
METHODS
K562 and KG-1 cells were the two cell lines selected. The MTT assay and flow cytometry were used to assess the cytotoxicity and for cell cycle analysis, respectively. The protein expressions were analyzed by Western blotting; the caspase activity was also checked.
RESULTS
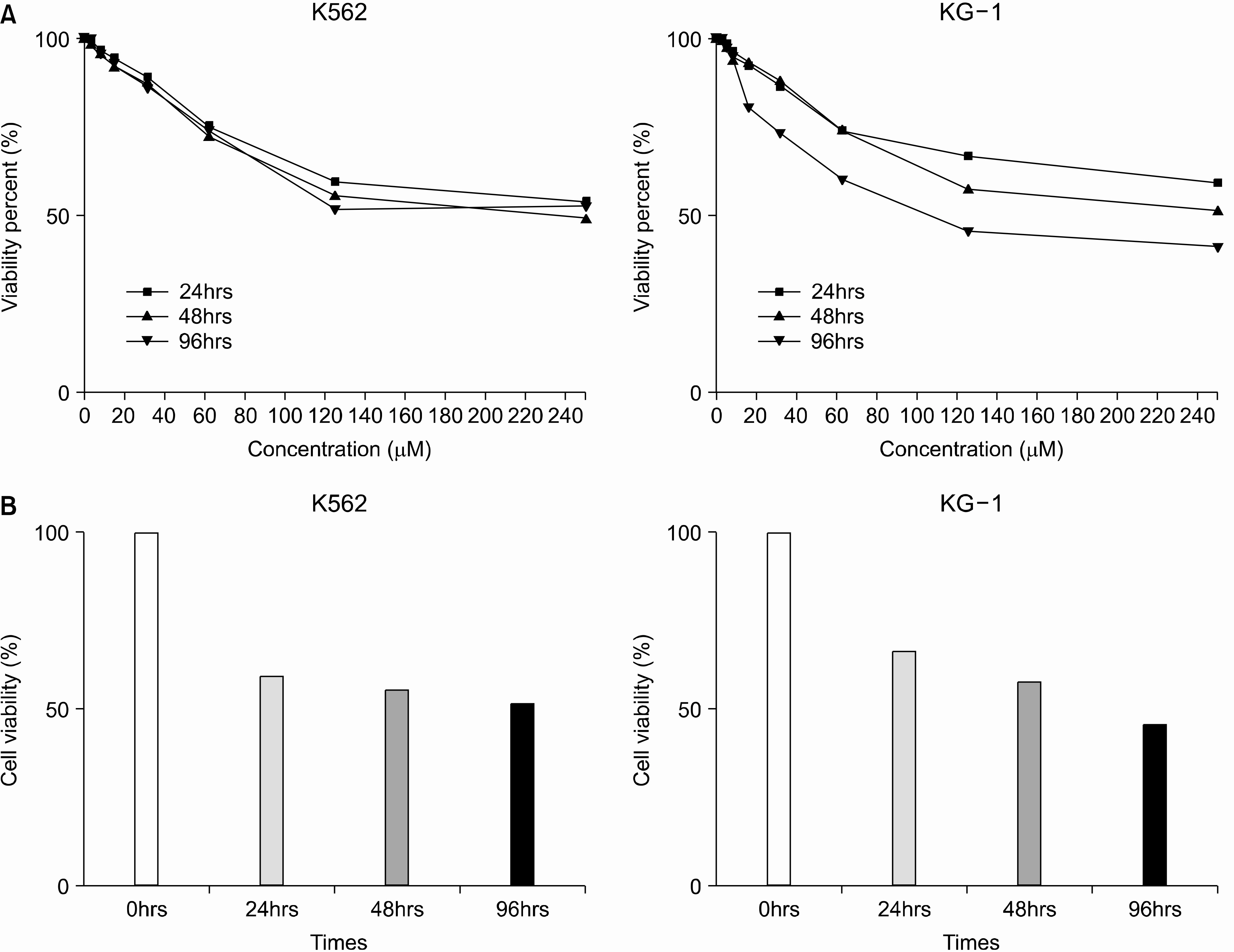
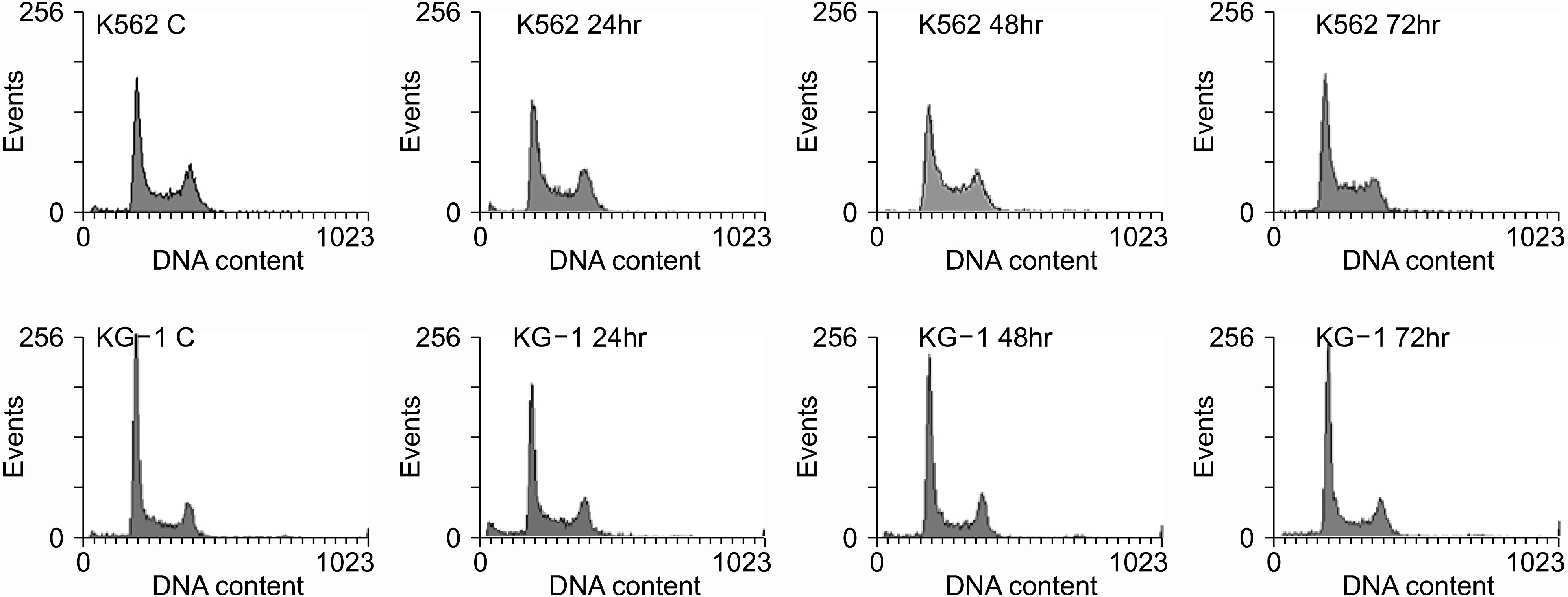
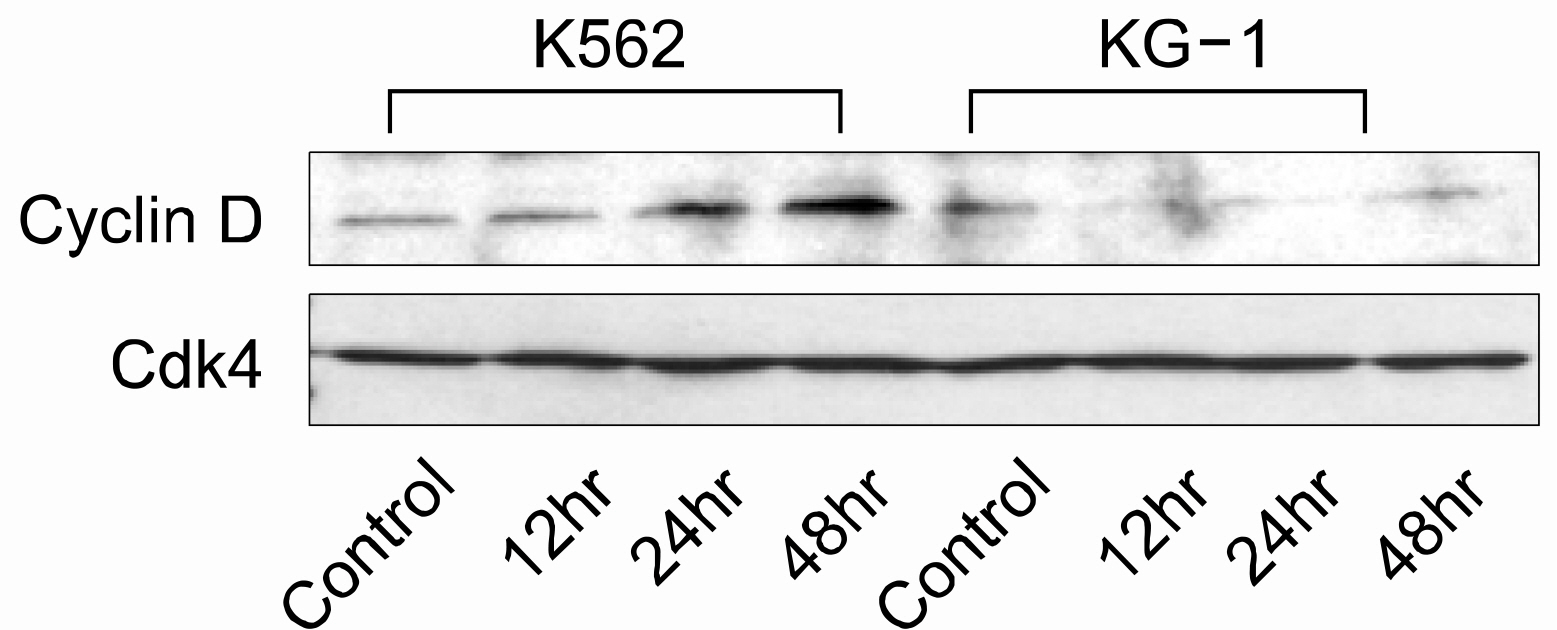
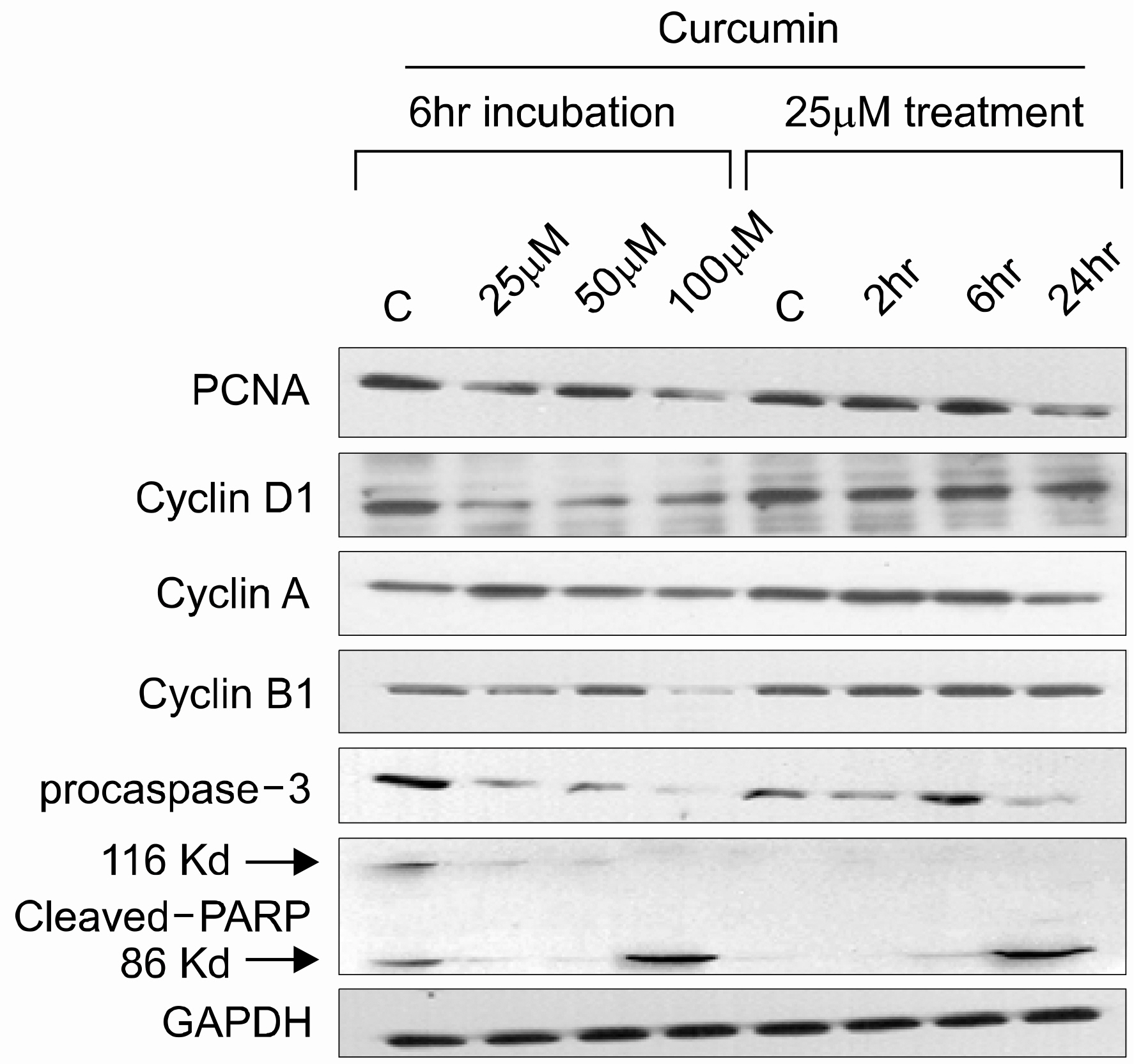
Both cell lines showed dose-dependent susceptibility to curcumin, and the cell cycle analysis showed an increased sub-G1 phase in the KG-1 cells. In the K562 cell, curcumin down regulated the expressions of PCNA (proliferating cell nuclear antigen) and cyclins D1 and B1. The expression of Akt was also down-regulated, but caspase-3 was activated to induce cleaved PARP (polyadenosine ribose polymerase) and apoptosis. However, the expression of phospho-Erk was unaffected. Co-treatment of cyclosporin A (CsA) with curcumin resulted in an attenuation of apoptosis in the K562 cells, implying curcumin-induced apoptosis is dependent on the release of cytochrome c from the mitochondria.
CONCLUSION
Curcumin induced cell cycle arrest and apoptosis in both human myelogenous leukemic cell lines, with the apoptosis appearing to be dependent on the release of cytochrome c from the mitochondria.
Keyword
MeSH Terms
-
Apoptosis*
Blotting, Western
Caspase 3
Cell Cycle
Cell Cycle Checkpoints
Cell Line*
Curcuma
Curcumin*
Cyclins
Cyclosporine
Cytochromes c
Flow Cytometry
Humans*
K562 Cells
Leukemia, Myeloid*
Mitochondria
Proliferating Cell Nuclear Antigen
Rhizome
Ribose
Caspase 3
Curcumin
Cyclins
Cyclosporine
Cytochromes c
Proliferating Cell Nuclear Antigen
Ribose
Figure
Reference
-
1). Liao S, Lin J, Dang MT, et al. Growth suppression of hamster flank organs by topical application of catechins, ali zarin, curcumin, and myristoleic acid. Arch Dermatol Res. 2001; 293:200–5.2). Ammon HP, Wahl MA. Pharmacology of Curcuma longa. Planta Med. 1991; 57:1–7.3). Kim KH, Park HY, Nam JH, et al. The inhibitory effect of curcumin on the growth of human colon cancer cells (HT-29, WiDr) in vitro. Korean J Gastroenterol. 2005; 45:277–84.4). Notarbartolo M, Poma P, Perri D, Dusonchet L, Cer-vello M, D'Alessandro N. Antitumor effects of curcumin, alone or in combination with cisplatin or doxorubicin, on human hepatic cancer cells. Analysis of their possible relationship to changes in NF-κB activation levels and in IAP gene expression. Cancer Lett. 2005; 224:53–65.5). Sun M, Yang Y, Li H, et al. The effect of curcumin on bladder cancer cell line EJ in vitro. Zhong Yao Cai. 2004; 27:848–50.6). Zhang J, Qi H, Wu C. Research of anti-proliferation of curcumin on A549 human lung cancer cells and its mechanism. Zhong Yao Cai. 2004; 27:923–7.7). Duvoix A, Morceau F, Schnekenburger M, et al. Cur-eumin-induced cell death in two leukemia cell lines: K562 and Jurkat. Ann N Y Acad Sci. 2003; 1010:389–92.8). Han SS, Keum YS, Seo HJ, Surh YJ. Curcumin suppresses activation of NF-kappaB and AP-1 induced by phorbol ester in cultured human promyelocytic leukemia cells. J Biochem Mol Biol. 2002; 35:337–42.9). Lin JK. Suppression of protein kinase C and nuclear oncogene expression as possible action mechanisms of cancer chemoprevention by curcumin. Arch Pharm Res. 2004; 27:683–92.
Article10). Korutla L, Kumar R. Inhibitory effect of curcumin on epidermal growth factor receptor kinase activity in A431 cells. Biochim Biophys Acta. 1994; 1224:597–600.
Article11). Lin JK, Pan MH, Lin-Shiau SY. Recent studies on the biofunctions and biotransformations of curcumin. Biofactors. 2000; 13:153–8.
Article12). Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC. Cleavage of poly (ADP-ribose) polymerase by a proteinase with properties like ICE. Nature. 1994; 371:346–7.13). Hansson MJ, Persson T, Friberg H, et al. Powerful cyclosporin inhibition of calcium-induced permeability transition in brain mitochondria. Brain Res. 2003; 960:99–111.
Article14). Ghribi O, DeWitt DA, Forbes MS, Arad A, Herman MM, Savory J. Cyclosporin A inhibits Al-induced cytochrome c release from mitochondria in aged rabbits. J Alzheimers Dis. 2001; 3:387–91.
Article15). Sporbert A, Domaing P, Leonhardt H, Cardoso MC. PCNA acts as a stationary loading platform for transiently interacting Okazaki fragment maturation proteins. Nucleic Acids Res. 2005; 33:3521–8.
Article16). Andreu EJ, Lledo E, Poch E, et al. BCR-ABL induces the expression of Skp2 through the PI3K pathway to promote p27Kip1 degradation and proliferation of chronic myelogenous leukemia cells. Cancer Res. 2005; 65:3264–72.
Article17). Kwon YK, Jun JM, Shin SW, Cho JW, Suh SI. Curcumin decreases cell proliferation rates through BTG2-mediated cyclin D1 down-regulation in U937 cells. Int J Oncol. 2005; 26:1597–603.
Article18). Chen A, Xu J. Activation of PPAR{gamma} by curcumin inhibits Moser cell growth and mediates suppression of gene expression of cyclin D1 and EGFR. Am J Physiol Gastrointest Liver Physiol. 2005; 288:G447–56.19). Chang F, Lee JT, Navolanic PM, et al. Involvement of PI3K/Akt pathway in cell cycle progression, apoptosis, and neoplastic transformation: a target for cancer chemotherapy. Leukemia. 2003; 17:590–603.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Curcumin on Apoptosis in SW480 Human Colon Cancer Cell Line
- Selective Effects of Curcumin on CdSe/ZnS Quantum-dot-induced Phototoxicity Using UVA Irradiation in Normal Human Lymphocytes and Leukemia Cells
- Induction of Apoptosis and Inhibition of Cellular Proliferation in Aspirin-treated SNU-668 Human Gastric Adenocarcinoma Cell Lines
- Cellular Effect of Curcumin and Citral Combination on Breast Cancer Cells: Induction of Apoptosis and Cell Cycle Arrest
- Mechanism Underlying Curcumin-induced Apoptosis and Cell Cycle Arrest on SCC25 Human Tongue Squamous Cell Carcinoma Cell Line