Korean Circ J.
2008 May;38(5):264-269. 10.4070/kcj.2008.38.5.264.
Effects of Cardiotrophin-1 on Adriamycin-Induced Apoptosis in H9c2 Cardiomyoblasts
- Affiliations
-
- 1Department of Medicine, Sungkyunkwan University School of Medicine, Cardiac and Vascular Center, Samsung Medical Center, Seoul, Korea. esjeon@smc.samsung.co.kr
- KMID: 2225788
- DOI: http://doi.org/10.4070/kcj.2008.38.5.264
Abstract
-
BACKGROUND AND OBJECTIVES: Adriamycin (doxorubicin, ADR) is a highly effective anti-neoplastic drug, but its clinical use is limited by its adverse side effects on the heart. Cardiotrophin (CT-1), a potent cardiac survival factor, is capable of inhibiting apoptosis in cardiac myocytes. The aim of this study was to investigate the cyto-protective effects of CT-1 against ADR-induced apoptosis in vitro.
MATERIALS AND METHODS
We determined a reasonable ADR concentration for inducing cell death by utilizing a cell survival test performed in a dose-dependent manner. To determine the requirements for apoptosis in ADR-treated cardiac myocytes (H9c2 cells), we examined the effect of CT-1 on survival and apoptotic changes using a cell counting kit (CCK), RT-PCR, and Western blotting.
RESULTS
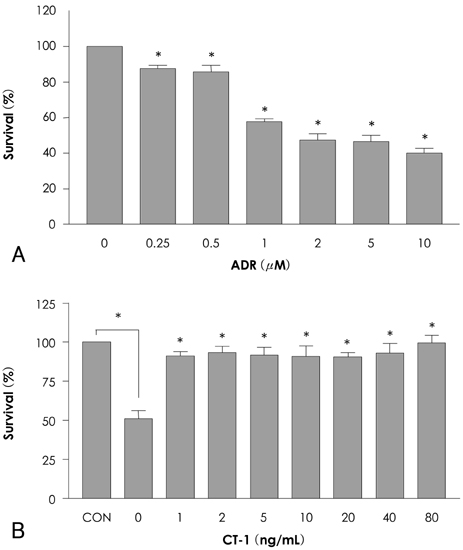
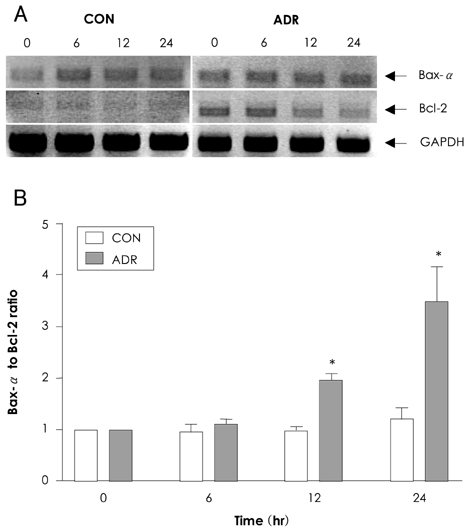
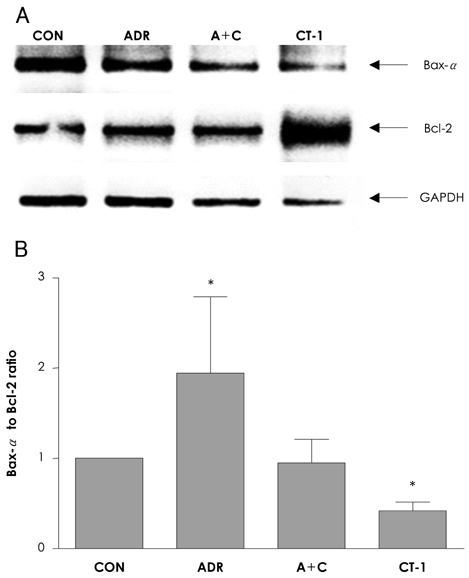
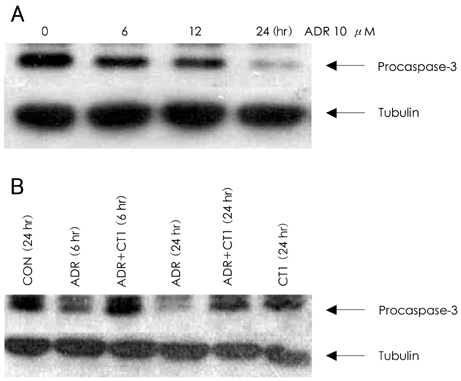
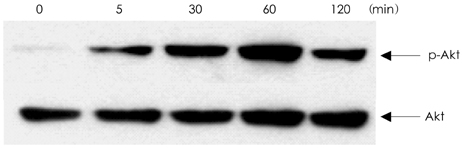
In analyzing cell survival as determined by CCK, ADR-induced cell death was found to occur in a dose-dependent manner (50% death at 24 hours after 2 micrometer of ADR), and ADR was shown to decrease procaspase-3. On RT-PCR, expression of Bax-alpha mRNA increased and Bcl-2 decreased during the 24 hours after ADR treatment. Consequently, the ratio of Bax-alpha/Bcl-2 mRNA peaked at 24 hours after ADR treatment. In contrast, CT-1 effectively attenuated the ADR-induced cell death in a dose-dependent manner. The changes in Bax-alpha and Bcl-2 mRNA expression after ADR treatment were reversed by CT-1 (1 ng/mL) treatment. The protein levels of procaspase-3 decreased after ADR treatment, an effect which was reversed by CT-1 treatment. Akt phosphorylation was also increased by CT-1, demonstrating that CT-1 inhibited apoptosis induced by ADR.
CONCLUSION
These data demonstrated that ADR-induced apoptosis of cardiomyocytes can be prevented by CT-1; therefore, it may be possible to use CT-1 as a cardioprotective agent during ADR chemotherapy in patients with cancer.
Keyword
MeSH Terms
Figure
Reference
-
1. Singal PK, Iliskovic N. Doxorubicin-induced cardiomyopathy. N Engl J Med. 1998. 339:900–905.2. Buzdar AU, Marcus C, Smith TL, Blumenschein GR. Early and delayed cardiotoxicity of doxorubicin. Cancer. 1985. 55:2761–2765.3. Doroshow JH. Effect of anthracycline antibiotics on oxygen radical formation in rat heart. Cancer Res. 1983. 43:460–472.4. Rajagopalan S, Politi PM, Sinha BK, Myers CE. Adriamycin-induced free radical formation in the perfused rat heart: implications for cardiotoxicity. Cancer Res. 1988. 48:4766–4769.5. Papadopoulou LC, Theophilidis G, Thomopoulos GN, Tsiftsoglou AS. Structural and functional impairment of mitochondria in adriamycin-induced cardiomyopathy in mice: suppression of cytochrome c oxidase II gene expression. Biochem Pharmacol. 1999. 57:481–489.6. Mijares A, Lopez JR. L-carnitine prevents increase in diastolic [Ca2+] induced by doxorubicin in cardiac cells. Eur J Pharmacol. 2001. 425:117–120.7. Mimnaugh EG, Trush MA, Gram TE. Enhancement of rat heart microsomal lipid peroxidation following doxorubicin treatment in vivo. Cancer Treat Rep. 1983. 67:731–733.8. Myers CE, McGuire WP, Liss RH, Ifrim I, Grotzinger K, Young RC. Adriamycin: the role of lipid peroxidation in cardiac toxicity and tumor response. Science. 1977. 197:165–167.9. Potmesil M, Israel M, Silber R. Two mechanisms of adriamycin-DNA interaction in L1210 cells. Biochem Pharmacol. 1984. 33:3137–3142.10. Tewey KM, Rowe TC, Yang L, Halligan BD, Liu LF. Adriamycin-induced DNA damage mediated by mammalian DNA topoisomerase II. Science. 1984. 226:466–468.11. Lampidis TJ, Johnson LV, Israel M. Effects of Adriamycin on rat heart cells in culture: increased accumulation and nucleoli fragmentation in cardiac muscle vs non-muscle cells. J Mol Cell Cardiol. 1981. 13:913–924.12. Goldenberg GJ, Wang H, Blair GW. Resistance to adriamycin: relationship of cytotoxicity to drug uptake and DNA single- and double-strand breakage in cloned cell lines of adriamycin-sensitive and -resistant P388 leukemia. Cancer Res. 1986. 46:2978–2983.13. Latchman DS. Cardiotrophin-1: a novel cytokine and its effects in the heart and other tissues. Pharmacol Ther. 2000. 85:29–37.14. Sheng Z, Knowlton K, Chen J, Hoshijima M, Brown JH, Chien KR. Cardiotrophin 1 (CT-1) inhibition of cardiac myocyte apoptosis via a mitogen-activated protein kinase-dependent pathway: divergence from downstream CT-1 signals for myocardial cell hypertrophy. J Biol Chem. 1997. 272:5783–5791.15. Kunisada K, Negoro S, Tone E, et al. Signal transducer and activator of transcription 3 in the heart transduces not only a hypertrophic signal but a protective signal against doxorubicin-induced cardiomyopathy. Proc Natl Acad Sci USA. 2000. 97:315–319.16. Lim BK, Choi JH, Jeon ES, et al. Virus receptor trap neutralizes coxackievirus in experimental murine viral myocarditis. Cardiovasc Res. 2006. 71:517–526.17. Kim JM, Lim BK, Jeon ES, et al. TNFR-Fc fusion protein expressed by in vivo electroporation improves survival rates and myocardial injury in coxackievirus induced murine myocarditis. Biochem Biophys Res Commun. 2006. 344:765–771.18. Wang L, Ma W, Markovic R, Chen JW, Wang PH. Regulation of cardiomyocyte apoptotic signaling by insulin-like growth factor I. Circ Res. 1998. 83:516–522.19. Jeon MH, Youn HJ, Lee JH. Adriamycin induced apoptosis of H9c2 cardiomycytes via a caspase-independent pathway. Korean Circ J. 2004. 34:76–83.20. Nakamura T, Ueda Y, Juan Y, Katsuda S, Takahashi H, Koh E. Fas-mediated apoptosis in adriamycin-induced cardiomyopathy in rats: In vivo study. Circulation. 2000. 102:572–578.21. Kumar D, Kirshenbaum LA, Li T, Danelisen I, Singal PK. Apoptosis in adriamycin cardiomyopathy and its modulation by probucol. Antioxid Redox Signal. 2001. 3:135–145.22. Park SW, Seo HS, Kim JW, et al. Effect of human Bcl-2 gene expression on the peripheral atherosclerotic lesions of apolipoprotein E-deficient mouse. Korean Circ J. 2005. 35:725–733.23. Park CS, Youn HJ, Cho EJ, et al. Cardioprotective effect of IGF-1 in mouse with adriamycin-induced cardiomyopathy. Korean Circ J. 2002. 32:1116–1123.24. Sung JD, Jeon ES. Pathogenesis of myocardial cell death and effect of cardiotrophin-1 in coxackieviral myocarditis. Korean J Cardiovasc Dis. 2002. 3:34–42.25. Lopez N, Diez J, Fortuno MA. Characterization of the protective effects of cardiotrophin-1 against non-ischemic death stimuli in adult cardiomyocytes. Cytokine. 2005. 30:282–292.26. Pennica D, King KL, Shaw KJ, et al. Expression cloning of cardiotrophin1, a cytokine that induces cardiac myocyte hypertrophy. Proc Natl Acad Sci USA. 1995. 92:1142–1146.27. Kuwahara K, Saito Y, Kishimoto I, et al. Cardiotrophin-1 phosphorylates akt and BAD, and prolongs cell survival via a PI3K-dependent pathway in cardiac myocytes. J Mol Cell Cardiol. 2000. 32:1385–1394.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Adriamycin Induced Apoptosis of H9c2 Cardiomyocytes via a Caspase-Independent Pathway
- Modulation of the caveolin-3 and Akt status in caveolae by insulin resistance in H9c2 cardiomyoblasts
- The Role of Apoptosis in Adriamycin Induced Cardiotoxicity and Preventive Effect of L-carnitine in Rat
- Effects of H2O2 on Cardiac Rate and Apoptosis of Primary Rat Cardiomyoblasts in in Vitro Culture Systems
- Effect of Calcium Channel Blocker on Primary Rat Cardiomyoblasts and Cardiac Rate in Vitro