Ann Dermatol.
2009 Nov;21(4):364-368. 10.5021/ad.2009.21.4.364.
Reduced Mitochondrial Properties in Putative Progenitor/Stem Cells of Human Keratinocytes
- Affiliations
-
- 1Department of Dermatology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 2Department of Nanopharmaceutical and Life Sciences and Department of Physiology, College of Medicine, Kyung Hee University, Seoul, Korea.
- 3Department of Otolaryngology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. sykim2@amc.seoul.kr
- KMID: 2156498
- DOI: http://doi.org/10.5021/ad.2009.21.4.364
Abstract
- BACKGROUND
The characterization of progenitor/keratinocyte stem cells (KSC) remains an unachieved goal. A previous study showed that rapid adhering cells to collagen IV had the characteristics of putative progenitor/KSCs.
OBJECTIVE
The purpose of this study was to investigate the genetic expression of rapid adhering cells compared to non adhering cells to determine the characteristic of KSCs.
METHODS
We isolated rapid adhering cells representative of KSCs from non adhering cells representative of transient amplifying cells. In addition, we differentiated cells from human tonsilar keratinocytes utilizing the adhering capability of the KSCs to collagen IV. Annealing control primer based differentially displayed polymerase chain reaction (PCR) was performed as well as Western blot analysis.
RESULTS
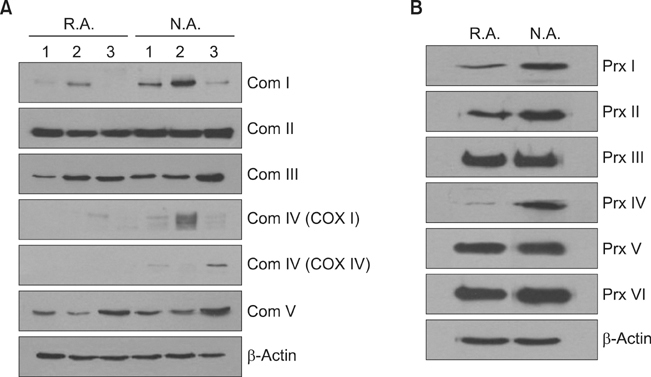
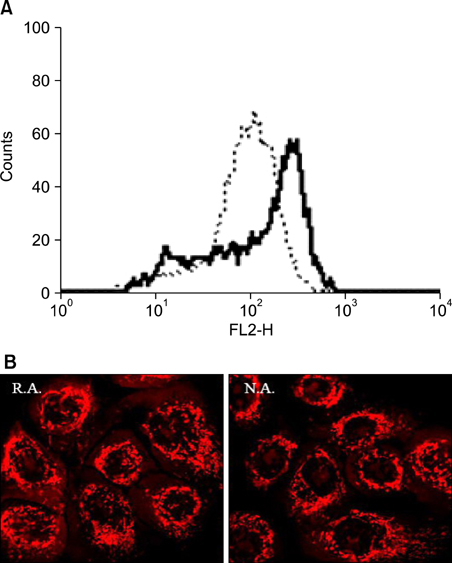
The levels of mitochondria-related gene expression were low in the rapid adhering cells compared to the non adhering cells. Mitochondrial complex I, COX IV, peroxiredoxins (I, II and IV) and mitochondrial membrane potential were all low in the rapid adhering cells compared to the non adhering cells.
CONCLUSION
Using an adhesion method on human collagen IV-coated plates, our results suggest that reduced mitochondrial function may be an important characteristic of KSCs.
MeSH Terms
Figure
Reference
-
1. Potten CS. Keratinocyte stem cells, label-retaining cells and possible genome protection mechanisms. J Investig Dermatol Symp Proc. 2004. 9:183–195.
Article2. Pellegrini G, Bondanza S, Guerra L, De Luca M. Cultivation of human keratinocyte stem cells: current and future clinical applications. Med Biol Eng Comput. 1998. 36:778–790.
Article3. Kaur P. Interfollicular epidermal stem cells: identification, challenges, potential. J Invest Dermatol. 2006. 126:1450–1458.
Article4. Kaur P, Li A. Adhesive properties of human basal epidermal cells: an analysis of keratinocyte stem cells, transit amplifying cells, and postmitotic differentiating cells. J Invest Dermatol. 2000. 114:413–420.
Article5. Potten CS, Booth C. Keratinocyte stem cells: a commentary. J Invest Dermatol. 2002. 119:888–899.6. Jones PH, Watt FM. Separation of human epidermal stem cells from transit amplifying cells on the basis of differences in integrin function and expression. Cell. 1993. 73:713–724.
Article7. Tani H, Morris RJ, Kaur P. Enrichment for murine keratinocyte stem cells based on cell surface phenotype. Proc Natl Acad Sci U S A. 2000. 97:10960–10965.
Article8. Jones PH, Harper S, Watt FM. Stem cell patterning and fate in human epidermis. Cell. 1995. 80:83–93.
Article9. Kim DS, Cho HJ, Choi HR, Kwon SB, Park KC. Isolation of human epidermal stem cells by adherence and the reconstruction of skin equivalents. Cell Mol Life Sci. 2004. 61:2774–2781.
Article10. Jeong EJ, Choi SH, Chang SE, Chang HW, Roh JL, Lee SW, et al. Putative progenitor/stem cells isolated from human oral mucosa are resistant to ionizing radiation. J Dermatol Sci. 2008. 50:65–68.
Article11. Radu E, Simionescu O, Regalia T, Dumitrescu D, Popescu LM. Stem cells (p63(+)) in keratinocyte cultures from human adult skin. J Cell Mol Med. 2002. 6:593–598.
Article12. Li S, Ross DT, Kadin ME, Brown PO, Wasik MA. Comparative genome-scale analysis of gene expression profiles in T cell lymphoma cells during malignant progression using a complementary DNA microarray. Am J Pathol. 2001. 158:1231–1237.
Article13. Hwang KC, Cui XS, Park SP, Shin MR, Park SY, Kim EY, et al. Identification of differentially regulated genes in bovine blastocysts using an annealing control primer system. Mol Reprod Dev. 2004. 69:43–51.
Article14. Hong KK, Lew BL, Kim YI, Lee JW, Kim NI. The effect of TNF-alpha and IFN-gamma on the telomerase activity of cultured human keratinocytes. Ann Dermatol. 2007. 19:147–152.
Article15. Dong R, Liu X, Liu Y, Deng Z, Nie X, Wang X, et al. Enrichment of epidermal stem cells by rapid adherence and analysis of the reciprocal interaction of epidermal stem cells with neighboring cells using an organotypic system. Cell Biol Int. 2007. 31:733–740.
Article16. Piccoli C, Ria R, Scrima R, Cela O, D'Aprile A, Boffoli D, et al. Characterization of mitochondrial and extra-mitochondrial oxygen consuming reactions in human hematopoietic stem cells. Novel evidence of the occurrence of NAD(P)H oxidase activity. J Biol Chem. 2005. 280:26467–26476.
Article17. Kim JY, Kim YH, Chang I, Kim S, Pak YK, Oh BH, et al. Resistance of mitochondrial DNA-deficient cells to TRAIL: role of Bax in TRAIL-induced apoptosis. Oncogene. 2002. 21:3139–3148.
Article18. Lee JB, Yun SJ, Chae HZ, Won YH, Kim YP, Lee SC. Expression of peroxiredoxin and thioredoxin in dermatological disorders. Br J Dermatol. 2002. 146:710–712.
Article19. Greaves LC, Preston SL, Tadrous PJ, Taylor RW, Barron MJ, Oukrif D, et al. Mitochondrial DNA mutations are established in human colonic stem cells, and mutated clones expand by crypt fission. Proc Natl Acad Sci U S A. 2006. 103:714–719.
Article20. Shidara Y, Yamagata K, Kanamori T, Nakano K, Kwong JQ, Manfredi G, et al. Positive contribution of pathogenic mutations in the mitochondrial genome to the promotion of cancer by prevention from apoptosis. Cancer Res. 2005. 65:1655–1663.
Article21. Finlan LE, Hupp TR. Epidermal stem cells and cancer stem cells: insights into cancer and potential therapeutic strategies. Eur J Cancer. 2006. 42:1283–1292.
Article22. Lee SC, Chae HZ, Lee JE, Kwon BD, Lee JB, Won YH, et al. Peroxiredoxin is ubiquitously expressed in rat skin: isotype-specific expression in the epidermis and hair follicle. J Invest Dermatol. 2000. 115:1108–1114.
Article23. Cho YM, Kwon S, Pak YK, Seol HW, Choi YM, Park DJ, et al. Dynamic changes in mitochondrial biogenesis and antioxidant enzymes during the spontaneous differentiation of human embryonic stem cells. Biochem Biophys Res Commun. 2006. 348:1472–1478.
Article24. Lonergan T, Brenner C, Bavister B. Differentiation-related changes in mitochondrial properties as indicators of stem cell competence. J Cell Physiol. 2006. 208:149–153.
Article25. St John JC, Ramalho-Santos J, Gray HL, Petrosko P, Rawe VY, Navara CS, et al. The expression of mitochondrial DNA transcription factors during early cardiomyocyte in vitro differentiation from human embryonic stem cells. Cloning Stem Cells. 2005. 7:141–153.26. St John JC, Amaral A, Bowles E, Oliveira JF, Lloyd R, Freitas M, et al. The analysis of mitochondria and mitochondrial DNA in human embryonic stem cells. Methods Mol Biol. 2006. 331:347–374.27. Koopman WJ, Visch HJ, Verkaart S, van den Heuvel LW, Smeitink JA, Willems PH. Mitochondrial network complexity and pathological decrease in complex I activity are tightly correlated in isolated human complex I deficiency. Am J Physiol Cell Physiol. 2005. 289:C881–C890.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- High Glucose Causes Human Cardiac Progenitor Cell Dysfunction by Promoting Mitochondrial Fission: Role of a GLUT1 Blocker
- Stem Cells for the Regeneration of Tendon and Ligament: A Perspective
- Adult Stem Cells: Beyond Regenerative Tool, More as a Bio-Marker in Obesity and Diabetes
- Endothelial progenitor cells and mesenchymal stem cells from human cord blood
- The Calcineurin-Drp1-Mediated Mitochondrial Fragmentation is Aligned with the Differentiation of c-Kit Cardiac Progenitor Cells