Aprotinin Inhibits Vascular Smooth Muscle Cell Inflammation and Proliferation via Induction of HO-1
- Affiliations
-
- 1Department of Cardiac Surgery, College of Medicine, Yeungnam University, Daegu 705-717, Korea.
- 2Department of Pharmacology and Aging-associated Vascular Disease Research Center, College of Medicine, Yeungnam University, Daegu 705-717, Korea. yjkang@med.yu.ac.kr
- KMID: 2071661
- DOI: http://doi.org/10.4196/kjpp.2009.13.2.123
Abstract
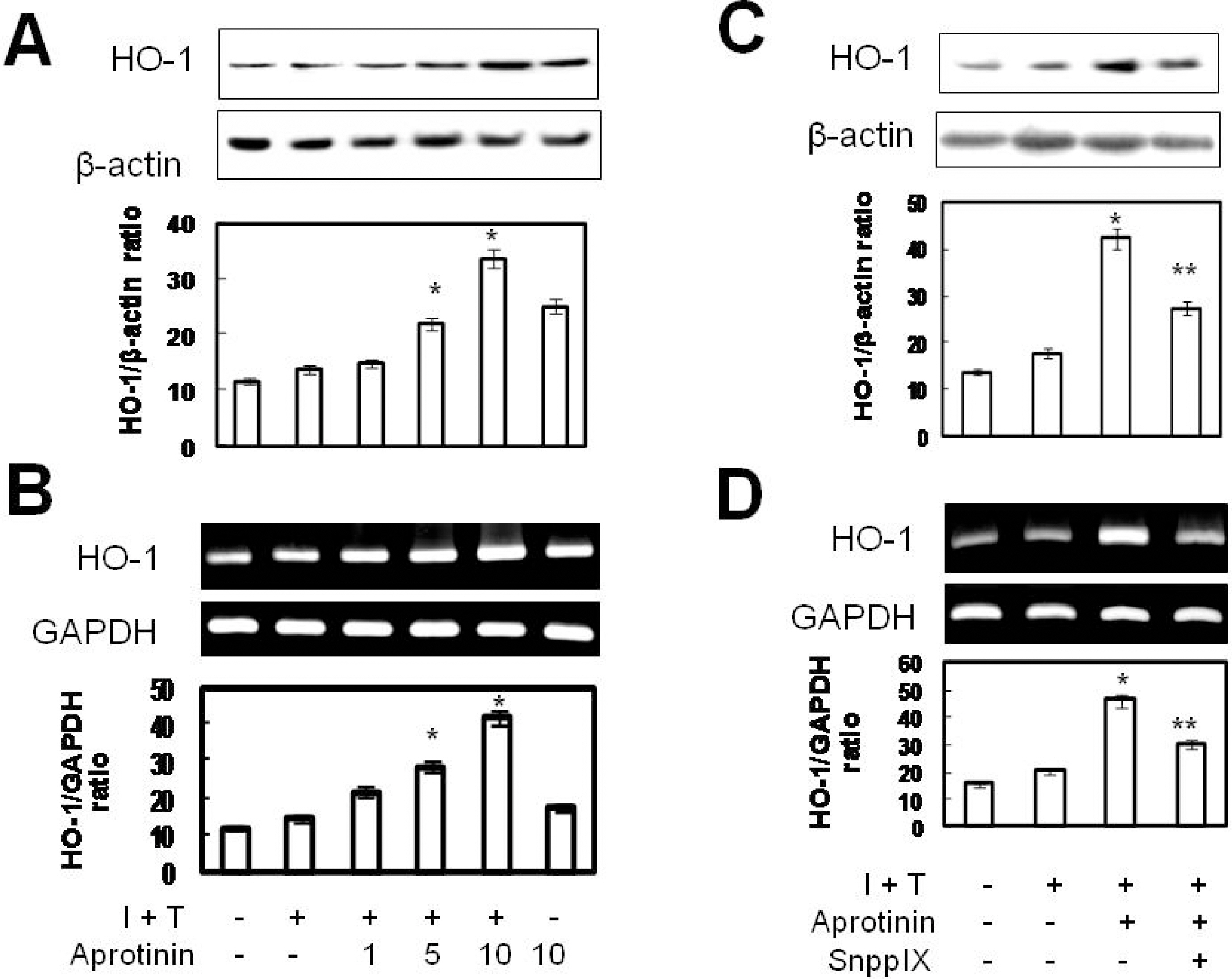
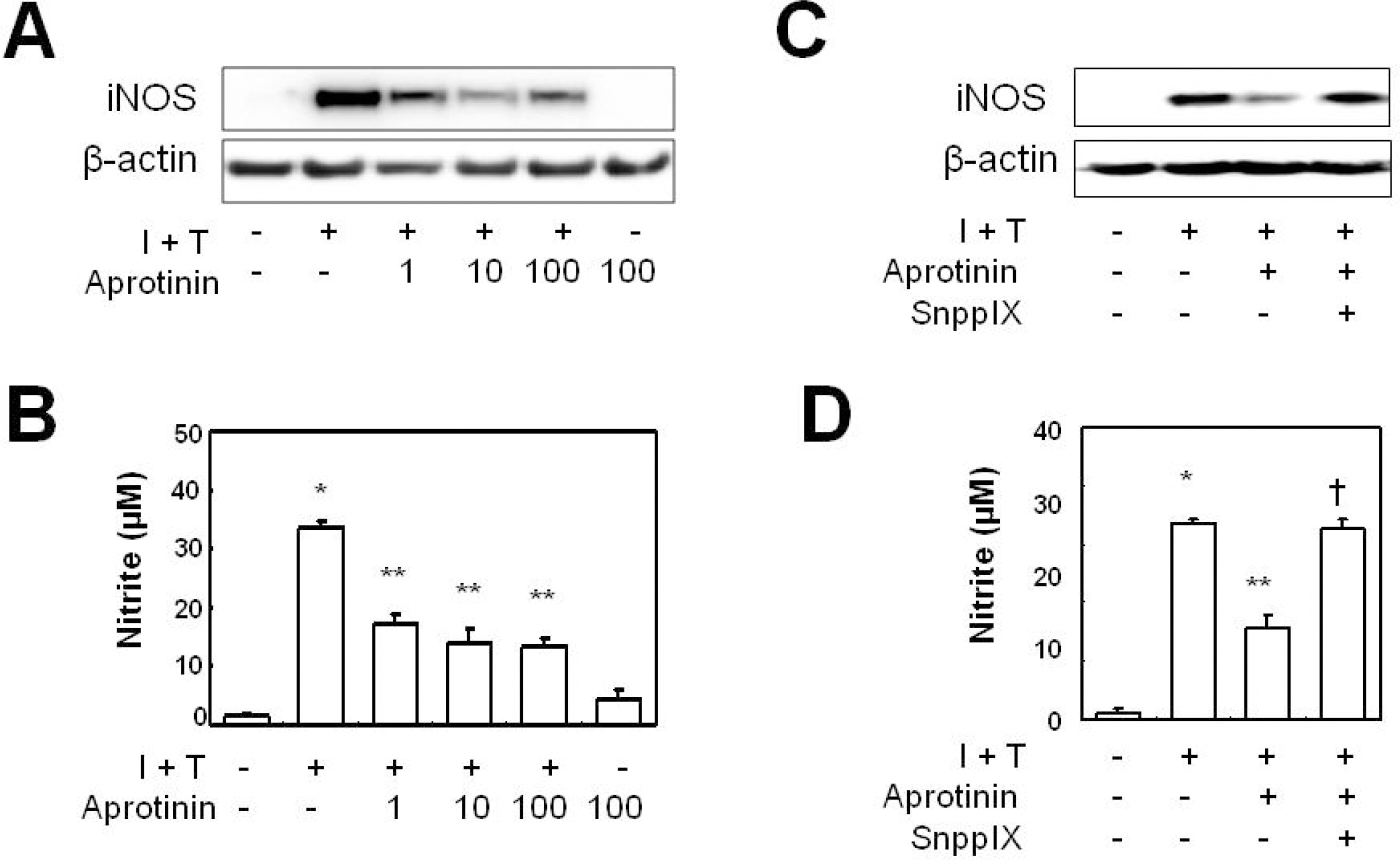
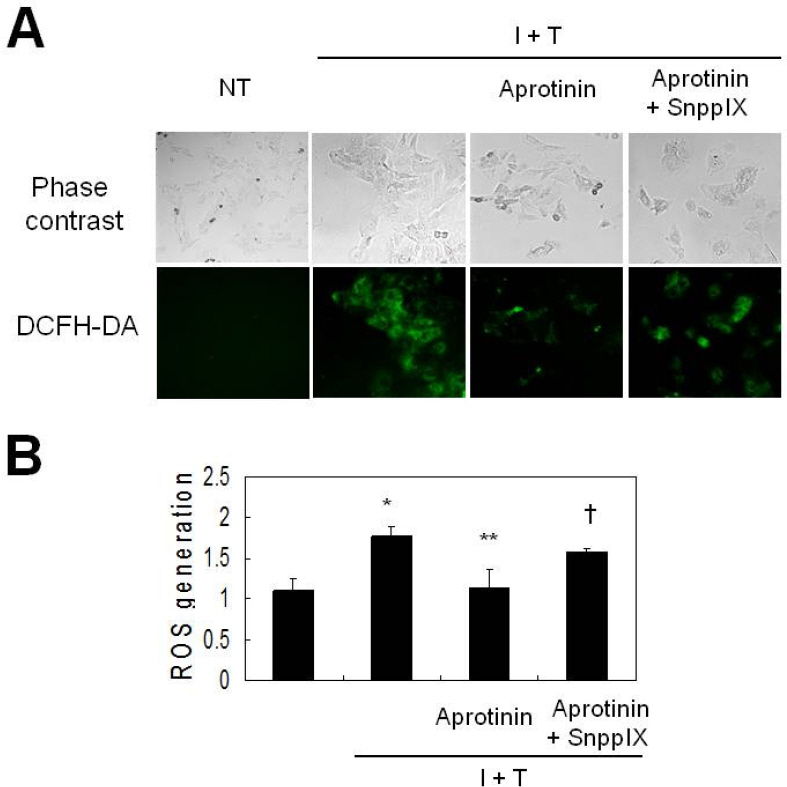
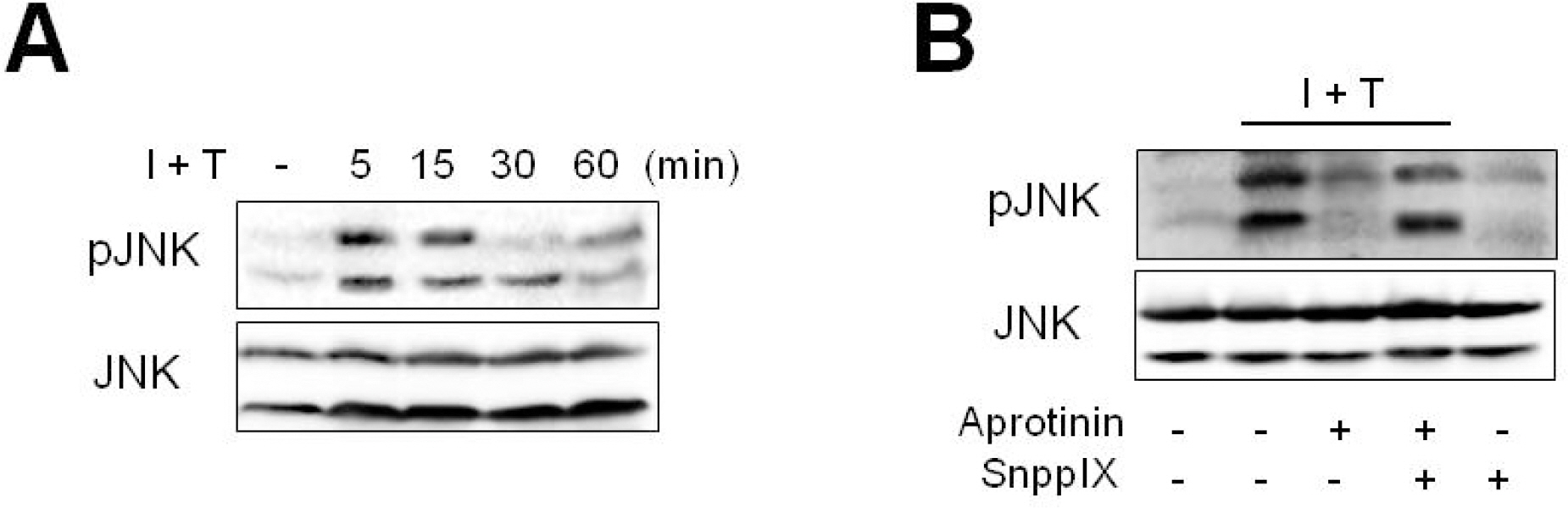
- Aprotinin is used clinically in cardiopulmonary bypass surgery to reduce transfusion requirements and the inflammatory response. The mechanism of action for the anti-inflammatory effects of aprotinin is still unclear. We examined our hypothesis whether inhibitory effects of aprotinin on cytokine-induced inducible nitric oxide synthase (iNOS) expression (IL-1beta plus TNF-alpha), reactive oxygen species (ROS) generation, and vascular smooth muscle cell (VSMC) proliferation were due to HO-1 induction in rat VSMCs. Aprotinin induced HO-1 protein expression in a dose-dependent manner, which was potentiated during inflammatory condition. Aprotinin reduced cytokine mixture (CM)-induced iNOS expression in a dose dependent manner. Furthermore, aprotinin reduced CM-induced ROS generation, cell proliferation, and phosphorylation of JNK but not of P38 and ERK1/2 kinases. Aprotinin effects were reversed by pre-treatment with the HO-1 inhibitor, tin protoporphyrin IX (SnPPIX). HO-1 is therefore closely involved in inflammatory-stimulated VSMC proliferation through the regulation of ROS generation and JNK phosphorylation. Our results suggest a new molecular basis for aprotinin anti-inflammatory properties.
Keyword
MeSH Terms
-
Animals
Aprotinin
Cardiopulmonary Bypass
Cell Proliferation
Inflammation
Metalloporphyrins
Muscle, Smooth, Vascular
Nitric Oxide Synthase Type II
Phosphorylation
Phosphotransferases
Protoporphyrins
Rats
Reactive Oxygen Species
Tin
Aprotinin
Metalloporphyrins
Nitric Oxide Synthase Type II
Phosphotransferases
Protoporphyrins
Reactive Oxygen Species
Tin
Figure
Cited by 3 articles
-
Cigarette Smoke Extract-induced Reduction in Migration and Contraction in Normal Human Bronchial Smooth Muscle Cells
Chul Ho Yoon, Hye-Jin Park, Young-Woo Cho, Eun-Jin Kim, Jong Deog Lee, Kee Ryeon Kang, Jaehee Han, Dawon Kang
Korean J Physiol Pharmacol. 2011;15(6):397-403. doi: 10.4196/kjpp.2011.15.6.397.Heme Oxygenase-1 Induced by Aprotinin Inhibits Vascular Smooth Muscle Cell Proliferation Through Cell Cycle Arrest in Hypertensive Rats
Hyoung Chul Choi, Kwang Youn Lee, Dong Hyup Lee, Young Jin Kang
Korean J Physiol Pharmacol. 2009;13(4):309-313. doi: 10.4196/kjpp.2009.13.4.309.Curcumin Induces Downregulation of E2F4 Expression and Apoptotic Cell Death in HCT116 Human Colon Cancer Cells; Involvement of Reactive Oxygen Species
Kyung-Chan Kim, ChuHee Lee
Korean J Physiol Pharmacol. 2010;14(6):391-397. doi: 10.4196/kjpp.2010.14.6.391.
Reference
-
Asimakopoulos G., Thompson R., Nourshargh S., Lidington EA., Mason JC., Ratnatunga CP., Haskard DO., Taylor KM., Landis RC. An anti-inflammatory property of aprotinin detected at the level of leukocyte extravasation. J Thorac Cardiovasc Surg. 120:361–369. 2000.
ArticleBrister SJ., Ofosu FA., Buchanan MR. Thrombin generation during cardiac surgery: is heparin the ideal anticoagulant? Thromb Haemost. 70:259–262. 1993.
ArticleClark JE., Foresti R., Green CJ., Motterlini R. Dynamics of haem oxygenase-1 expression and bilirubin production in cellular protection against oxidative stress. Biochem J. 348:615–619. 2000.
ArticleDay JR., Taylor KM., Lidington EA., Mason JC., Haskard DO., Randi AM., Landis RC. Aprotinin inhibits proinflammatory activation of endothelial cells by thrombin through the protease-activated receptor 1. J Thorac Cardiovasc Surg. 131:21–27. 2006.
ArticleDuckers HJ., Boehm M., True AL., Yet SF., San H., Park JL., Clinton Webb R., Lee ME., Nabel GJ., Nabel EG. Heme oxygenase-1 protects against vascular constriction and proliferation. Nat Med. 7:693–698. 2001.
ArticleDurante W., Kroll MH., Christodoulides N., Peyton KJ., Schafer AI. Nitric oxide induces heme oxygenase-1 gene expression and carbon monoxide production in vascular smooth muscle cells. Circ Res. 80:557–64. 1997.
ArticleHill GE., Springall DR., Robbins RA. Aprotinin is associated with a decrease in nitric oxide production during cardiopulmonary bypass. Surgery. 121:449–455. 1997.
ArticleGuikema BJ., Ginnan R., Singer HA., Jourd'heuil D. Catalase potentiates interleukin-1beta-induced expression of nitric oxide synthase in rat vascular smooth muscle cells. Free Radic Biol Med. 38:597–605. 2005.Johnson RA., Lavesa M., Askari B., Abraham NG., Nasjletti A. A heme oxygenase product, presumably carbon monoxide, mediates a vasodepressor function in rats. Hypertension. 25:166–169. 1995.
ArticleJuan SH., Cheng TH., Lin HC., Chu YL., Lee WS. Mechanism of concentration-dependent induction of heme oxygenase-1 by resveratrol in human aortic smooth muscle cells. Biochem Pharmacol. 69:41–48. 2005.
ArticleKapturczak MH., Wasserfall C., Brusko T., Campbell-Thompson M., Ellis TM., Atkinson MA., Agarwal A. Heme oxygenase-1 modulates early inflammatory responses: evidence from the heme oxygenase-1-deficient mouse. Am J Pathol. 165:1045–1053. 2004.Keyse SM., Tyrrell RM. Heme oxygenase is the major 32-kDa stress protein induced in human skin fibroblasts by UVA radiation, hydrogen peroxide, and sodium arsenite. Proc Natl Acad Sci USA. 86:99–103. 1989.
ArticleLandis RC., Asimakopoulos G., Poullis M., Haskard DO., Taylor KM. The antithrombotic and antiinflammatory mechanisms of action of aprotinin. Ann Thorac Surg. 72:2169–2175. 2001.
ArticleLandis RC., Haskard DO., Taylor KM. New antiinflammatory and platelet-preserving effects of aprotinin. Ann Thorac Surg. 72:S1808–1813. 2001.
ArticleLee TS., Chang CC., Zhu Y., Shyy JY. Simvastatin induces heme oxygenase-1: a novel mechanism of vessel protection. Circulation. 110:1296–1302. 2004.Lee TS., Chau LY. Heme oxygenase-1 mediates the anti-inflammatory effect of interleukin-10 in mice. Nat Med. 8:240–246. 2002.
ArticleLee TS., Tsai HL., Chau LY. Induction of heme oxygenase-1 expression in murine macrophages is essential for the anti-inflammatory effect of low dose 15-deoxy-Δ12,14-prostaglandin J2. J Biol Chem. 278:19325–19330. 2003.Maines MD. Heme oxygenase: function, multiplicity, regulatory mechanisms, and clinical applications. FASEB J. 2:2557–2568. 1988.
ArticleMcCoubrey WK Jr., Huang TJ., Maines MD. Isolation and characterization of a cDNA from the rat brain that encodes hemoprotein heme oxygenase-3. Eur J Bioche. 247:725–732. 1997.
ArticleMinamino T., Christou H., Hsieh CM., Liu Y., Dhawan V., Abraham NG., Perrella MA., Mitsialis SA., Kourembanas S. Targeted expression of heme oxygenase-1 prevents the pulmonary inflammatory and vascular responses to hypoxia. Proc Natl Acad Sci USA. 98:8798–8803. 2001.
ArticleMotterlini R., Foresti R., Intaglietta M., Winslow RM. NO-mediated activation of heme oxygenase: endogenous cytoprotection against oxidative stress to endothelium. Am J Physio. 270:H107–H114. 1996.
ArticleOtterbein LE., Zuckerbraun BS., Haga M., Liu F., Song R., Usheva A., Stachulak C., Bodyak N., Smith RN., Csizmadia E., Tyagi S., Akamatsu Y., Flavell RJ., Billiar TR., Tzeng E., Bach FH., Choi AM., Soares MP. Carbon monoxide suppresses arteriosclerotic lesions associated with chronic graft rejection and with balloon injury. Nat Med. 9:183–190. 2003.
ArticlePoss KD., Tonegawa S. Reduced stress defense in heme oxygenase deficient cells. Proc Natl Acad Sci USA. 94:10925–10930. 1997.Poullis M., Manning R., Laffan M., Haskard DO., Taylor KM., Landis RC. The antithrombotic effect of aprotinin: actions mediated via the protease activated receptor 1. J Thorac Cardiovasc Surg. 120:370–378. 2000.Tenhunen R., MArver HS., Schmid R. The enzymatic conversion of heme to bilirubin by microsomal heme owygenase. Proc Natl Acad Sci USA. 61:748–755. 1968.Wachtfogel YT., Kucich U., Hack CE., Gluszko P., Niewiarowski S., Colman RW., Edmunds LH Jr. Aprotinin inhibits the contact, neutrophil, and platelet activation systems during simulated extracorporeal perfusion. J Thorac Cardiovasc Surg. 106:1–9. 1993.
ArticleWagner CT., Durante W., Christodoulides N., Hellums JD., Schafer AI. Hemodynamic forces induce the expression of heme oxygenase in cultured vascular smooth muscle cells. J Clin Invest. 100:589–596. 1997.
ArticleWestaby S. Aprotinin in perspective. Ann Thorac Surg. 55:1033–1041. 1993.
ArticleYachie A., Niida Y., Wada T., Igarashi N., Kaneda H. Oxidative stress causes enhanced endothelial cell injury in human heme oxygenase-1 deficiency. J Clin Invest. 103:129–135. 1999.
ArticleYoshinaga T., Sassa S., Kappas A. Purification and properties of bovine spleen heme oxygenase: amino acid composition and sites of action of inhibitors of heme oxygenase. J Biol Chem. 257:7778–7785. 1982.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Heme Oxygenase-1 Induced by Aprotinin Inhibits Vascular Smooth Muscle Cell Proliferation Through Cell Cycle Arrest in Hypertensive Rats
- Intimal Hyperplasia
- Insulin Enhances Suppressive Effect of Lipopolysaccharide on Glucose-induced Proliferation of Vascular Smooth Muscle Cells
- Losartan Inhibits Vascular Smooth Muscle Cell Proliferation through Activation of AMP-Activated Protein Kinase
- The effect of advanced glycation and products on the proliferation of vascular smooth muscle cell