Korean J Physiol Pharmacol.
2013 Apr;17(2):139-147. 10.4196/kjpp.2013.17.2.139.
Signaling Pathway of Lysophosphatidic Acid-Induced Contraction in Feline Esophageal Smooth Muscle Cells
- Affiliations
-
- 1Department of Pharmacology, College of Pharmacy, Chung-Ang University, Seoul 156-756, Korea. udsohn@cau.ac.kr
- KMID: 1429385
- DOI: http://doi.org/10.4196/kjpp.2013.17.2.139
Abstract
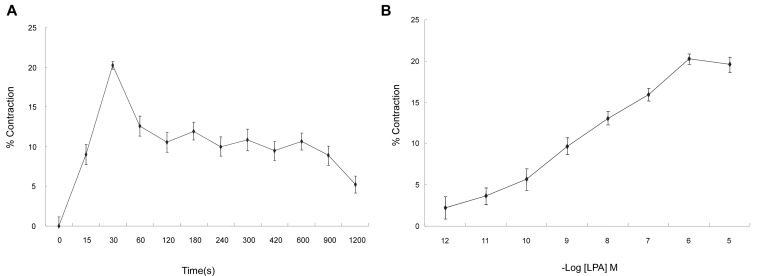
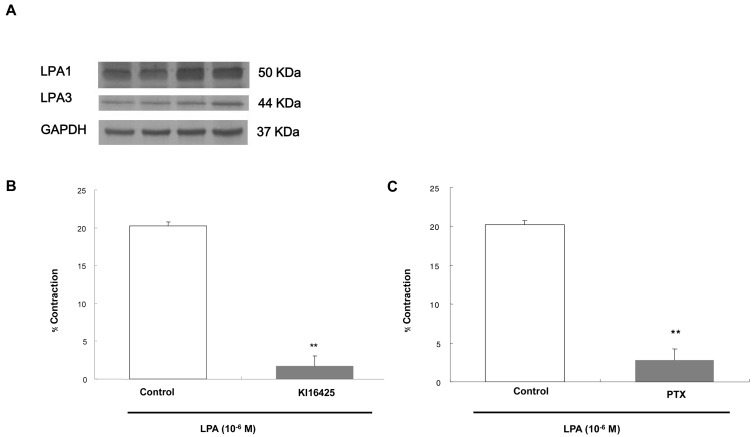
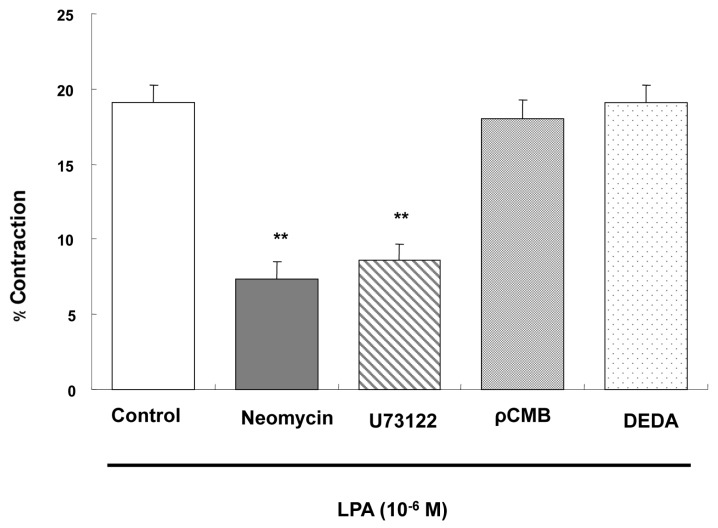
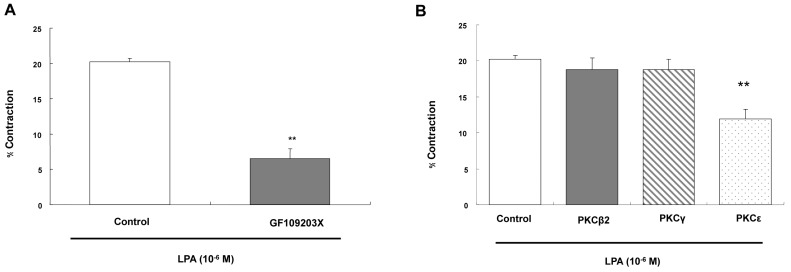
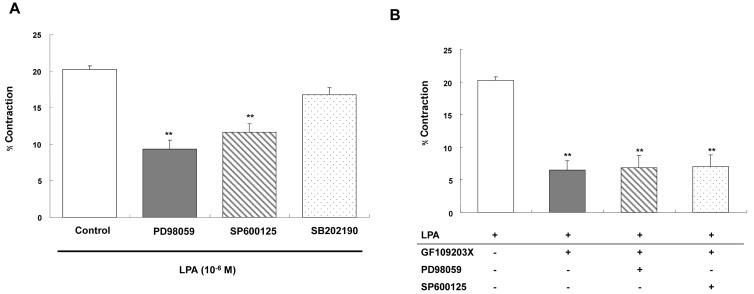
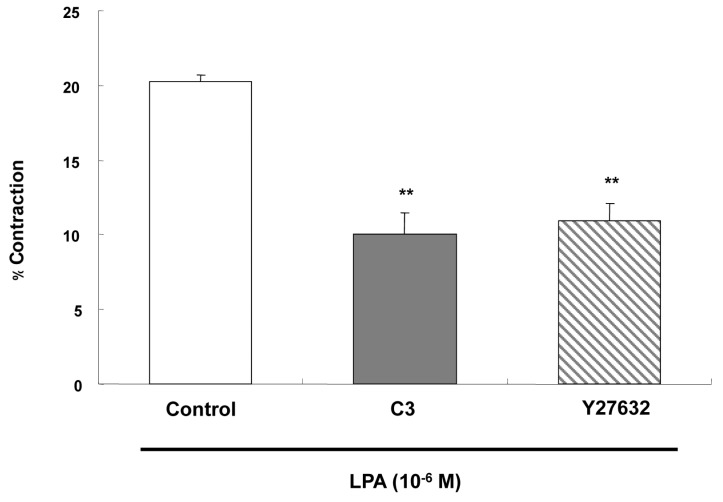
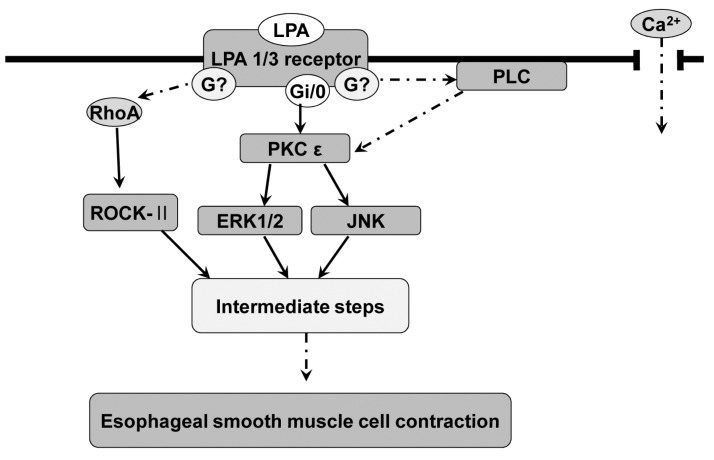
- Lysolipids such as LPA, S1P and SPC have diverse biological activities including cell proliferation, differentiation, and migration. We investigated signaling pathways of LPA-induced contraction in feline esophageal smooth muscle cells. We used freshly isolated smooth muscle cells and permeabilized cells from cat esophagus to measure the length of cells. Maximal contraction occurred at 10(-6) M and the response peaked at 30s. To identify LPA receptor subtypes in cells, western blot analysis was performed with antibodies to LPA receptor subtypes. LPA1 and LPA3 receptor were detected at 50 kDa and 44 kDa. LPA-induced contraction was almost completely blocked by LPA receptor (1/3) antagonist KI16425. Pertussis toxin (PTX) inhibited the contraction induced by LPA, suggesting that the contraction is mediated by a PTX-sensitive G protein. Phospholipase C (PLC) inhibitors U73122 and neomycin, and protein kinase C (PKC) inhibitor GF109203X also reduced the contraction. The PKC-mediated contraction may be isozyme-specific since only PKCepsilon antibody inhibited the contraction. MEK inhibitor PD98059 and JNK inhibitor SP600125 blocked the contraction. However, there is no synergistic effect of PKC and MAPK on the LPA-induced contraction. In addition, RhoA inhibitor C3 exoenzyme and ROCK inhibitor Y27632 significantly, but not completely, reduced the contraction. The present study demonstrated that LPA-induced contraction seems to be mediated by LPA receptors (1/3), coupled to PTX-sensitive G protein, resulting in activation of PLC, PKC-epsilon pathway, which subsequently mediates activation of ERK and JNK. The data also suggest that RhoA/ROCK are involved in the LPA-induced contraction.
Keyword
MeSH Terms
-
Amides
Animals
Anthracenes
Antibodies
Blotting, Western
Cats
Cell Proliferation
Contracts
Esophagus
Estrenes
Flavonoids
GTP-Binding Proteins
Indoles
Isoxazoles
Maleimides
Muscle, Smooth
Myocytes, Smooth Muscle
Neomycin
Pertussis Toxin
Propionates
Protein Kinase C
Pyridines
Pyrrolidinones
Receptors, Lysophosphatidic Acid
Type C Phospholipases
Amides
Anthracenes
Antibodies
Estrenes
Flavonoids
GTP-Binding Proteins
Indoles
Isoxazoles
Maleimides
Neomycin
Pertussis Toxin
Propionates
Protein Kinase C
Pyridines
Pyrrolidinones
Receptors, Lysophosphatidic Acid
Type C Phospholipases
Figure
Reference
-
1. Chun J, Goetzl EJ, Hla T, Igarashi Y, Lynch KR, Moolenaar W, Pyne S, Tigyi G. International Union of Pharmacology. XXXIV. Lysophospholipid receptor nomenclature. Pharmacol Rev. 2002; 54:265–269. PMID: 12037142.
Article2. Moolenaar WH, Kranenburg O, Postma FR, Zondag GC. Lysophosphatidic acid: G-protein signalling and cellular responses. Curr Opin Cell Biol. 1997; 9:168–173. PMID: 9069262.
Article3. Goetzl EJ, An S. Diversity of cellular receptors and functions for the lysophospholipid growth factors lysophosphatidic acid and sphingosine 1-phosphate. FASEB J. 1998; 12:1589–1598. PMID: 9837849.
Article4. Siess W. Athero- and thrombogenic actions of lysophosphatidic acid and sphingosine-1-phosphate. Biochim Biophys Acta. 2002; 1582:204–215. PMID: 12069830.
Article5. Ando T, Ishiguro H, Kuwabara Y, Kimura M, Mitsui A, Kurehara H, Sugito N, Tomoda K, Mori R, Takashima N, Ogawa R, Fujii Y. Expression of ACP6 is an independent prognostic factor for poor survival in patients with esophageal squamous cell carcinoma. Oncol Rep. 2006; 15:1551–1555. PMID: 16685394.
Article6. Toews ML, Ustinova EE, Schultz HD. Lysophosphatidic acid enhances contractility of isolated airway smooth muscle. J Appl Physiol. 1997; 83:1216–1222. PMID: 9338431.7. Bischoff A, Czyborra P, Fetscher C, Meyer Zu Heringdorf D, Jakobs KH, Michel MC. Sphingosine-1-phosphate and sphingosylphosphorylcholine constrict renal and mesenteric microvessels in vitro. Br J Pharmacol. 2000; 130:1871–1877. PMID: 10952677.8. Ohmori T, Yatomi Y, Osada M, Kazama F, Takafuta T, Ikeda H, Ozaki Y. Sphingosine 1-phosphate induces contraction of coronary artery smooth muscle cells via S1P2. Cardiovasc Res. 2003; 58:170–177. PMID: 12667959.9. Tokumura A, Fukuzawa K, Yamada S, Tsukatani H. Stimulatory effect of lysophosphatidic acids on uterine smooth muscles of non-pregant rats. Arch Int Pharmacodyn Ther. 1980; 245:74–83. PMID: 6902643.10. Meyer zu Heringdorf D, van Koppen CJ, Jakobs KH. Molecular diversity of sphingolipid signalling. FEBS Lett. 1997; 410:34–38. PMID: 9247118.
Article11. Van Brocklyn JR, Lee MJ, Menzeleev R, Olivera A, Edsall L, Cuvillier O, Thomas DM, Coopman PJ, Thangada S, Liu CH, Hla T, Spiegel S. Dual actions of sphingosine-1-phosphate: extracellular through the Gi-coupled receptor Edg-1 and intracellular to regulate proliferation and survival. J Cell Biol. 1998; 142:229–240. PMID: 9660876.
Article12. van Koppen C, Meyer zu Heringdorf M, Laser KT, Zhang C, Jakobs KH, Bünemann M, Pott L. Activation of a high affinity Gi protein-coupled plasma membrane receptor by sphingosine-1-phosphate. J Biol Chem. 1996; 271:2082–2087. PMID: 8567663.
Article13. Contos JJ, Ishii I, Chun J. Lysophosphatidic acid receptors. Mol Pharmacol. 2000; 58:1188–1196. PMID: 11093753.
Article14. Ishii I, Fukushima N, Ye X, Chun J. Lysophospholipid receptors: signaling and biology. Annu Rev Biochem. 2004; 73:321–354. PMID: 15189145.
Article15. Kostenis E. Novel clusters of receptors for sphingosine-1-phosphate, sphingosylphosphorylcholine, and (lyso)-phosphatidic acid: new receptors for “old” ligands. J Cell Biochem. 2004; 92:923–936. PMID: 15258916.
Article16. Parrill AL, Sardar VM, Yuan H. Sphingosine 1-phosphate and lysophosphatidic acid receptors: agonist and antagonist binding and progress toward development of receptor-specific ligands. Semin Cell Dev Biol. 2004; 15:467–476. PMID: 15271292.
Article17. Radeff-Huang J, Seasholtz TM, Matteo RG, Brown JH. G protein mediated signaling pathways in lysophospholipid induced cell proliferation and survival. J Cell Biochem. 2004; 92:949–966. PMID: 15258918.
Article18. Renbäck K, Inoue M, Ueda H. Lysophosphatidic acid-induced, pertussis toxin-sensitive nociception through a substance P release from peripheral nerve endings in mice. Neurosci Lett. 1999; 270:59–61. PMID: 10454146.
Article19. Yang SJ, An JY, Shim JO, Park CH, Huh IH, Sohn UD. The mechanism of contraction by 2-chloroadenosine in cat detrusor muscle cells. J Urol. 2000; 163:652–658. PMID: 10647703.
Article20. Jung S, Lee Y, Han S, Kim Y, Nam T, Ahn D. Lysophosphatidylcholine increases ca current via activation of protein kinase C in rabbit portal vein smooth muscle cells. Korean J Physiol Pharmacol. 2008; 12:31–35. PMID: 20157391.21. Violin JD, Newton AC. Pathway illuminated: visualizing protein kinase C signaling. IUBMB Life. 2003; 55:653–660. PMID: 14769001.
Article22. Cao W, Sohn UD, Bitar KN, Behar J, Biancani P, Harnett KM. MAPK mediates PKC-dependent contraction of cat esophageal and lower esophageal sphincter circular smooth muscle. Am J Physiol Gastrointest Liver Physiol. 2003; 285:G86–G95. PMID: 12799309.23. Shin CY, Lee YP, Lee TS, Je HD, Kim DS, Sohn UD. The signal transduction of endothelin-1-induced circular smooth muscle cell contraction in cat esophagus. J Pharmacol Exp Ther. 2002; 302:924–934. PMID: 12183648.
Article24. Howe LR, Marshall CJ. Lysophosphatidic acid stimulates mitogen-activated protein kinase activation via a G-protein-coupled pathway requiring p21ras and p74raf-1. J Biol Chem. 1993; 268:20717–20720. PMID: 8407893.
Article25. Cobb MH, Goldsmith EJ. How MAP kinases are regulated. J Biol Chem. 1995; 270:14843–14846. PMID: 7797459.
Article26. Shirai H, Autieri M, Eguchi S. Small GTP-binding proteins and mitogen-activated protein kinases as promising therapeutic targets of vascular remodeling. Curr Opin Nephrol Hypertens. 2007; 16:111–115. PMID: 17293685.
Article27. Biancani P, Hillemeier C, Bitar KN, Makhlouf GM. Contraction mediated by Ca2+ influx in esophageal muscle and by Ca2+ release in the LES. Am J Physiol. 1987; 253:G760–G766. PMID: 3122586.28. Wang P, Bitar KN. Rho a regulates sustained smooth muscle contraction through cytoskeletal reorganization of HSP27. Am J Physiol. 1998; 275:G1454–G1462. PMID: 9843784.29. Fabiato A, Fabiato F. Calculator programs for computing the composition of the solutions containing multiple metals and ligands used for experiments in skinned muscle cells. J Physiol (Paris). 1979; 75:463–505. PMID: 533865.30. Patel CA, Rattan S. Cellular regulation of basal tone in internal anal sphincter smooth muscle by RhoA/ROCK. Am J Physiol Gastrointest Liver Physiol. 2007; 292:G1747–G1756. PMID: 17379756.
Article31. Sohn UD, Han B, Tashjian AH Jr, Behar J, Biancani P. Agonist-independent, muscle-type-specific signal transduction pathways in cat esophageal and lower esophageal sphincter circular smooth muscle. J Pharmacol Exp Ther. 1995; 273:482–491. PMID: 7536246.32. Song HJ, Choi TS, Chung FY, Park SY, Ryu JS, Woo JG, Min YS, Shin CY, Sohn UD. Sphingosine 1-phosphate-induced signal transduction in cat esophagus smooth muscle cells. Mol Cells. 2006; 21:42–51. PMID: 16511346.33. Sohn UD, Zoukhri D, Dartt D, Sergheraert C, Harnett KM, Behar J, Biancani P. Different protein kinase C isozymes mediate lower esophageal sphincter tone and phasic contraction of esophageal circular smooth muscle. Mol Pharmacol. 1997; 51:462–470. PMID: 9058601.34. Baek I, Jeon SB, Song MJ, Yang E, Sohn UD, Kim IK. Flavone attenuates vascular contractions by inhibiting RhoA/Rho kinase pathway. Korean J Physiol Pharmacol. 2009; 13:201–207. PMID: 19885038.
Article35. Kim YS, Song HJ, Park SY, Min YS, Im BO, Ko SK, Whang WK, Sohn UD. The signaling mechanism of the sphingosylphosphorylcholine-induced contraction in cat esophageal smooth muscle cells. Arch Pharm Res. 2007; 30:1608–1618. PMID: 18254249.
Article36. van der Bend RL, Brunner J, Jalink K, van Corven EJ, Moolenaar WH, van Blitterswijk WJ. Identification of a putative membrane receptor for the bioactive phospholipid, lysophosphatidic acid. EMBO J. 1992; 11:2495–2501. PMID: 1321033.
Article37. Fukushima N, Ishii I, Contos JJ, Weiner JA, Chun J. Lysophospholipid receptors. Annu Rev Pharmacol Toxicol. 2001; 41:507–534. PMID: 11264467.
Article38. Ohta H, Sato K, Murata N, Damirin A, Malchinkhuu E, Kon J, Kimura T, Tobo M, Yamazaki Y, Watanabe T, Yagi M, Sato M, Suzuki R, Murooka H, Sakai T, Nishitoba T, Im DS, Nochi H, Tamoto K, Tomura H, Okajima F. Ki16425, a subtype-selective antagonist for EDG-family lysophosphatidic acid receptors. Mol Pharmacol. 2003; 64:994–1005. PMID: 14500756.
Article39. Bitar KN, Hillemeier C, Biancani P, Balazovich KJ. Regulation of smooth muscle contraction in rabbit internal anal sphincter by protein kinase C and Ins(1,4,5)P3. Am J Physiol. 1991; 260:G537–G542. PMID: 2018129.
Article40. Ma T, Qi QH, Xu J, Dong ZL, Yang WX. Signal pathways involved in emodin-induced contraction of smooth muscle cells from rat colon. World J Gastroenterol. 2004; 10:1476–1479. PMID: 15133857.
Article41. Nishizuka Y. Protein kinase C and lipid signaling for sustained cellular responses. FASEB J. 1995; 9:484–496. PMID: 7737456.
Article42. Tangkijvanich P, Melton AC, Santiskulvong C, Yee HF Jr. Rho and p38 MAP kinase signaling pathways mediate LPA-stimulated hepatic myofibroblast migration. J Biomed Sci. 2003; 10:352–358. PMID: 12711863.
Article43. Payne DM, Rossomando AJ, Martino P, Erickson AK, Her JH, Shabanowitz J, Hunt DF, Weber MJ, Sturgill TW. Identification of the regulatory phosphorylation sites in pp42/mitogen-activated protein kinase (MAP kinase). EMBO J. 1991; 10:885–892. PMID: 1849075.
Article44. Bennett BL, Sasaki DT, Murray BW, O'Leary EC, Sakata ST, Xu W, Leisten JC, Motiwala A, Pierce S, Satoh Y, Bhagwat SS, Manning AM, Anderson DW. SP600125, an anthrapyrazolone inhibitor of Jun N-terminal kinase. Proc Natl Acad Sci U S A. 2001; 98:13681–13686. PMID: 11717429.
Article45. Fox T, Coll JT, Xie X, Ford PJ, Germann UA, Porter MD, Pazhanisamy S, Fleming MA, Galullo V, Su MS, Wilson KP. A single amino acid substitution makes ERK2 susceptible to pyridinyl imidazole inhibitors of p38 MAP kinase. Protein Sci. 1998; 7:2249–2255. PMID: 9827991.
Article46. Olofsson B. Rho guanine dissociation inhibitors: pivotal molecules in cellular signalling. Cell Signal. 1999; 11:545–554. PMID: 10433515.47. Chen XQ, Tan I, Ng CH, Hall C, Lim L, Leung T. Characterization of RhoA-binding kinase ROKalpha implication of the pleckstrin homology domain in ROKalpha function using region-specific antibodies. J Biol Chem. 2002; 277:12680–12688. PMID: 11815607.48. Gong MC, Fujihara H, Somlyo AV, Somlyo AP. Translocation of rhoA associated with Ca2+ sensitization of smooth muscle. J Biol Chem. 1997; 272:10704–10709. PMID: 9099720.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Relaxation Effect of Synthetic Ceramide Analogues in Cat Esophageal Smooth Muscle Cells
- The Effects of Heparin and Protamine on Contraction of Tracheal Smooth Muscle Induced by Carbachol in the Guinea Pig
- G protein-coupled receptor signaling in gastrointestinal smooth muscle
- Characteristics of 5-Hydroxytryptamine Receptors Involved in Contraction of Feline Ileal Longitudinal Smooth Muscle
- The Role of Lysophosphatidic Acid in Adult Stem Cells