J Breast Cancer.
2019 Sep;22(3):375-386. 10.4048/jbc.2019.22.e43.
In Vitro and In Vivo Study on the Effect of Lysosome-associated Protein Transmembrane 4 Beta on the Progression of Breast Cancer
- Affiliations
-
- 1Department of Oncological Surgery, Enze Hospital of Taizhou Enze Medical Group, Luqiao, Zhejiang, China.
- 2The Affiliated People's Hospital of Inner Mongolia Medical University, Hohhot, Inner Mongolia, China.
- 3Gynecology of Taizhou Enze Medical Center (Group) Enze Hospital, Taizhou, Zhejiang, China.
- 4Shanghai Key Laboratory of New Drug Design, School of Pharmacy, East China University of Science and Technology, Shanghai, China. wuxia_imu@163.com
- KMID: 2458859
- DOI: http://doi.org/10.4048/jbc.2019.22.e43
Abstract
- PURPOSE
Although the effect of lysosome-associated protein transmembrane 4 beta (LAPTM4B) on the proliferation, migration, and invasion of breast cancer (BC) cells has already been studied, its specific role in BC progression is still elusive. Here, we evaluated the effect of different levels of LAPTM4B expression on the proliferation, invasion, adhesion, and tumor formation abilities of BC cells in vitro, as well as on breast tumor progression in vivo.
METHODS
We investigated the influence of LAPTM4B expression on MCF-7 cell proliferation, invasion, adhesion, and tube formation abilities in vitro through its overexpression or knockdown and on breast tumor progression in vivo.
RESULTS
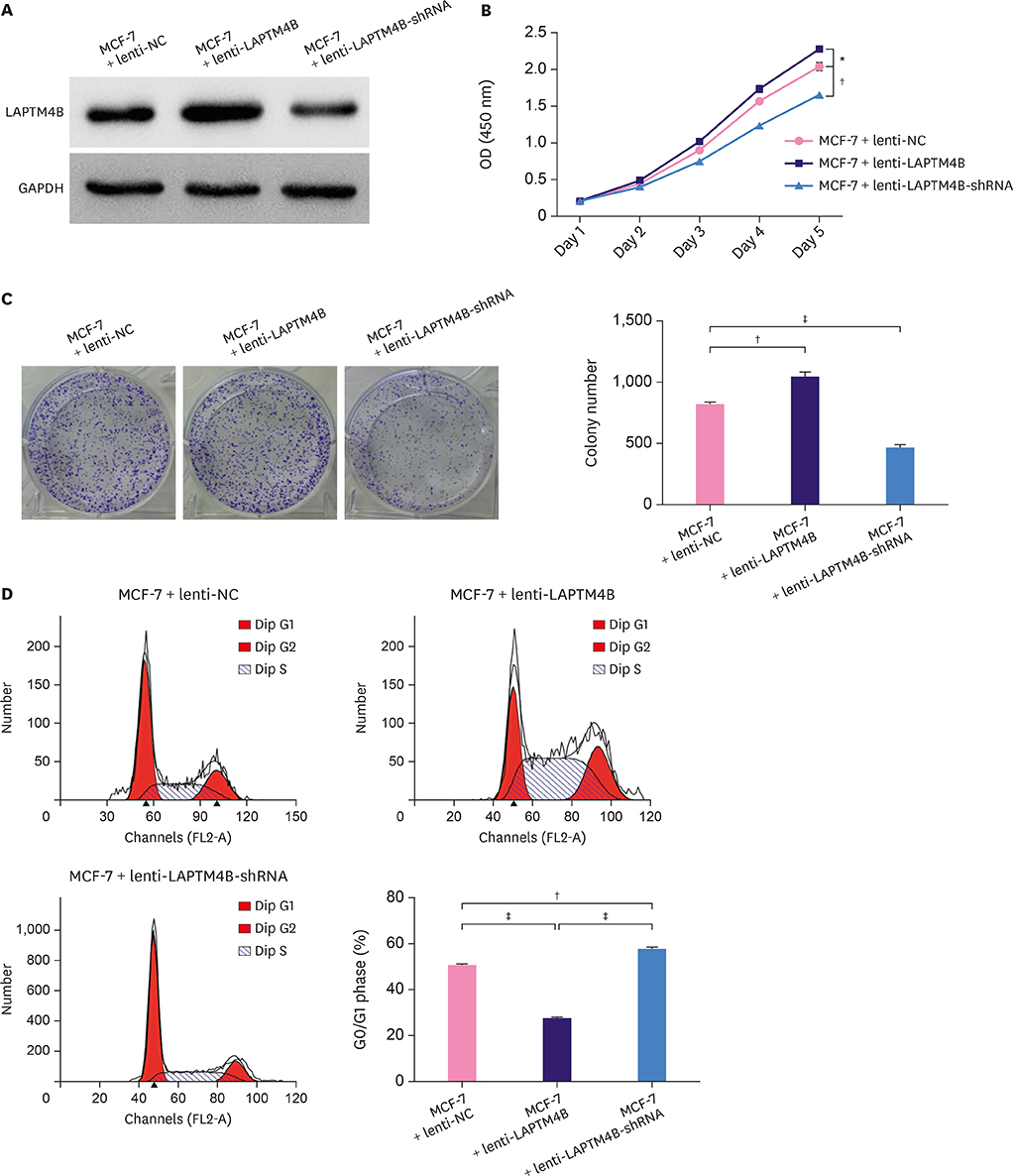
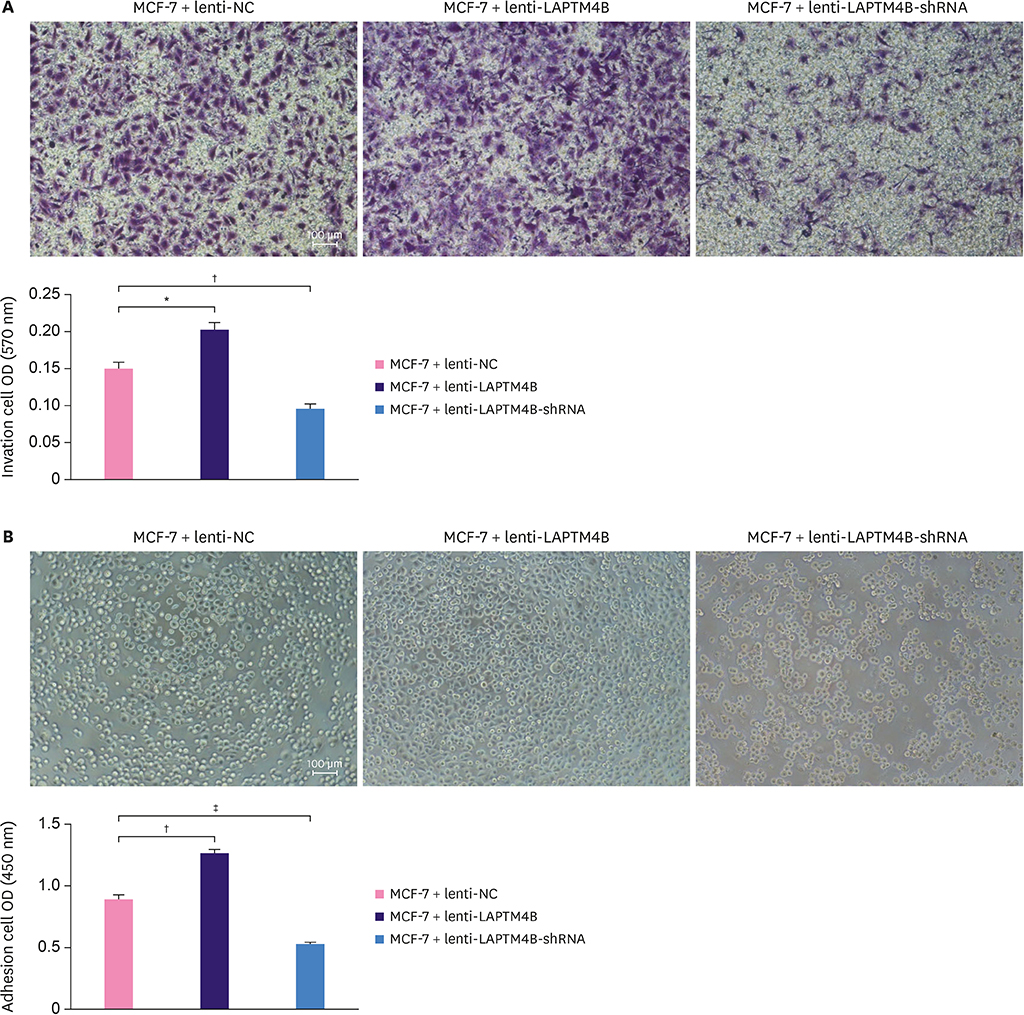
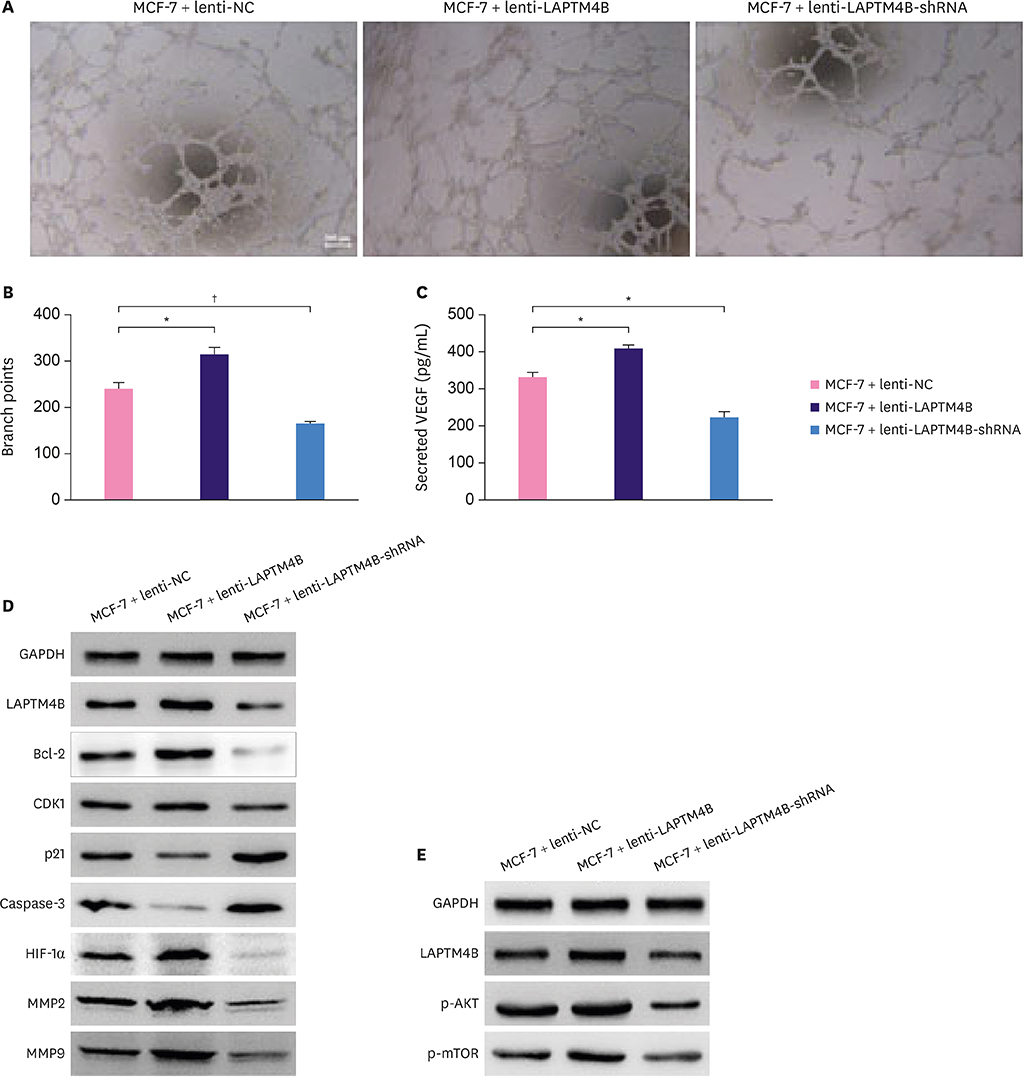
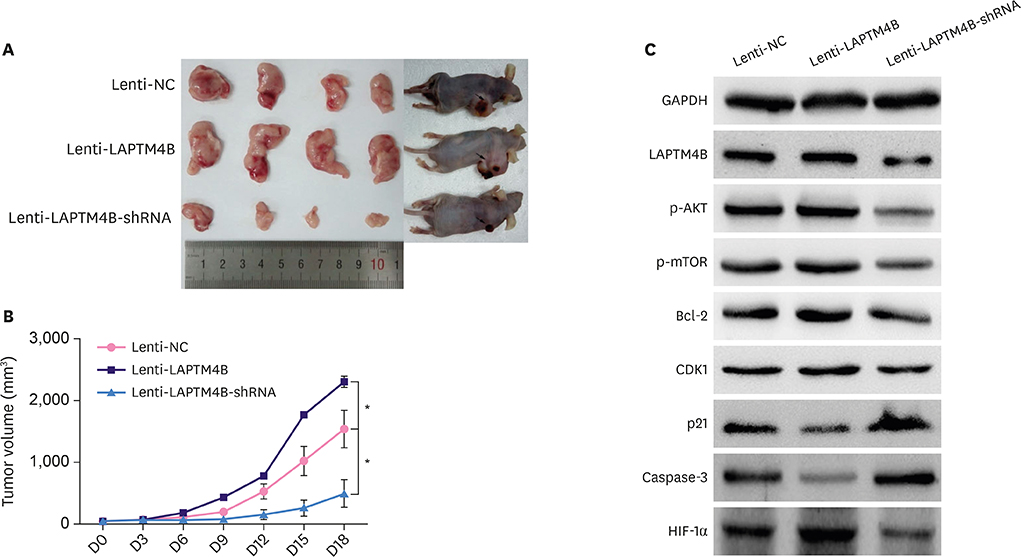
Cell growth curves and colony formation assays showed that LAPTM4B promoted the proliferation of breast tumor cells. Cell cycle analysis results revealed that LAPTM4B promoted the entry of cells from the G1 into the S phase. Transwell invasion and cell extracellular matrix adhesion assays showed that LAPTM4B overexpression increased the invasion and adhesion capabilities of MCF-7 cells. More branches were observed in MCF-7 cells overexpressing LAPTM4B under an electron microscope. In comparison with LAPTM4B overexpression, LAPTM4B knockdown decreased the expression of vascular endothelial growth factor-A and significantly inhibited the vasculogenic tube formation ability of tumors. These results were also verified with western blot analysis.
CONCLUSION
LAPTM4B promoted the proliferation of MCF-7 cells through the downregulation of p21 (WAF1/CIP1) and caspase-3, and induced cell invasion, adhesion, and angiogenesis through the upregulation of hypoxia-inducible factor 1 alpha, matrix metalloproteinase 2 (MMP2), and MMP9 expression. This specific role deems LAPTM4B as a potential therapeutic target for BC treatment.
MeSH Terms
-
Blotting, Western
Breast Neoplasms*
Breast*
Caspase 3
Cell Cycle
Disease Progression
Down-Regulation
Extracellular Matrix
Hypoxia-Inducible Factor 1
In Vitro Techniques*
Matrix Metalloproteinase 2
MCF-7 Cells
S Phase
Up-Regulation
Vascular Endothelial Growth Factor A
Caspase 3
Hypoxia-Inducible Factor 1
Matrix Metalloproteinase 2
Vascular Endothelial Growth Factor A
Figure
Reference
-
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011; 61:69–90.
Article2. Youlden DR, Cramb SM, Yip CH, Baade PD. Incidence and mortality of female breast cancer in the Asia-Pacific region. Cancer Biol Med. 2014; 11:101–115.3. Ginsburg O, Bray F, Coleman MP, Vanderpuye V, Eniu A, Kotha SR, et al. The global burden of women's cancers: a grand challenge in global health. Lancet. 2017; 389:847–860.
Article4. Bertos NR, Park M. Breast cancer - one term, many entities? J Clin Invest. 2011; 121:3789–3796.
Article5. Curigliano G, Burstein HJ, Winer EP, Gnant M, Dubsky P, Loibl S, et al. De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann Oncol. 2017; 28:1700–1712.
Article6. Tono Y, Ishihara M, Miyahara Y, Tamaru S, Oda H, Yamashita Y, et al. Pertuzumab, trastuzumab and eribulin mesylate therapy for previously treated advanced HER2-positive breast cancer: a feasibility study with analysis of biomarkers. Oncotarget. 2018; 9:14909–14921.
Article7. Li F, Dou J, Wei L, Li S, Liu J. The selective estrogen receptor modulators in breast cancer prevention. Cancer Chemother Pharmacol. 2016; 77:895–903.
Article8. Chen Y, Wang L, Zhu Y, Chen Z, Qi X, Jin L, et al. Breast cancer resistance protein (BCRP)-containing circulating microvesicles contribute to chemoresistance in breast cancer. Oncol Lett. 2015; 10:3742–3748.
Article9. Meng F, Song H, Luo C, Yin M, Xu Y, Liu H, et al. Correlation of LAPTM4B polymorphisms with cervical carcinoma. Cancer. 2011; 117:2652–2658.
Article10. Shao GZ, Zhou RL, Zhang QY, Zhang Y, Liu JJ, Rui JA, et al. Molecular cloning and characterization of LAPTM4B, a novel gene upregulated in hepatocellular carcinoma. Oncogene. 2003; 22:5060–5069.
Article11. Liu X, Zhou R, Zhang Q, Zhang Y, Shao G, Jin Y, et al. Identification and characterization of LAPTM4B encoded by a human hepatocellular carcinoma-associated novel gene. Beijing Da Xue Xue Bao. 2003; 35:340–347.12. Li Y, Zou L, Li Q, Haibe-Kains B, Tian R, Li Y, et al. Amplification of LAPTM4B and YWHAZ contributes to chemotherapy resistance and recurrence of breast cancer. Nat Med. 2010; 16:214–218.
Article13. Xiao M, Jia S, Wang H, Wang J, Huang Y, Li Z. Overexpression of LAPTM4B: an independent prognostic marker in breast cancer. J Cancer Res Clin Oncol. 2013; 139:661–667.
Article14. Fan M, Liu Y, Zhou R, Zhang Q. Association of LAPTM4B gene polymorphism with breast cancer susceptibility. Cancer Epidemiol. 2012; 36:364–368.
Article15. Zhou L, He XD, Yu JC, Zhou RL, Shan Y, Rui JA. Overexpression of LAPTM4B-35 attenuates epirubucin-induced apoptosis of gallbladder carcinoma GBC-SD cells. Surgery. 2011; 150:25–31.
Article16. Kang Y, Yin M, Jiang W, Zhang H, Xia B, Xue Y, et al. Overexpression of LAPTM4B-35 is associated with poor prognosis in colorectal carcinoma. Am J Surg. 2012; 204:677–683.
Article17. Gomez-Pinillos A, Ferrari AC. mTOR signaling pathway and mTOR inhibitors in cancer therapy. Hematol Oncol Clin North Am. 2012; 26:483–505.
Article18. Cordes N, Rödel F, Rodemann HP. Molecular signaling pathways. Mechanisms and clinical use. Strahlenther Onkol. 2012; 188:Suppl 3. 308–311.19. Chen PS, Shih YW, Huang HC, Cheng HW. Diosgenin, a steroidal saponin, inhibits migration and invasion of human prostate cancer PC-3 cells by reducing matrix metalloproteinases expression. PLoS One. 2011; 6:e20164.
Article20. González-Arriaga P, Pascual T, García-Alvarez A, Fernández-Somoano A, López-Cima MF, Tardón A. Genetic polymorphisms in MMP 2, 9 and 3 genes modify lung cancer risk and survival. BMC Cancer. 2012; 12:121.
Article21. Akhtar S, Mahjabeen I, Akram Z, Kayani MA. CYP1A1 and GSTP1 gene variations in breast cancer: a systematic review and case-control study. Fam Cancer. 2016; 15:201–214.
Article22. Baldwin RM, Bejide M, Trinkle-Mulcahy L, Côté J. Identification of the PRMT1v1 and PRMT1v2 specific interactomes by quantitative mass spectrometry in breast cancer cells. Proteomics. 2015; 15:2187–2197.
Article23. Xiao M, Yang S, Meng F, Qin Y, Yang Y, Jia S, et al. LAPTM4B predicts axillary lymph node metastasis in breast cancer and promotes breast cancer cell aggressiveness in vitro. Cell Physiol Biochem. 2017; 41:1072–1082.
Article24. Abeyrathna P, Su Y. The critical role of Akt in cardiovascular function. Vascul Pharmacol. 2015; 74:38–48.
Article25. Raina D, Kharbanda S, Kufe D. The MUC1 oncoprotein activates the anti-apoptotic phosphoinositide 3-kinase/Akt and Bcl-xL pathways in rat 3Y1 fibroblasts. J Biol Chem. 2004; 279:20607–20612.
Article26. Wang CD, Yuan CF, Bu YQ, Wu XM, Wan JY, Zhang L, et al. Fangchinoline inhibits cell proliferation via Akt/GSK-3beta/ cyclin D1 signaling and induces apoptosis in MDA-MB-231 breast cancer cells. Asian Pac J Cancer Prev. 2014; 15:769–773.
Article27. Tang C, Lu YH, Xie JH, Wang F, Zou JN, Yang JS, et al. Downregulation of survivin and activation of caspase-3 through the PI3K/Akt pathway in ursolic acid-induced HepG2 cell apoptosis. Anticancer Drugs. 2009; 20:249–258.
Article28. Wang XX, Cheng Q, Zhang SN, Qian HY, Wu JX, Tian H, et al. PAK5-Egr1-MMP2 signaling controls the migration and invasion in breast cancer cell. Tumour Biol. 2013; 34:2721–2729.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Vps34 Inhibits Hepatocellular Carcinoma Invasion by Regulating Endosome-Lysosome Trafficking via Rab7-RILP and Rab11
- Amygdalin Regulates Apoptosis and Adhesion in Hs578T Triple-Negative Breast Cancer Cells
- Diallyl Trisulfide Inhibits Leptin-induced OncogenicSignaling in Human Breast Cancer Cells but Fails toPrevent Chemically-induced Luminal-type Cancer in Rats
- Natural Products for Chemoprevention of Breast Cancer
- Clinicopathological Significance of SMAD4 Expression in Breast Cancer