Korean Circ J.
2018 Dec;48(12):1135-1144. 10.4070/kcj.2018.0061.
Protective Effect of Right Ventricular Mitochondrial Damage by Cyclosporine A in Monocrotaline-induced Pulmonary Hypertension
- Affiliations
-
- 1Department of Pediatrics, Dongguk University School of Medicine, Gyeongju, Korea. lds117@dongguk.ac.kr
- 2Department of Anatomy, Dongguk University School of Medicine, Gyeongju, Korea.
- KMID: 2424563
- DOI: http://doi.org/10.4070/kcj.2018.0061
Abstract
- BACKGROUND AND OBJECTIVES
Mitochondria play a key role in the pathophysiology of heart failure and mitochondrial permeability transition pore (MPTP) play a critical role in cell death and a critical target for cardioprotection. The aim of this study was to evaluate the protective effects of cyclosporine A (CsA), one of MPTP blockers, and morphological changes of mitochondria and MPTP related proteins in monocrotaline (MCT) induced pulmonary arterial hypertension (PAH).
METHODS
Eight weeks old Sprague-Dawley rats were randomized to control, MCT (60 mg/kg) and MCT plus CsA (10 mg/kg/day) treatment groups. Four weeks later, right ventricular hypertrophy (RVH) and morphological changes of right ventricle (RV) were done. Western blot and reverse transcription polymerase chain reaction (RT-PCR) for MPTP related protein were performed.
RESULTS
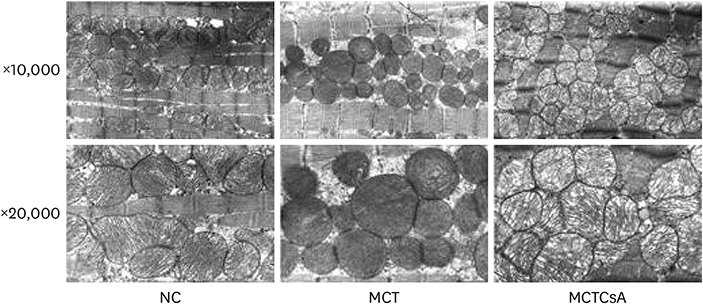
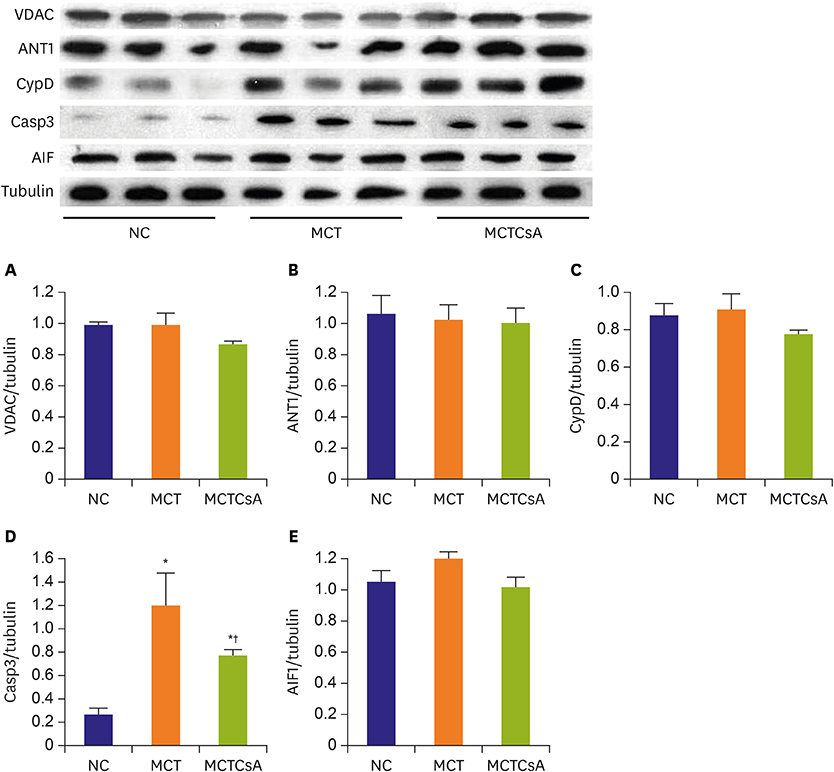
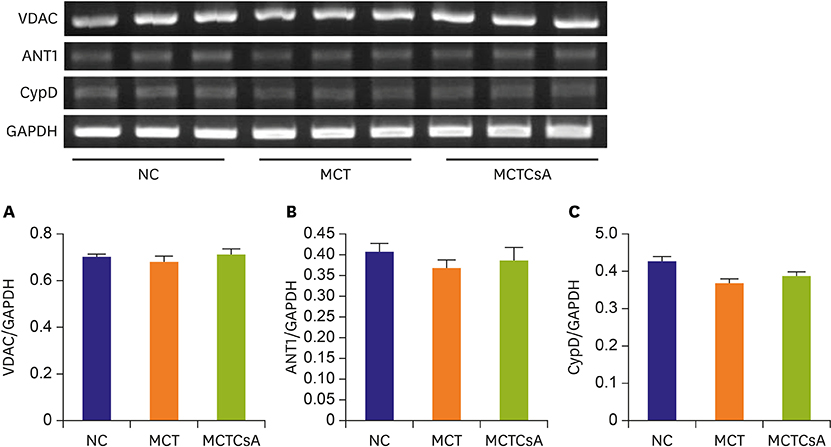
In electron microscopy, CsA treatment prevented MCT-induced mitochondrial disruption of RV. RVH was significantly increased in MCT group compared to that of the controls but RVH was more increased with CsA treatment. Thickened medial wall thickness of pulmonary arteriole in PAH was not changed after CsA treatment. In western blot, caspase-3 was significantly increased in MCT group, and was attenuated in CsA treatment. There were no significant differences in voltage-dependent anion channel, adenine nucleotide translocator 1 and cyclophilin D expression in western blot and RT-PCR between the 3 groups.
CONCLUSIONS
CsA reduces MCT induced RV mitochondrial damage. Although, MPTP blocking does not reverse pulmonary pathology, it may reduce RV dysfunction in PAH. The results suggest that it could serve as an adjunctive therapy to PAH treatment.
MeSH Terms
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
Adenine Nucleotide Translocator 1
Arterioles
Blotting, Western
Caspase 3
Cell Death
Cyclophilins
Cyclosporine*
Heart Failure
Heart Ventricles
Hypertension
Hypertension, Pulmonary*
Hypertrophy, Right Ventricular
Microscopy, Electron
Mitochondria
Monocrotaline
Pathology
Permeability
Polymerase Chain Reaction
Pulmonary Circulation
Rats, Sprague-Dawley
Reverse Transcription
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
Adenine Nucleotide Translocator 1
Caspase 3
Cyclophilins
Cyclosporine
Monocrotaline
Figure
Cited by 1 articles
-
New Therapeutic Target for Pulmonary Arterial Hypertension
Kyung Lim Yoon
Korean Circ J. 2018;48(12):1145-1147. doi: 10.4070/kcj.2018.0250.
Reference
-
1. Humbert M, Morrell NW, Archer SL, et al. Cellular and molecular pathobiology of pulmonary arterial hypertension. J Am Coll Cardiol. 2004; 43:13S–24S.
Article2. Martin KB, Klinger JR, Rounds SI. Pulmonary arterial hypertension: new insights and new hope. Respirology. 2006; 11:6–17.
Article3. Lee SH, Rubin LJ. Current treatment strategies for pulmonary arterial hypertension. J Intern Med. 2005; 258:199–215.
Article4. Rosca MG, Hoppel CL. Mitochondrial dysfunction in heart failure. Heart Fail Rev. 2013; 18:607–622.
Article5. Kwong JQ, Molkentin JD. Physiological and pathological roles of the mitochondrial permeability transition pore in the heart. Cell Metab. 2015; 21:206–214.
Article6. Haworth RA, Hunter DR. The Ca2+-induced membrane transition in mitochondria. II. Nature of the Ca2+ trigger site. Arch Biochem Biophys. 1979; 195:460–467.7. Halestrap AP. What is the mitochondrial permeability transition pore? J Mol Cell Cardiol. 2009; 46:821–831.
Article8. Crompton M, Costi A. Kinetic evidence for a heart mitochondrial pore activated by Ca2+, inorganic phosphate and oxidative stress. A potential mechanism for mitochondrial dysfunction during cellular Ca2+ overload. Eur J Biochem. 1988; 178:489–501.
Article9. Griffiths EJ, Halestrap AP. Mitochondrial non-specific pores remain closed during cardiac ischaemia, but open upon reperfusion. Biochem J. 1995; 307:93–98.
Article10. Sheu JJ, Chua S, Sun CK, et al. Intra-coronary administration of cyclosporine limits infarct size, attenuates remodeling and preserves left ventricular function in porcine acute anterior infarction. Int J Cardiol. 2011; 147:79–87.
Article11. Hausenloy DJ, Boston-Griffiths EA, Yellon DM. Cyclosporin A and cardioprotection: from investigative tool to therapeutic agent. Br J Pharmacol. 2012; 165:1235–1245.
Article12. Lim SY, Hausenloy DJ, Arjun S, et al. Mitochondrial cyclophilin-D as a potential therapeutic target for post-myocardial infarction heart failure. J Cell Mol Med. 2011; 15:2443–2451.
Article13. Bach PH, Lock EA. Nephrotoxicity: In Vitro and in Vivo, Animals to Man. 1st ed. New York (NY): Springer Science and Business Media;2013. p. 707.14. Voelkel NF, Natarajan R, Drake JI, Bogaard HJ. Right ventricle in pulmonary hypertension. Compr Physiol. 2011; 1:525–540.
Article15. Daicho T, Yagi T, Abe Y, et al. Possible involvement of mitochondrial energy-producing ability in the development of right ventricular failure in monocrotaline-induced pulmonary hypertensive rats. J Pharmacol Sci. 2009; 111:33–43.
Article16. Hong YM, Kwon JH, Choi S, Kim KC. Apoptosis and inflammation associated gene expressions in monocrotaline-induced pulmonary hypertensive rats after bosentan treatment. Korean Circ J. 2014; 44:97–104.
Article17. Campian ME, Verberne HJ, Hardziyenka M, et al. Serial noninvasive assessment of apoptosis during right ventricular disease progression in rats. J Nucl Med. 2009; 50:1371–1377.
Article18. Zuo XR, Wang Q, Cao Q, et al. Nicorandil prevents right ventricular remodeling by inhibiting apoptosis and lowering pressure overload in rats with pulmonary arterial hypertension. PLoS One. 2012; 7:e44485.
Article19. Zungu-Edmondson M, Shults NV, Wong CM, Suzuki YJ. Modulators of right ventricular apoptosis and contractility in a rat model of pulmonary hypertension. Cardiovasc Res. 2016; 110:30–39.
Article20. Kinnally KW, Peixoto PM, Ryu SY, Dejean LM. Is mPTP the gatekeeper for necrosis, apoptosis, or both? Biochim Biophys Acta. 2011; 1813:616–622.
Article21. Cregan SP, Dawson VL, Slack RS. Role of AIF in caspase-dependent and caspase-independent cell death. Oncogene. 2004; 23:2785–2796.
Article22. Halestrap AP, Richardson AP. The mitochondrial permeability transition: a current perspective on its identity and role in ischaemia/reperfusion injury. J Mol Cell Cardiol. 2015; 78:129–141.
Article23. Baines CP, Kaiser RA, Purcell NH, et al. Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature. 2005; 434:658–662.
Article24. Ramachandran A, Lebofsky M, Baines CP, Lemasters JJ, Jaeschke H. Cyclophilin D deficiency protects against acetaminophen-induced oxidant stress and liver injury. Free Radic Res. 2011; 45:156–164.
Article25. Lim WY, Messow CM, Berry C. Cyclosporin variably and inconsistently reduces infarct size in experimental models of reperfused myocardial infarction: a systematic review and meta-analysis. Br J Pharmacol. 2012; 165:2034–2043.
Article26. Gomez L, Li B, Mewton N, et al. Inhibition of mitochondrial permeability transition pore opening: translation to patients. Cardiovasc Res. 2009; 83:226–233.
Article27. Piot C, Croisille P, Staat P, et al. Effect of cyclosporine on reperfusion injury in acute myocardial infarction. N Engl J Med. 2008; 359:473–481.
Article28. Ghaffari S, Kazemi B, Toluey M, Sepehrvand N. The effect of prethrombolytic cyclosporine-A injection on clinical outcome of acute anterior ST-elevation myocardial infarction. Cardiovasc Ther. 2013; 31:e34–e39.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Angiotensin-(1-9) ameliorates pulmonary arterial hypertension via angiotensin type II receptor
- The Protective Effect of Simvastatin on Monocrotaline-Induced Pulmonary Hypertension in Rats
- Changes of Pulmonary Pathology and Gene Expressions After Simvastatin Treatment in the Monocrotaline-Induced Pulmonary Hypertension Rat Model
- Enhancement of cyclosporine-induced oxidative damage of kidney mitochondria by iron
- Effect of endothelin receptor blockade on monocrotaline-induced pulmonary hypertension in rats