Korean J Physiol Pharmacol.
2017 Sep;21(5):547-554. 10.4196/kjpp.2017.21.5.547.
Hydroquinone suppresses IFN-β expression by targeting AKT/IRF3 pathway
- Affiliations
-
- 1Department of Genetic Engineering, Sungkyunkwan University, Suwon 16419, Korea. kjhmlkjhml@hanmail.net, jaecho@skku.edu
- 2Department of Information Statistics, Kangwon National University, Chucheon 24341, Korea.
- 3Department of Pharmaceutical Engineering, Cheongju University, Cheongju 28503, Korea. ysyi@cjy.ac.kr
- KMID: 2388699
- DOI: http://doi.org/10.4196/kjpp.2017.21.5.547
Abstract
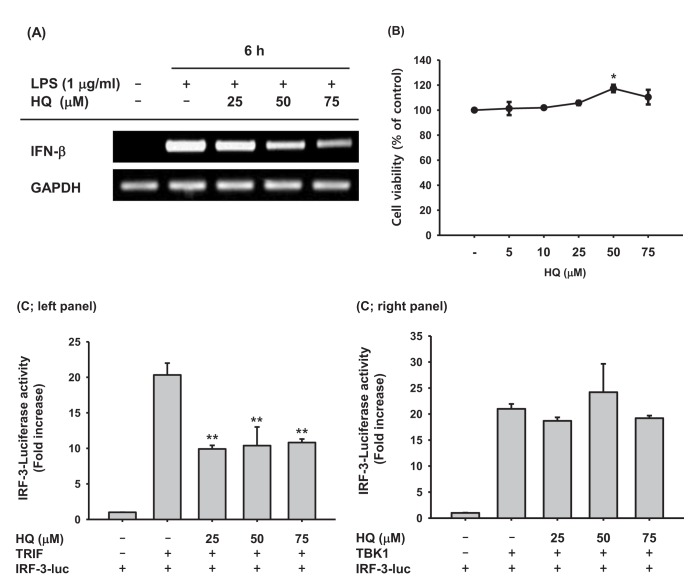
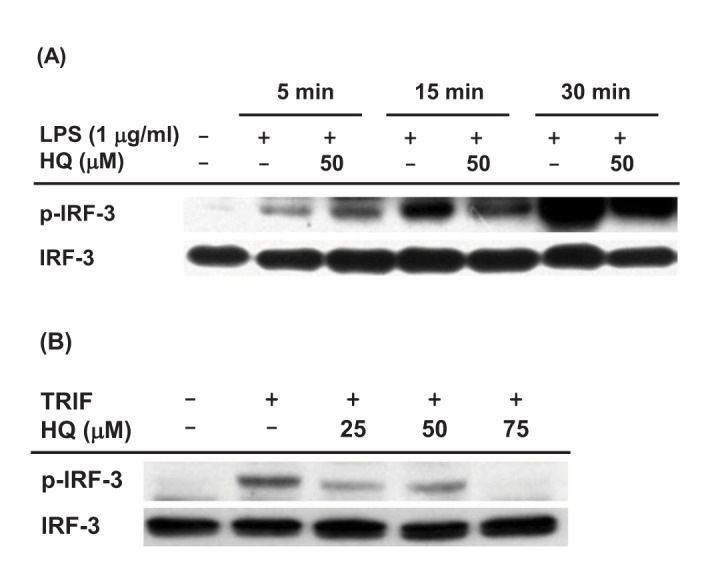
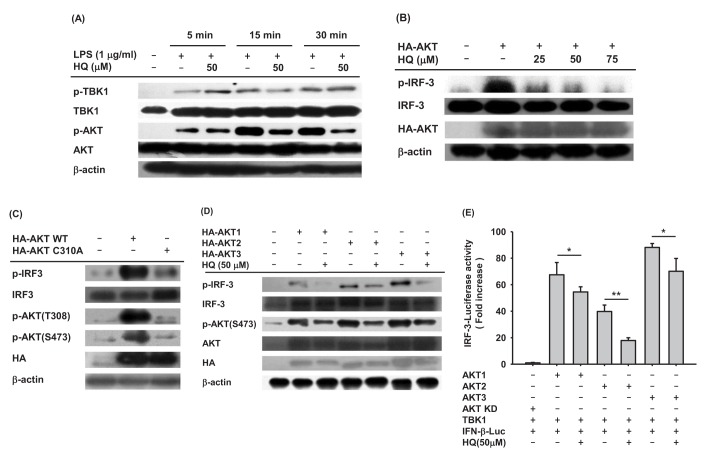
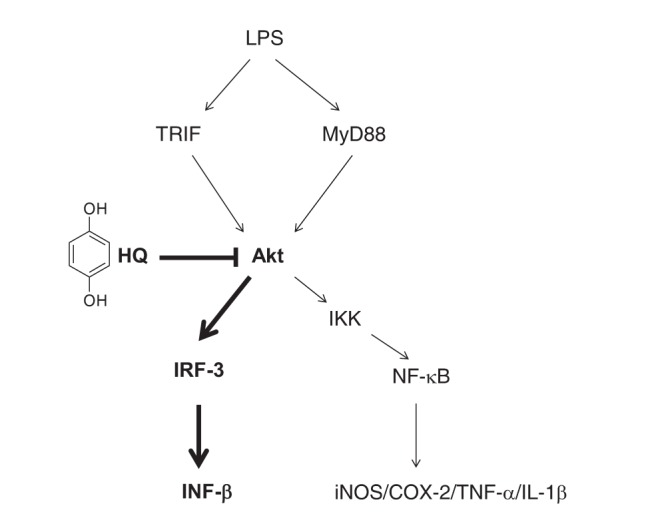
- Previous studies have demonstrated the role of hydroquinone (HQ), a hydroxylated benzene metabolite, in modulating various immune responses; however, its role in macrophage-mediated inflammatory responses is not fully understood. In this study, the role of HQ in inflammatory responses and the underlying molecular mechanism were explored in macrophages. HQ down-regulated the expression of interferon (IFN)-β mRNA in LPS-stimulated RAW264.7 cells without any cytotoxicity and suppressed interferon regulatory factor (IRF)-3-mediated luciferase activity induced by TIR-domain-containing adapter-inducing interferon-β (TRIF) and TANK-binding kinase 1 (TBK1). A mechanism study revealed that HQ inhibited IRF-3 phosphorylation induced by lipopolysaccharide (LPS), TRIF, and AKT by suppressing phosphorylation of AKT, an upstream kinase of the IRF-3 signaling pathway. IRF-3 phosphorylation is highly induced by wild-type AKT and poorly induced by an AKT mutant, AKT C310A, which is mutated at an inhibitory target site of HQ. We also showed that HQ inhibited IRF-3 phosphorylation by targeting all three AKT isoforms (AKT1, AKT2, and AKT3) in RAW264.7 cells and suppressed IRF-3-mediated luciferase activities induced by AKT in HEK293 cells. Taken together, these results strongly suggest that HQ inhibits the production of a type I IFN, IFN-β, by targeting AKTs in the IRF-3 signaling pathway during macrophage-mediated inflammation.
Keyword
MeSH Terms
Figure
Reference
-
1. Ferrero-Miliani L, Nielsen OH, Andersen PS, Girardin SE. Chronic inflammation: importance of NOD2 and NALP3 in interleukin-1beta generation. Clin Exp Immunol. 2007; 147:227–235. PMID: 17223962.2. Kaur M, Singh M, Silakari O. Inhibitors of switch kinase ‘spleen tyrosine kinase’ in inflammation and immune-mediated disorders: a review. Eur J Med Chem. 2013; 67:434–446. PMID: 23917087.
Article3. Baek KS, Yi YS, Son YJ, Yoo S, Sung NY, Kim Y, Hong S, Aravinthan A, Kim JH, Cho JY. In vitro and in vivo anti-inflammatory activities of Korean Red Ginseng-derived components. J Ginseng Res. 2016; 40:437–444. PMID: 27746698.4. Vance RE, Isberg RR, Portnoy DA. Patterns of pathogenesis: discrimination of pathogenic and nonpathogenic microbes by the innate immune system. Cell Host Microbe. 2009; 6:10–21. PMID: 19616762.
Article5. Janeway CA Jr, Medzhitov R. Innate immune recognition. Annu Rev Immunol. 2002; 20:197–216. PMID: 11861602.
Article6. Pandey S, Kawai T, Akira S. Microbial sensing by Toll-like receptors and intracellular nucleic acid sensors. Cold Spring Harb Perspect Biol. 2014; 7:a016246. PMID: 25301932.
Article7. Zhao H, Gonzalezgugel E, Cheng L, Richbourgh B, Nie L, Liu C. The roles of interferon-inducible p200 family members IFI16 and p204 in innate immune responses, cell differentiation and proliferation. Genes Dis. 2015; 2:46–56. PMID: 25815367.
Article8. Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol. 2001; 2:675–680. PMID: 11477402.
Article9. Massarotti EM. Clinical and patient-reported outcomes in clinical trials of abatacept in the treatment of rheumatoid arthritis. Clin Ther. 2008; 30:429–442. PMID: 18405783.
Article10. Vo VA, Lee JW, Chang JE, Kim JY, Kim NH, Lee HJ, Kim SS, Chun W, Kwon YS. Avicularin inhibits lipopolysaccharide-induced inflammatory response by suppressing ERK phosphorylation in RAW 264.7 macrophages. Biomol Ther (Seoul). 2012; 20:532–537. PMID: 24009846.
Article11. Taylor PR, Martinez-Pomares L, Stacey M, Lin HH, Brown GD, Gordon S. Macrophage receptors and immune recognition. Annu Rev Immunol. 2005; 23:901–944. PMID: 15771589.
Article12. Kwon IS, Yim JH, Lee HK, Pyo S. Lobaric acid inhibits VCAM-1 expression in TNF-α-stimulated vascular smooth muscle cells via modulation of NF-κB and MAPK signaling pathways. Biomol Ther (Seoul). 2016; 24:25–32. PMID: 26759698.
Article13. Byeon SE, Yi YS, Oh J, Yoo BC, Hong S, Cho JY. The role of Src kinase in macrophage-mediated inflammatory responses. Mediators Inflamm. 2012; 2012:512926. PMID: 23209344.
Article14. Yi YS, Son YJ, Ryou C, Sung GH, Kim JH, Cho JY. Functional roles of Syk in macrophage-mediated inflammatory responses. Mediators Inflamm. 2014; 2014:270302. PMID: 25045209.
Article15. Yu T, Yi YS, Yang Y, Oh J, Jeong D, Cho JY. The pivotal role of TBK1 in inflammatory responses mediated by macrophages. Mediators Inflamm. 2012; 2012:979105. PMID: 23304064.
Article16. Yang Y, Kim SC, Yu T, Yi YS, Rhee MH, Sung GH, Yoo BC, Cho JY. Functional roles of p38 mitogen-activated protein kinase in macrophage-mediated inflammatory responses. Mediators Inflamm. 2014; 2014:352371. PMID: 24771982.
Article17. Yang S, Kim Y, Jeong D, Kim JH, Kim S, Son YJ, Yoo BC, Jeong EJ, Kim TW, Lee IH, Cho JY. Pyrrole-derivative of chalcone, (E)-3-phenyl-1-(2-pyrrolyl)-2-propenone, inhibits inflammatory responses via inhibition of Src, Syk, and TAK1 kinase activities. Biomol Ther (Seoul). 2016; 24:595–603. PMID: 27469142.
Article18. DeCaprio AP. The toxicology of hydroquinone−relevance to occupational and environmental exposure. Crit Rev Toxicol. 1999; 29:283–330. PMID: 10379810.19. McDonald TA, Holland NT, Skibola C, Duramad P, Smith MT. Hypothesis: phenol and hydroquinone derived mainly from diet and gastrointestinal flora activity are causal factors in leukemia. Leukemia. 2001; 15:10–20. PMID: 11243376.
Article20. Moerloose KB, Pauwels RA, Joos GF. Short-term cigarette smoke exposure enhances allergic airway inflammation in mice. Am J Respir Crit Care Med. 2005; 172:168–172. PMID: 15831841.
Article21. Kim E, Kang BY, Kim TS. Inhibition of interleukin-12 production in mouse macrophages by hydroquinone, a reactive metabolite of benzene, via suppression of nuclear factor-kappaB binding activity. Immunol Lett. 2005; 99:24–29. PMID: 15894107.22. Kim AR, Cho JY, Lee JY, Choi JS, Chung HY. Hydroquinone modulates reactivity of peroxynitrite and nitric oxide production. J Pharm Pharmacol. 2005; 57:475–481. PMID: 15831208.
Article23. Pyatt DW, Yang Y, Stillman WS, Cano LL, Irons RD. Hydroquinone inhibits PMA-induced activation of NFkappaB in primary human CD19+ B lymphocytes. Cell Biol Toxicol. 2000; 16:41–51. PMID: 10890505.24. Lee JY, Lee YG, Lee J, Yang KJ, Kim AR, Kim JY, Won MH, Park J, Yoo BC, Kim S, Cho WJ, Cho JY. Akt Cys-310-targeted inhibition by hydroxylated benzene derivatives is tightly linked to their immunosuppressive effects. J Biol Chem. 2010; 285:9932–9948. PMID: 20054000.
Article25. Ma Q, Kinneer K, Ye J, Chen BJ. Inhibition of nuclear factor kappaB by phenolic antioxidants: interplay between antioxidant signaling and inflammatory cytokine expression. Mol Pharmacol. 2003; 64:211–219. PMID: 12869625.26. Lee JY, Kim JY, Lee YG, Shin WC, Chun T, Rhee MH, Cho JY. Hydroquinone, a reactive metabolite of benzene, reduces macrophage-mediated immune responses. Mol Cells. 2007; 23:198–206. PMID: 17464197.27. Cho JY. Suppressive effect of hydroquinone, a benzene metabolite, on in vitro inflammatory responses mediated by macrophages, monocytes, and lymphocytes. Mediators Inflamm. 2008; 2008:298010. PMID: 19148301.
Article28. Yang Y, Yang WS, Yu T, Yi YS, Park JG, Jeong D, Kim JH, Oh JS, Yoon K, Kim JH, Cho JY. Novel anti-inflammatory function of NSC95397 by the suppression of multiple kinases. Biochem Pharmacol. 2014; 88:201–215. PMID: 24468133.
Article29. Keung MH, Chan LS, Kwok HH, Wong RN, Yue PY. Role of microRNA-520h in 20(R)-ginsenoside-Rg3-mediated angiosuppression. J Ginseng Res. 2016; 40:151–159. PMID: 27158236.
Article30. Yi YS, Cho JY, Kim D. Cerbera manghas methanol extract exerts anti-inflammatory activity by targeting c-Jun N-terminal kinase in the AP-1 pathway. J Ethnopharmacol. 2016; 193:387–396. PMID: 27562319.
Article31. Baek KS, Hong YD, Kim Y, Sung NY, Yang S, Lee KM, Park JY, Park JS, Rho HS, Shin SS, Cho JY. Anti-inflammatory activity of AP-SF, a ginsenoside-enriched fraction, from Korean ginseng. J Ginseng Res. 2015; 39:155–161. PMID: 26045689.
Article32. Gerlier D, Thomasset N. Use of MTT colorimetric assay to measure cell activation. J Immunol Methods. 1986; 94:57–63. PMID: 3782817.
Article33. Park JG, Son YJ, Aravinthan A, Kim JH, Cho JY. Korean Red Ginseng water extract arrests growth of xenografted lymphoma cells. J Ginseng Res. 2016; 40:431–436. PMID: 27746697.
Article34. Jeong D, Yi YS, Sung GH, Yang WS, Park JG, Yoon K, Yoon DH, Song C, Lee Y, Rhee MH, Kim TW, Kim JH, Cho JY. Anti-inflammatory activities and mechanisms of Artemisia asiatica ethanol extract. J Ethnopharmacol. 2014; 152:487–496. PMID: 24503036.
Article35. Yang Y, Lee J, Rhee MH, Yu T, Baek KS, Sung NY, Kim Y, Yoon K, Kim JH, Kwak YS, Hong S, Kim JH, Cho JY. Molecular mechanism of protopanaxadiol saponin fraction-mediated anti-inflammatory actions. J Ginseng Res. 2015; 39:61–68. PMID: 25535478.
Article36. Lee MH, Chung SW, Kang BY, Kim KM, Kim TS. Hydroquinone, a reactive metabolite of benzene, enhances interleukin-4 production in CD4+ T cells and increases immunoglobulin E levels in antigen-primed mice. Immunology. 2002; 106:496–502. PMID: 12153512.
Article37. Kim S, Oh MH, Kim BS, Kim WI, Cho HS, Park BY, Park C, Shin GW, Kwon J. Upregulation of heme oxygenase-1 by ginsenoside Ro attenuates lipopolysaccharide-induced inflammation in macrophage cells. J Ginseng Res. 2015; 39:365–370. PMID: 26869829.
Article38. Shen T, Yang WS, Yi YS, Sung GH, Rhee MH, Poo H, Kim MY, Kim KW, Kim JH, Cho JY. AP-1/IRF-3 targeted anti-inflammatory activity of andrographolide isolated from andrographis paniculata. Evid Based Complement Alternat Med. 2013; 2013:210736. PMID: 23840248.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- JS-III-49, a hydroquinone derivative, exerts anti-inflammatory activity by targeting Akt and p38
- Inhibition of the interaction between Hippo/YAP and Akt signaling with ursolic acid and 3′3-diindolylmethane suppresses esophageal cancer tumorigenesis
- Identification of Small GTPases That Phosphorylate IRF3 through TBK1 Activation Using an Active Mutant Library Screen
- miR-450a-5p Eliminates MGO-Induced Insulin Resistance via Targeting CREB
- Allergic contact dermatitis to hydroquinone in bleaching cream