J Dent Anesth Pain Med.
2017 Mar;17(1):21-28. 10.17245/jdapm.2017.17.1.21.
Propofol protects human keratinocytes from oxidative stress via autophagy expression
- Affiliations
-
- 1Department of Dental Anesthesia and Pain Medicine, School of Dentistry, Pusan National University, Dental Research Institute, Yangsan, Republic of Korea. dentiwon@hanmail.net
- 2Department of Anesthesia and Pain Medicine, School of Medicine, Pusan National University, Yangsan, Republic of Korea.
- 3Department of Oral Anatomy, School of Dentistry, Pusan National University, Yangsan, Republic of Korea.
- KMID: 2374720
- DOI: http://doi.org/10.17245/jdapm.2017.17.1.21
Abstract
- BACKGROUND
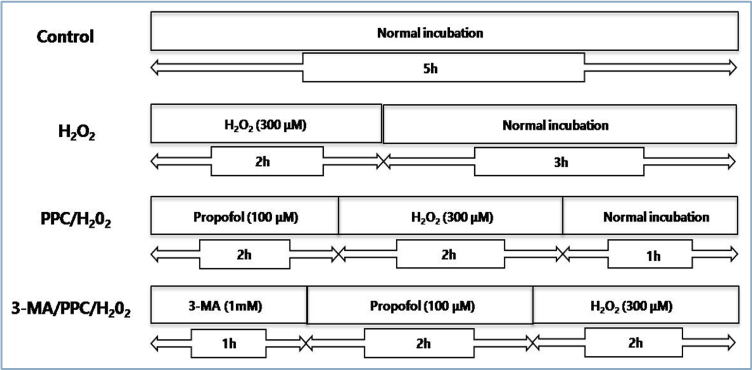
The skin consists of tightly connected keratinocytes, and prevents extensive water loss while simultaneously protecting against the entry of microbial pathogens. Excessive cellular levels of reactive oxygen species can induce cell apoptosis and also damage skin integrity. Propofol (2,6-diisopropylphenol) has antioxidant properties. In this study, we investigated how propofol influences intracellular autophagy and apoptotic cell death induced by oxidative stress in human keratinocytes. METHOD: The following groups were used for experimentation: control, cells were incubated under normoxia (5% COâ‚‚, 21% Oâ‚‚, and 74% Nâ‚‚) without propofol; hydrogen peroxide (Hâ‚‚Oâ‚‚), cells were exposed to Hâ‚‚Oâ‚‚ (300 µM) for 2 h; propofol preconditioning (PPC)/Hâ‚‚Oâ‚‚, cells pretreated with propofol (100 µM) for 2 h were exposed to Hâ‚‚Oâ‚‚; and 3-methyladenine (3-MA)/PPC/Hâ‚‚Oâ‚‚, cells pretreated with 3-MA (1 mM) for 1 h and propofol were exposed to Hâ‚‚Oâ‚‚. Cell viability, apoptosis, and migration capability were evaluated. Relation to autophagy was detected by western blot analysis.
RESULTS
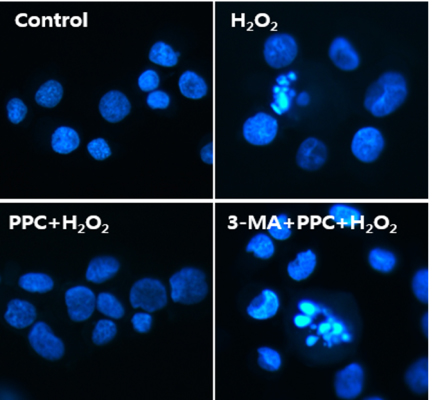
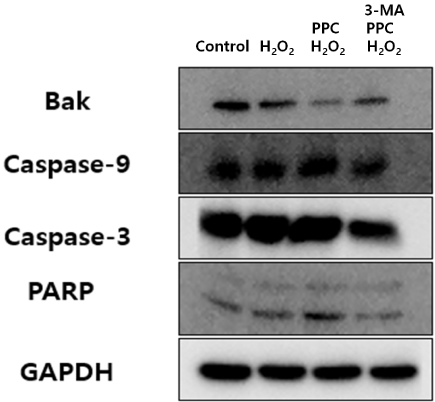
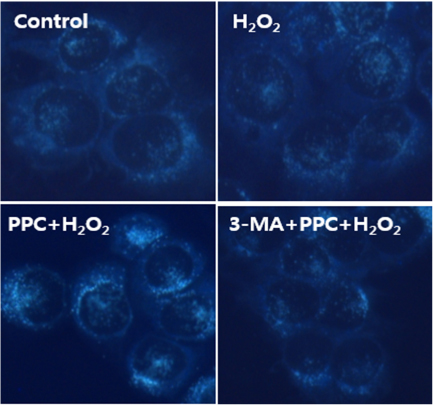
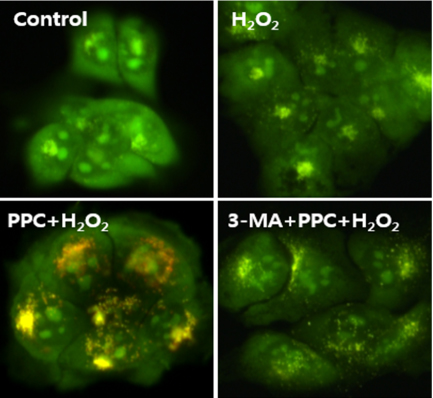
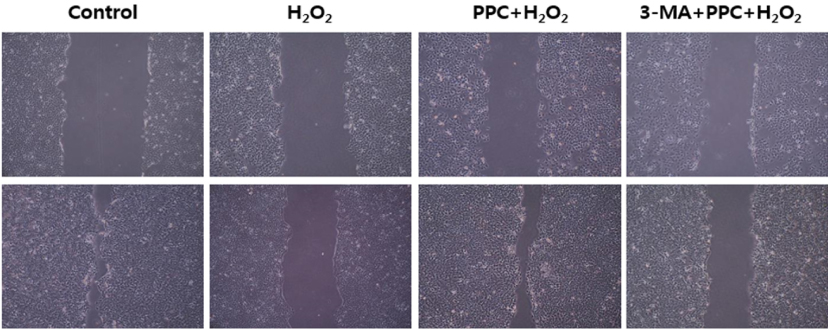
Cell viability decreased significantly in the Hâ‚‚Oâ‚‚ group compared to that in the control group and was improved by propofol preconditioning. Propofol preconditioning effectively decreased Hâ‚‚Oâ‚‚-induced cell apoptosis and increased cell migration. However, pretreatment with 3-MA inhibited the protective effect of propofol on cell apoptosis. Autophagy was activated in the PPC/Hâ‚‚Oâ‚‚ group compared to that in the Hâ‚‚Oâ‚‚ group as demonstrated by western blot analysis and autophagosome staining.
CONCLUSION
The results suggest that propofol preconditioning induces an endogenous cellular protective effect in human keratinocytes against oxidative stress through the activation of signaling pathways related to autophagy.
Keyword
MeSH Terms
Figure
Reference
-
1. Proksch E, Folster-Holst R, Brautigam M, Sepehrmanesh M, Pfeiffer S, Jensen JM. Role of the epidermal barrier in atopic dermatitis. J Dtsch Dermatol Ges. 2009; 7:899–910.
Article2. Schafer M, Werner S. Oxidative stress in normal and impaired wound repair. Pharmacol Res. 2008; 58:165–171.
Article3. Dunham LJ. Cancer in man at site of prior benign lesion of skin or mucous membrane: a review. Cancer Res. 1972; 32:1359–1374.4. Gulcin I, Alici HA, Cesur M. Determination of in vitro antioxidant and radical scavenging activities of propofol. Chem Pharm Bull. 2005; 53:281–285.
Article5. Wang B, Shravah J, Luo H, Raedschelders K, Chen DD, Ansley DM. Propofol protects against hydrogen peroxide-induced injury in cardiac H9c2 cells via Akt activation and Bcl-2 up-regulation. Biochem Biophys Res Commun. 2009; 389:105–111.
Article6. Jin YC, Kim W, Ha YM, Shin IW, Sohn JT, Kim HJ, et al. Propofol limits rat myocardial ischemia and reperfusion injury with an associated reduction in apoptotic cell death in vivo. Vascul Pharmacol. 2009; 50:71–77.
Article7. Svobodova A, Zdarilova A, Maliskova J, Mikulkova H, Walterova D, Vostalova J. Attenuation of UVA-induced damage to human keratinocytes by silymarin. J Dermatol Sci. 2007; 46:21–30.
Article8. Andreassi M, Andreassi L. Antioxidants in dermocosmetology: from the laboratory to clinical application. J Cosmet Dermatol. 2003; 2:153–160.
Article9. Pillai S, Oresajo C, Hayward J. Ultraviolet radiation and skin aging: roles of reactive oxygen species, inflammation and protease activation, and strategies for prevention of inflammation-induced matrix degradation - a review. Int J Cosmet Sci. 2005; 27:17–34.
Article10. Syed DN, Malik A, Hadi N, Sarfaraz S, Afaq F, Mukhtar H. Photochemopreventive effect of pomegranate fruit extract on UVA-mediated activation of cellular pathways in normal human epidermal keratinocytes. Photochem Photobiol. 2006; 82:398–405.
Article11. Lippens S, Denecker G, Ovaere P, Vandenabeele P, Declercq W. Death penalty for keratinocytes: apoptosis versus cornification. Cell Death Differ. 2005; 12:1497–1508.
Article12. Kulms D, Schwarz T. Molecular mechanisms of UV-induced apoptosis. Photodermatol Photoimmunol Photomed. 2000; 16:195–201.
Article13. Levine B, Klionsky DJ. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell. 2004; 6:463–477.14. Corcoran TB, Engel A, Sakamoto H, O'Shea A, O'Callaghan-Enright S, Shorten GD. The effects of propofol on neutrophil function, lipid peroxidation and inflammatory response during elective coronary artery bypass grafting in patients with impaired ventricular function. Br J Anaesth. 2006; 97:825–831.
Article15. Murphy PG, Bennett JR, Myers DS, Davies MJ, Jones JG. The effect of propofol anaesthesia on free radical-induced lipid peroxidation in rat liver microsomes. Eur J Anaesthesiol. 1993; 10:261–266.16. Wickley PJ, Shiga T, Murray PA, Damron DS. Propofol decreases myofilament Ca2+ sensitivity via a protein kinase C-, nitric oxide synthase-dependent pathway in diabetic cardiomyocytes. Anesthesiology. 2006; 104:978–987.
Article17. Lakhani SA, Masud A, Kuida K, Porter GA Jr, Booth CJ, Mehal WZ, et al. Caspases 3 and 7: key mediators of mitochondrial events of apoptosis. Science. 2006; 311:847–851.
Article18. Cepero E, King AM, Coffey LM, Perez RG, Boise LH. Caspase-9 and effector caspases have sequential and distinct effects on mitochondria. Oncogene. 2005; 24:6354–6366.
Article19. Sedlak TW, Oltvai ZN, Yang E, Wang K, Boise LH, Thompson CB, et al. Multiple Bcl-2 family members demonstrate selective dimerizations with Bax. Proc Natl Acad Sci U S A. 1995; 92:7834–7838.
Article20. Santoro MM, Gaudino G. Cellular and molecular facets of keratinocyte reepithelization during wound healing. Exp Cell Res. 2005; 304:274–286.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Propofol protects against oxidative-stress-induced COS-7 cell apoptosis by inducing autophagy
- Effects of propofol-induced autophagy against oxidative stress in human osteoblasts
- Expression of Antioxidant Response and Autophagy Markers in Patients with Vitiligo: An Immunohistochemical Study
- Aquaporin-3 Downregulation in Vitiligo Keratinocytes Increases Oxidative Stress of Melanocytes
- Protective Effect of Propofol against Hypoxia-reoxygenation Injury in HaCaT Human Keratinocytes