J Korean Med Sci.
2017 Mar;32(3):542-551. 10.3346/jkms.2017.32.3.542.
The Effects of Remifentanil on Expression of High Mobility Group Box 1 in Septic Rats
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Saint Vincent's Hospital, The College of Medicine, The Catholic University of Korea, Suwon, Korea. joojd@catholic.ac.kr
- KMID: 2366654
- DOI: http://doi.org/10.3346/jkms.2017.32.3.542
Abstract
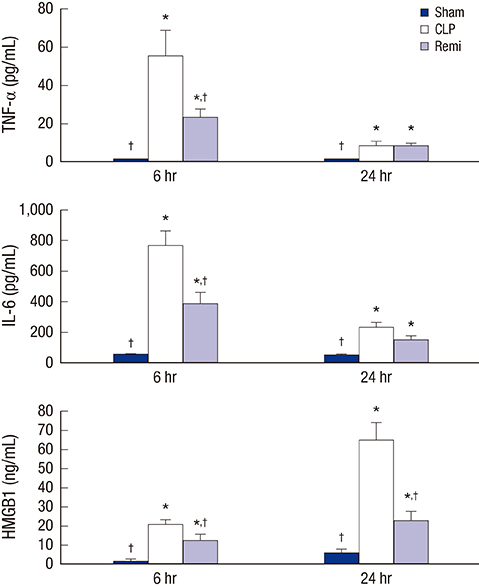
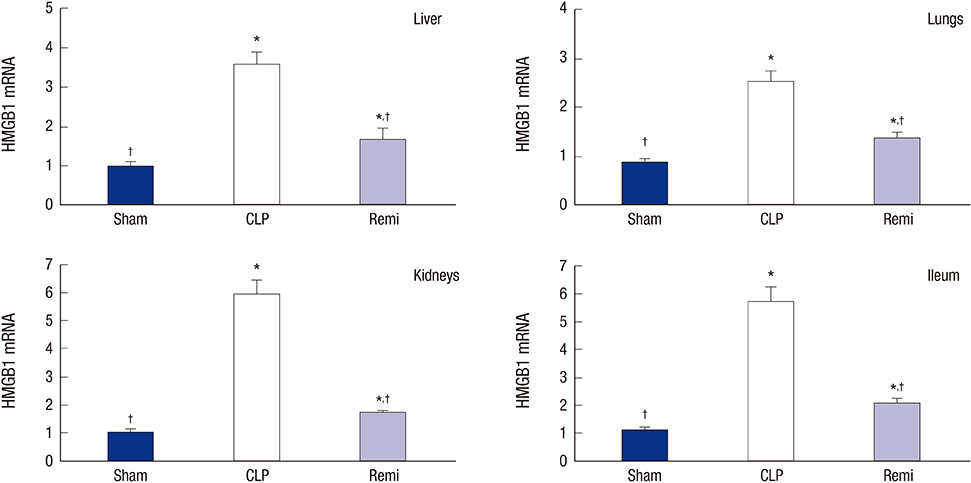
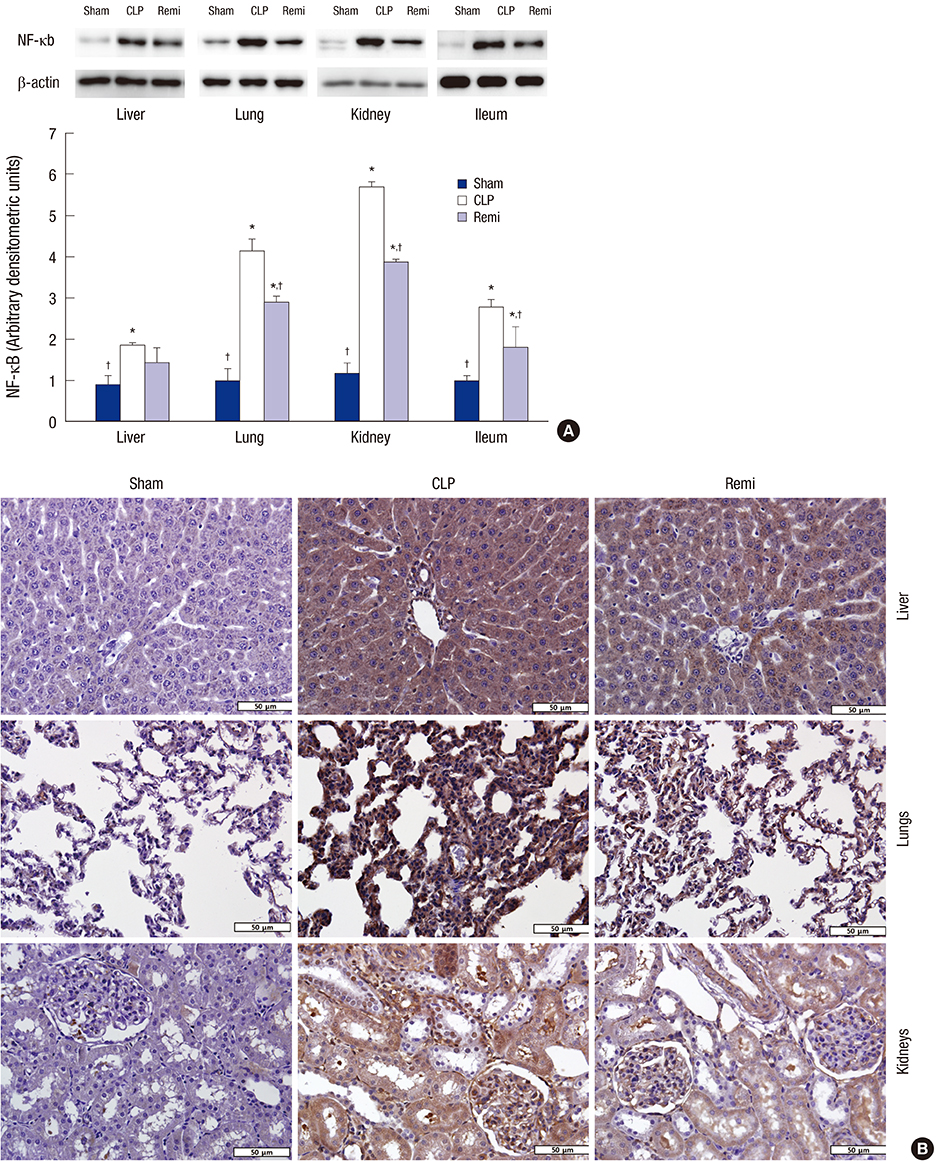
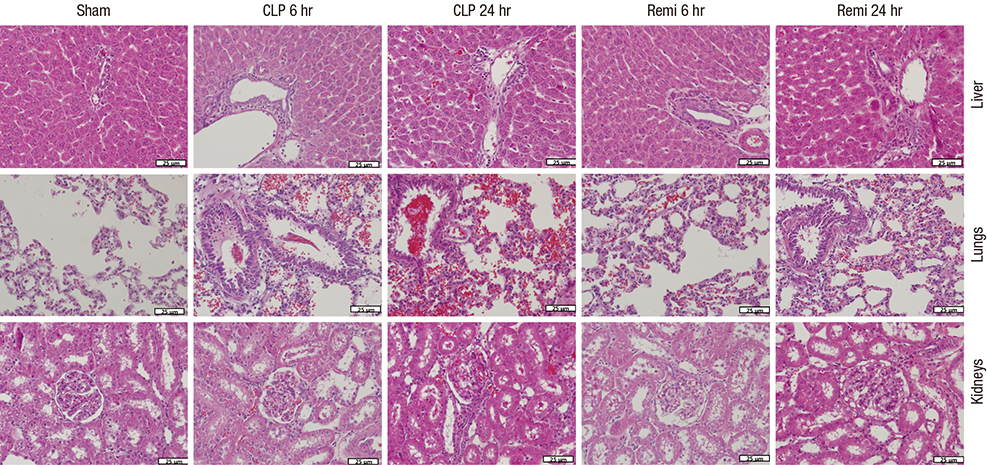
- High mobility group box 1 (HMGB1) is a pivotal mediator of sepsis progression. Remifentanil, an opioid agonist, has demonstrated anti-inflammatory effects in septic mice. However, it is not yet known whether remifentanil affects the expression of HMGB1. We investigated the effects of remifentanil on HMGB1 expression and the underlying mechanism in septic rats. Forty-eight male Sprague-Dawley rats were randomly divided into 3 groups; a sham group, a cecal ligation and puncture (CLP) group, and a CLP with remifentanil treatment (Remi) group. The rat model of CLP was used to examine plasma concentrations of proinflammatory cytokines, tissue HMGB1 mRNA and the activity of nuclear factor (NF)-κB in the liver, lungs, kidneys, and ileum. Pathologic changes and immunohistochemical staining of NF-κB in the liver, lungs, and kidneys tissue were observed. We found that remifentanil treatment suppressed the level of serum interleukin (IL)-6 and tumor necrosis factor (TNF)-α 6 hours after CLP, and serum HMGB1 24 hours after CLP. HMGB1 mRNA levels and the activity of NF-κB in multiple organs decreased by remifentanil treatment 24 hours after CLP. Remifentanil treatment also attenuated nuclear expression of NF-κB in immunohistochemical staining and mitigated pathologic changes in multiple organs. Altogether, these results suggested that remifentanil inhibited expression of HMGB1 in vital organs and release of HMGB1 into plasma. The mechanism was related to the inhibitory effect of remifentanil on the release of proinflammatory cytokines and activation of NF-κB.
Keyword
MeSH Terms
Figure
Reference
-
1. Sagy M, Al-Qaqaa Y, Kim P. Definitions and pathophysiology of sepsis. Curr Probl Pediatr Adolesc Health Care. 2013; 43:260–263.2. van Beijnum JR, Buurman WA, Griffioen AW. Convergence and amplification of toll-like receptor (TLR) and receptor for advanced glycation end products (RAGE) signaling pathways via high mobility group B1 (HMGB1). Angiogenesis. 2008; 11:91–99.3. Wang H, Yang H, Tracey KJ. Extracellular role of HMGB1 in inflammation and sepsis. J Intern Med. 2004; 255:320–331.4. Gibot S, Massin F, Cravoisy A, Barraud D, Nace L, Levy B, Bollaert PE. High-mobility group box 1 protein plasma concentrations during septic shock. Intensive Care Med. 2007; 33:1347–1353.5. Hou LC, Qin MZ, Zheng LN, Lu Y, Wang Q, Peng DR, Yu XP, Xin YC, Ji GL, Xiong LZ. Severity of sepsis is correlated with the elevation of serum high-mobility group box 1 in rats. Chin Med J (Engl). 2009; 122:449–454.6. Sundén-Cullberg J, Norrby-Teglund A, Rouhiainen A, Rauvala H, Herman G, Tracey KJ, Lee ML, Andersson J, Tokics L, Treutiger CJ. Persistent elevation of high mobility group box-1 protein (HMGB1) in patients with severe sepsis and septic shock. Crit Care Med. 2005; 33:564–573.7. Wang H, Bloom O, Zhang M, Vishnubhakat JM, Ombrellino M, Che J, Frazier A, Yang H, Ivanova S, Borovikova L, et al. HMG-1 as a late mediator of endotoxin lethality in mice. Science. 1999; 285:248–251.8. Yang H, Ochani M, Li J, Qiang X, Tanovic M, Harris HE, Susarla SM, Ulloa L, Wang H, DiRaimo R, et al. Reversing established sepsis with antagonists of endogenous high-mobility group box 1. Proc Natl Acad Sci USA. 2004; 101:296–301.9. Hofbauer R, Frass M, Gmeiner B, Sandor N, Schumann R, Wagner O, Kaye AD. Effects of remifentanil on neutrophil adhesion, transmigration, and intercellular adhesion molecule expression. Acta Anaesthesiol Scand. 2000; 44:1232–1237.10. Sacerdote P, Gaspani L, Rossoni G, Panerai AE, Bianchi M. Effect of the opioid remifentanil on cellular immune response in the rat. Int Immunopharmacol. 2001; 1:713–719.11. Zongze Z, Jia Z, Chang C, Kai C, Yanlin W. Protective effects of remifentanil on septic mice. Mol Biol Rep. 2010; 37:2803–2808.12. Zhang Y, Du Z, Zhou Q, Wang Y, Li J. Remifentanil attenuates lipopolysaccharide-induced acute lung injury by downregulating the NF-kappaB signaling pathway. Inflammation. 2014; 37:1654–1660.13. Dejager L, Pinheiro I, Dejonckheere E, Libert C. Cecal ligation and puncture: the gold standard model for polymicrobial sepsis? Trends Microbiol. 2011; 19:198–208.14. Ayala A, Chaudry IH. Immune dysfunction in murine polymicrobial sepsis: mediators, macrophages, lymphocytes and apoptosis. Shock. 1996; 6:Suppl 1. S27–38.15. Gårdlund B, Sjölin J, Nilsson A, Roll M, Wickerts CJ, Wretlind B. Plasma levels of cytokines in primary septic shock in humans: correlation with disease severity. J Infect Dis. 1995; 172:296–301.16. Ebong S, Call D, Nemzek J, Bolgos G, Newcomb D, Remick D. Immunopathologic alterations in murine models of sepsis of increasing severity. Infect Immun. 1999; 67:6603–6610.17. Hack CE, De Groot ER, Felt-Bersma RJ, Nuijens JH, Strack Van Schijndel RJ, Eerenberg-Belmer AJ, Thijs LG, Aarden LA. Increased plasma levels of interleukin-6 in sepsis. Blood. 1989; 74:1704–1710.18. Song XM, Wang YL, Li JG, Wang CY, Zhou Q, Zhang ZZ, Liang H. Effects of propofol on pro-inflammatory cytokines and nuclear factor kappaB during polymicrobial sepsis in rats. Mol Biol Rep. 2009; 36:2345–2351.19. Xu L, Bao H, Si Y, Wang X. Effects of dexmedetomidine on early and late cytokines during polymicrobial sepsis in mice. Inflamm Res. 2013; 62:507–514.20. Huang LF, Yao YM, Sheng ZY. Novel insights for high mobility group box 1 protein-mediated cellular immune response in sepsis: a systemic review. World J Emerg Med. 2012; 3:165–171.21. Karlsson S, Pettilä V, Tenhunen J, Laru-Sompa R, Hynninen M, Ruokonen E. HMGB1 as a predictor of organ dysfunction and outcome in patients with severe sepsis. Intensive Care Med. 2008; 34:1046–1053.22. Ji MH, Zhu XL, Liu FF, Li GM, Tian M, Wu J, Fan YX, Li N, Yang JJ. Alpha 2A-adrenoreceptor blockade improves sepsis-induced acute lung injury accompanied with depressed high mobility group box-1 levels in rats. Cytokine. 2012; 60:639–645.23. Wang HL, Xing YQ, Xu YX, Rong F, Lei WF, Zhang WH. The protective effect of lidocaine on septic rats via the inhibition of high mobility group box 1 expression and NF-kappaB activation. Mediators Inflamm. 2013; 2013:570370.24. Zhang LT, Yao YM, Lu JQ, Yan XJ, Yu Y, Sheng ZY. Sodium butyrate prevents lethality of severe sepsis in rats. Shock. 2007; 27:672–677.25. Li X, Stark GR. NFκB-dependent signaling pathways. Exp Hematol. 2002; 30:285–296.26. Sharp BM, Roy S, Bidlack JM. Evidence for opioid receptors on cells involved in host defense and the immune system. J Neuroimmunol. 1998; 83:45–56.27. Mellon RD, Bayer BM. Evidence for central opioid receptors in the immunomodulatory effects of morphine: review of potential mechanism(s) of action. J Neuroimmunol. 1998; 83:19–28.28. Hyejin J, Mei L, Seongheon L, Cheolwon J, Seokjai K, Hongbeom B, Minsun K, Sungsu C, Sanghyun K. Remifentanil attenuates human neutrophils activation induced by lipopolysaccharide. Immunopharmacol Immunotoxicol. 2013; 35:264–271.29. Ke JJ, Zhan J, Feng XB, Wu Y, Rao Y, Wang YL. A comparison of the effect of total intravenous anaesthesia with propofol and remifentanil and inhalational anaesthesia with isoflurane on the release of pro- and anti-inflammatory cytokines in patients undergoing open cholecystectomy. Anaesth Intensive Care. 2008; 36:74–78.30. von Dossow V, Luetz A, Haas A, Sawitzki B, Wernecke KD, Volk HD, Spies CD. Effects of remifentanil and fentanyl on the cell-mediated immune response in patients undergoing elective coronary artery bypass graft surgery. J Int Med Res. 2008; 36:1235–1247.31. Winterhalter M, Brandl K, Rahe-Meyer N, Osthaus A, Hecker H, Hagl C, Adams HA, Piepenbrock S. Endocrine stress response and inflammatory activation during CABG surgery. A randomized trial comparing remifentanil infusion to intermittent fentanyl. Eur J Anaesthesiol. 2008; 25:326–335.32. Cho SS, Rudloff I, Berger PJ, Irwin MG, Nold MF, Cheng W, Nold-Petry CA. Remifentanil ameliorates intestinal ischemia-reperfusion injury. BMC Gastroenterol. 2013; 13:69.33. Ge Y, Hu S, Zhang Y, Wang W, Xu Q, Zhou L, Mao H. Levobupivacaine inhibits lipopolysaccharide-induced high mobility group box 1 release in vitro and in vivo. J Surg Res. 2014; 192:582–591.34. Yu M, Shao D, Liu J, Zhu J, Zhang Z, Xu J. Effects of ketamine on levels of cytokines, NF-kappaB and TLRs in rat intestine during CLP-induced sepsis. Int Immunopharmacol. 2007; 7:1076–1082.35. Breen D, Karabinis A, Malbrain M, Morais R, Albrecht S, Jarnvig IL, Parkinson P, Kirkham AJ. Decreased duration of mechanical ventilation when comparing analgesia-based sedation using remifentanil with standard hypnotic-based sedation for up to 10 days in intensive care unit patients: a randomised trial [ISRCTN47583497]. Crit Care. 2005; 9:R200–R210.36. Battershill AJ, Keating GM. Remifentanil: a review of its analgesic and sedative use in the intensive care unit. Drugs. 2006; 66:365–385.37. Zhang Z, Zhang L, Zhou C, Wu H. Ketamine inhibits LPS-induced HGMB1 release in vitro and in vivo. Int Immunopharmacol. 2014; 23:14–26.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Up-to-date information on polymyxin B-immobilized fiber column direct hemoperfusion for septic shock
- Exploring the beneficial role of telmisartan in sepsis-induced myocardial injury through inhibition of high-mobility group box 1 and glycogen synthase kinase-3β/nuclear factor-κB pathway
- Sry-related High Mobility Group Box 17 Functions as a Tumor Suppressor by Antagonizing the Wingless-related Integration Site Pathway
- The Effects of Remifentanil on the LMA Insertion with the Administration of Propofol
- Increased Serum High Mobility Group Box 1 (HMGB1) Concentration and the Altered Expression of HMGB1 and Its Receptor Advanced Glycation Endproducts in Pemphigus