J Korean Med Sci.
2016 Apr;31(4):547-552. 10.3346/jkms.2016.31.4.547.
The Glucotoxicity Protecting Effect of Ezetimibe in Pancreatic Beta Cells via Inhibition of CD36
- Affiliations
-
- 1Department of Internal Medicine, Yeungnam University College of Medicine, Daegu, Korea. helee@med.yu.ac.kr
- 2Department of Physiology, Yeungnam University College of Medicine, Daegu, Korea.
- KMID: 2363693
- DOI: http://doi.org/10.3346/jkms.2016.31.4.547
Abstract
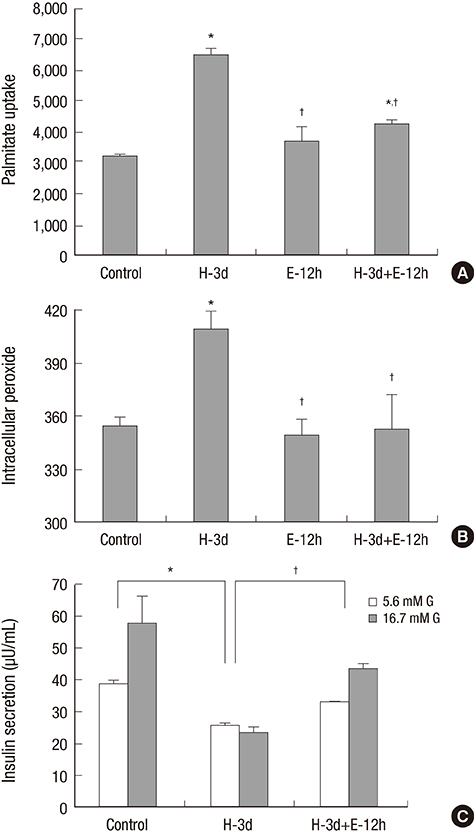
- Inhibition of CD36, a fatty acid transporter, has been reported to prevent glucotoxicity and ameliorate high glucose induced beta cell dysfunction. Ezetimibe is a selective cholesterol absorption inhibitor that blocks Niemann Pick C1-like 1 protein, but may exert its effect through suppression of CD36. We attempted to clarify the beneficial effect of ezetimibe on insulin secreting cells and to determine whether this effect is related to change of CD36 expression. mRNA expression of insulin and CD36, intracellular peroxide level and glucose stimulated insulin secretion (GSIS) under normal (5.6 mM) or high glucose (30 mM) condition in INS-1 cells and primary rat islet cells were compared. Changes of the aforementioned factors with treatment with ezetimibe (20 μM) under normal or high glucose condition were also assessed. mRNA expression of insulin was decreased with high glucose, which was reversed by ezetimibe in both INS-1 cells and primary rat islets. CD36 mRNA expression was increased with high glucose, but decreased by ezetimibe in INS-1 cells and primary rat islets. Three-day treatment with high glucose resulted in an increase in intracellular peroxide level; however, it was decreased by treatment with ezetimibe. Decrease in GSIS by three-day treatment with high glucose was reversed by ezetimibe. Palmitate uptake following exposure to high glucose conditions for three days was significantly elevated, which was reversed by ezetimibe in INS-1 cells. Ezetimibe may prevent glucotoxicity in pancreatic β-cells through a decrease in fatty acid influx via inhibition of CD36.
Keyword
MeSH Terms
-
Animals
Anticholesteremic Agents/*pharmacology
Antigens, CD36/antagonists & inhibitors/genetics/*metabolism
Cells, Cultured
Ezetimibe/*pharmacology
Flow Cytometry
Glucose/toxicity
Insulin/genetics/metabolism/secretion
Insulin-Secreting Cells/cytology/*drug effects/metabolism
Male
Palmitic Acid/metabolism
RNA, Messenger/metabolism
Rats
Rats, Sprague-Dawley
Reactive Oxygen Species/metabolism
Real-Time Polymerase Chain Reaction
Anticholesteremic Agents
Antigens, CD36
Ezetimibe
Glucose
Insulin
Palmitic Acid
RNA, Messenger
Reactive Oxygen Species
Figure
Cited by 2 articles
-
The Efficacy and Safety of Moderate-Intensity Rosuvastatin with Ezetimibe versus High-Intensity Rosuvastatin in High Atherosclerotic Cardiovascular Disease Risk Patients with Type 2 Diabetes Mellitus: A Randomized, Multicenter, Open, Parallel, Phase 4 Study
Jun Sung Moon, Il Rae Park, Sang Soo Kim, Hye Soon Kim, Nam Hoon Kim, Sin Gon Kim, Seung Hyun Ko, Ji Hyun Lee, Inkyu Lee, Bo Kyeong Lee, Kyu Chang Won
Diabetes Metab J. 2023;47(6):818-825. doi: 10.4093/dmj.2023.0171.Overcoming β-Cell Dysfunction in Type 2 Diabetes Mellitus: CD36 Inhibition and Antioxidant System
Il Rae Park, Yong Geun Chung, Kyu Chang Won
Diabetes Metab J. 2025;49(1):1-12. doi: 10.4093/dmj.2024.0796.
Reference
-
1. Poitout V, Robertson RP. Glucolipotoxicity: fuel excess and beta-cell dysfunction. Endocr Rev. 2008; 29:351–366.2. Kim JW, Yoon KH. Glucolipotoxicity in pancreatic β-cells. Diabetes Metab J. 2011; 35:444–450.3. Jacqueminet S, Briaud I, Rouault C, Reach G, Poitout V. Inhibition of insulin gene expression by long-term exposure of pancreatic beta cells to palmitate is dependent on the presence of a stimulatory glucose concentration. Metabolism. 2000; 49:532–536.4. Wallin T, Ma Z, Ogata H, Jørgensen IH, Iezzi M, Wang H, Wollheim CB, Björklund A. Facilitation of fatty acid uptake by CD36 in insulin-producing cells reduces fatty-acid-induced insulin secretion and glucose regulation of fatty acid oxidation. Biochim Biophys Acta. 2010; 1801:191–197.5. Kim YW, Moon JS, Seo YJ, Park SY, Kim JY, Yoon JS, Lee IK, Lee HW, Won KC. Inhibition of fatty acid translocase cluster determinant 36 (CD36), stimulated by hyperglycemia, prevents glucotoxicity in INS-1 cells. Biochem Biophys Res Commun. 2012; 420:462–466.6. Altmann SW, Davis HR Jr, Zhu LJ, Yao X, Hoos LM, Tetzloff G, Iyer SP, Maguire M, Golovko A, Zeng M, et al. Niemann-Pick C1 Like 1 protein is critical for intestinal cholesterol absorption. Science. 2004; 303:1201–1204.7. Kramer W, Girbig F, Corsiero D, Pfenninger A, Frick W, Jähne G, Rhein M, Wendler W, Lottspeich F, Hochleitner EO, et al. Aminopeptidase N (CD13) is a molecular target of the cholesterol absorption inhibitor ezetimibe in the enterocyte brush border membrane. J Biol Chem. 2005; 280:1306–1320.8. Mina-Osorio P, Ortega E, Aminopeptidase N. CD13) functionally interacts with FcgammaRs in human monocytes. J Leukoc Biol. 2005; 77:1008–1017.9. Orsó E, Werner T, Wolf Z, Bandulik S, Kramer W, Schmitz G. Ezetimib influences the expression of raft-associated antigens in human monocytes. Cytometry A. 2006; 69:206–208.10. Hiramitsu S, Miyagishima K, Ishii J, Matsui S, Naruse H, Shiino K, Kitagawa F, Ozaki Y. Effect of ezetimibe on lipid and glucose metabolism after a fat and glucose load. J Cardiol. 2012; 60:395–400.11. Tsunoda T, Nozue T, Yamada M, Mizuguchi I, Sasaki M, Michishita I. Effects of ezetimibe on atherogenic lipoproteins and glucose metabolism in patients with diabetes and glucose intolerance. Diabetes Res Clin Pract. 2013; 100:46–52.12. Nomura M, Ishii H, Kawakami A, Yoshida M. Inhibition of hepatic Niemann-Pick C1-like 1 improves hepatic insulin resistance. Am J Physiol Endocrinol Metab. 2009; 297:E1030–8.13. Zhong Y, Wang J, Gu P, Shao J, Lu B, Jiang S. Effect of ezetimibe on insulin secretion in db/db diabetic mice. Exp Diabetes Res. 2012; 2012:420854.14. Yang SJ, Choi JM, Kim L, Kim BJ, Sohn JH, Kim WJ, Park SE, Rhee EJ, Lee WY, Oh KW, et al. Chronic administration of ezetimibe increases active glucagon-like peptide-1 and improves glycemic control and pancreatic beta cell mass in a rat model of type 2 diabetes. Biochem Biophys Res Commun. 2011; 407:153–157.15. Won KC, Moon JS, Eun MJ, Yoon JS, Chun KA, Cho IH, Kim YW, Lee HW. A protective role for heme oxygenase-1 in INS-1 cells and rat islets that are exposed to high glucose conditions. J Korean Med Sci. 2006; 21:418–424.16. Robertson RP, Harmon J, Tran PO, Poitout V. Beta-cell glucose toxicity, lipotoxicity, and chronic oxidative stress in type 2 diabetes. Diabetes. 2004; 53:Suppl 1. S119–24.17. Boden G, Chen X, Rosner J, Barton M. Effects of a 48-h fat infusion on insulin secretion and glucose utilization. Diabetes. 1995; 44:1239–1242.18. Paolisso G, Tataranni PA, Foley JE, Bogardus C, Howard BV, Ravussin E. A high concentration of fasting plasma non-esterified fatty acids is a risk factor for the development of NIDDM. Diabetologia. 1995; 38:1213–1217.19. Leung N, Sakaue T, Carpentier A, Uffelman K, Giacca A, Lewis GF. Prolonged increase of plasma non-esterified fatty acids fully abolishes the stimulatory effect of 24 hours of moderate hyperglycaemia on insulin sensitivity and pancreatic beta-cell function in obese men. Diabetologia. 2004; 47:204–213.20. Piro S, Anello M, Di Pietro C, Lizzio MN, Patanè G, Rabuazzo AM, Vigneri R, Purrello M, Purrello F. Chronic exposure to free fatty acids or high glucose induces apoptosis in rat pancreatic islets: possible role of oxidative stress. Metabolism. 2002; 51:1340–1347.21. Giacca A, Xiao C, Oprescu AI, Carpentier AC, Lewis GF. Lipid-induced pancreatic β-cell dysfunction: focus on in vivo studies. Am J Physiol Endocrinol Metab. 2011; 300:E255–62.22. Lupi R, Dotta F, Marselli L, Del Guerra S, Masini M, Santangelo C, Patané G, Boggi U, Piro S, Anello M, et al. Prolonged exposure to free fatty acids has cytostatic and pro-apoptotic effects on human pancreatic islets: evidence that β-cell death is caspase mediated, partially dependent on ceramide pathway, and Bcl-2 regulated. Diabetes. 2002; 51:1437–1442.23. Eitel K, Staiger H, Rieger J, Mischak H, Brandhorst H, Brendel MD, Bretzel RG, Häring HU, Kellerer M. Protein kinase C delta activation and translocation to the nucleus are required for fatty acid-induced apoptosis of insulin-secreting cells. Diabetes. 2003; 52:991–997.24. Busch AK, Cordery D, Denyer GS, Biden TJ. Expression profiling of palmitate- and oleate-regulated genes provides novel insights into the effects of chronic lipid exposure on pancreatic beta-cell function. Diabetes. 2002; 51:977–987.25. Kharroubi I, Ladrière L, Cardozo AK, Dogusan Z, Cnop M, Eizirik DL. Free fatty acids and cytokines induce pancreatic β-cell apoptosis by different mechanisms: role of nuclear factor-kappaB and endoplasmic reticulum stress. Endocrinology. 2004; 145:5087–5096.26. Hajri T, Abumrad NA. Fatty acid transport across membranes: relevance to nutrition and metabolic pathology. Annu Rev Nutr. 2002; 22:383–415.27. Griffin E, Re A, Hamel N, Fu C, Bush H, McCaffrey T, Asch AS. A link between diabetes and atherosclerosis: glucose regulates expression of CD36 at the level of translation. Nat Med. 2001; 7:840–846.28. Dalgaard LT, Thams P, Gaarn LW, Jensen J, Lee YC, Nielsen JH. Suppression of FAT/CD36 mRNA by human growth hormone in pancreatic β-cells. Biochem Biophys Res Commun. 2011; 410:345–350.29. Noushmehr H, D’Amico E, Farilla L, Hui H, Wawrowsky KA, Mlynarski W, Doria A, Abumrad NA, Perfetti R. Fatty acid translocase (FAT/CD36) is localized on insulin-containing granules in human pancreatic beta-cells and mediates fatty acid effects on insulin secretion. Diabetes. 2005; 54:472–481.30. Kikuchi K, Nezu U, Inazumi K, Miyazaki T, Ono K, Orime K, Shirakawa J, Sato K, Koike H, Wakasugi T, et al. Double-blind randomized clinical trial of the effects of ezetimibe on postprandial hyperlipidaemia and hyperglycaemia. J Atheroscler Thromb. 2012; 19:1093–1101.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Role of CD36 in Type 2 Diabetes Mellitus: β-Cell Dysfunction and Beyond
- Overcoming β-Cell Dysfunction in Type 2 Diabetes Mellitus: CD36 Inhibition and Antioxidant System
- Aloe-Emodin Protects RIN-5F (Pancreatic beta-cell) Cell from Glucotoxicity via Regulation of Pro-Inflammatory Cytokine and Downregulation of Bax and Caspase 3
- Can Tea Extracts Exert a Protective Effect Against Diabetes by Reducing Oxidative Stress and Decreasing Glucotoxicity in Pancreatic beta-Cells?
- Padina arborescens extract protects high glucose-induced apoptosis in pancreatic beta cells by reducing oxidative stress