Clin Exp Otorhinolaryngol.
2016 Dec;9(4):332-338. 10.21053/ceo.2015.01599.
Effect of Long-Term Antiorthostatic Suspension in a Murine Model of Acute Lung Injury
- Affiliations
-
- 1Department of Otorhinolaryngology-Head and Neck Surgery, Inha University School of Medicine, Incheon, Korea. inhaorl@inha.ac.kr
- KMID: 2360764
- DOI: http://doi.org/10.21053/ceo.2015.01599
Abstract
OBJECTIVES
Antiorthostatic suspension (AOS) is ground-based model of simulated microgravity. There is still no study about the effect of long-term microgravity on the clinical course of acute lung injury. We evaluated the effect of simulated microgravity using AOS in a murine model of acute lung injury by lipopolysaccharide (LPS).
METHODS
Thirty BALB/c mice were used. During 4 weeks, mice were equally allocated to control (free movement), restraint (tail suspended, but hindlimbs not unloaded), and AOS group (hindlimb unloaded). After then, mice got intranasal challenge with LPS (20 mg/kg, 50 μL). We measured: weight gain before and after AOS, the number of inflammatory cells and titers of cytokines (interleukin [IL]-1β, IL-6, IL-10, tumor necrosis factor-α, and interferon-γ) in bronchoalveolar lavage (BAL) fluid, titer of myeloperoxidase (MPO) in serum and lung homogenate, and histopathologic examination of lung tissue.
RESULTS
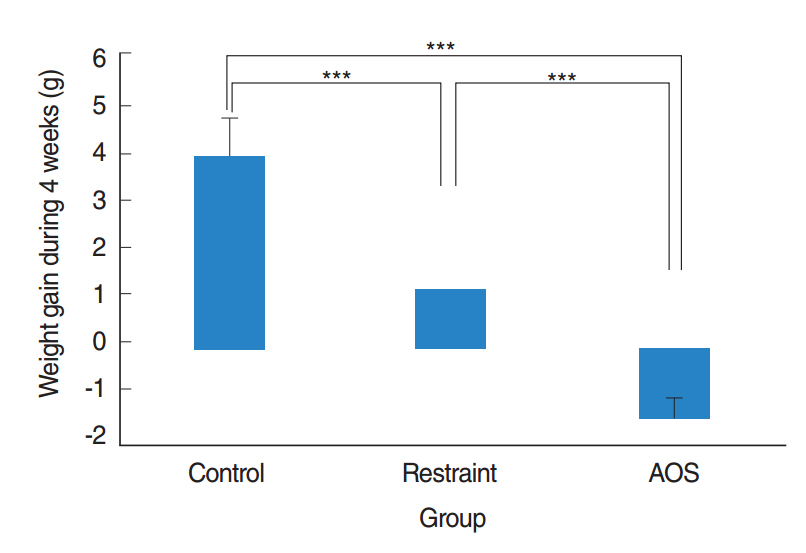
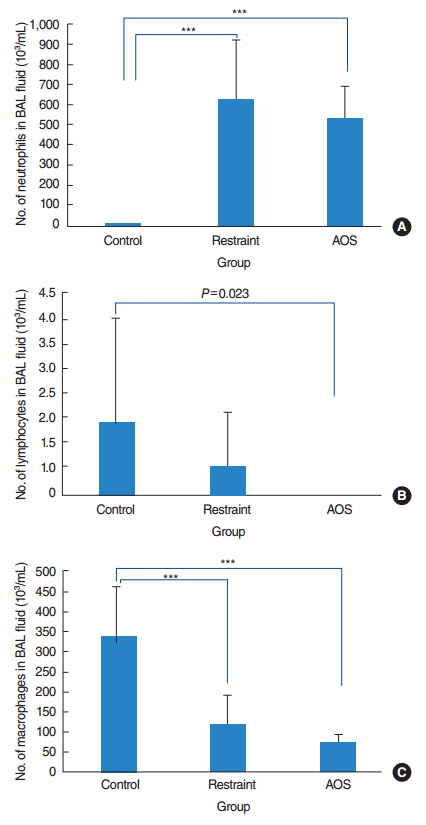
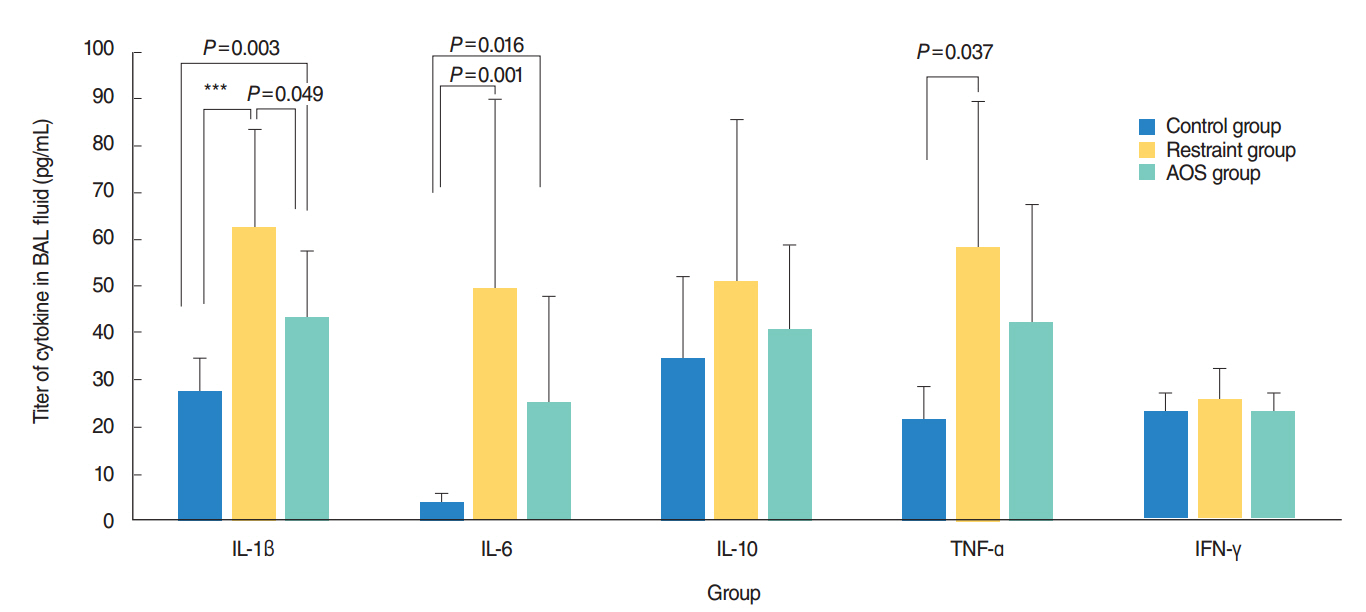
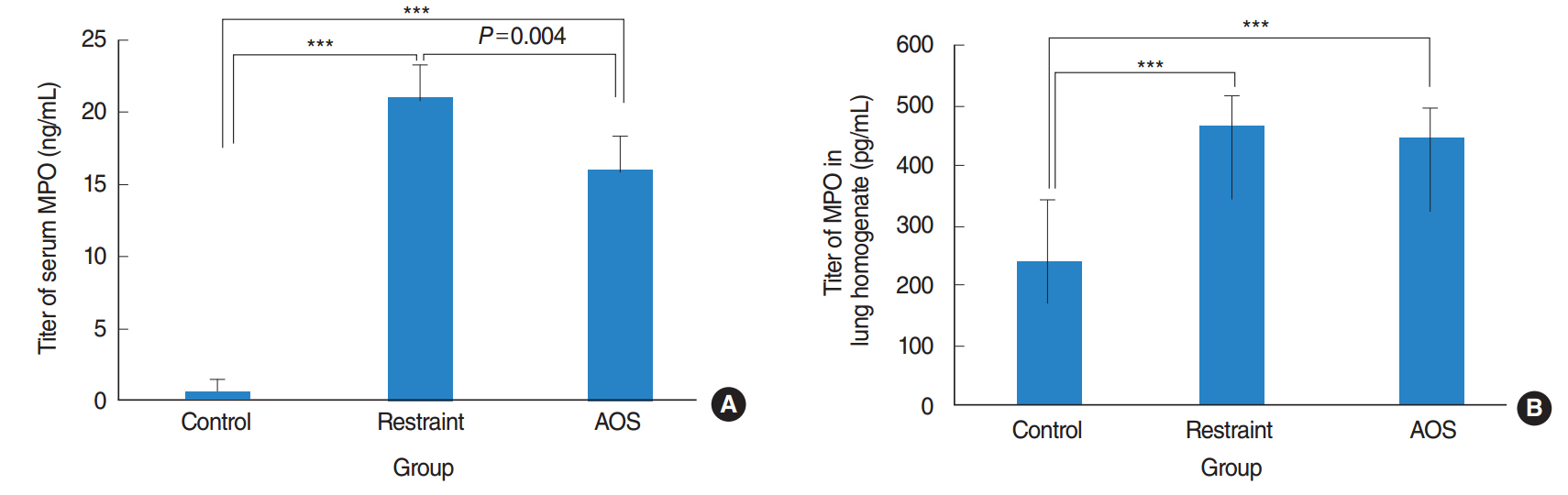
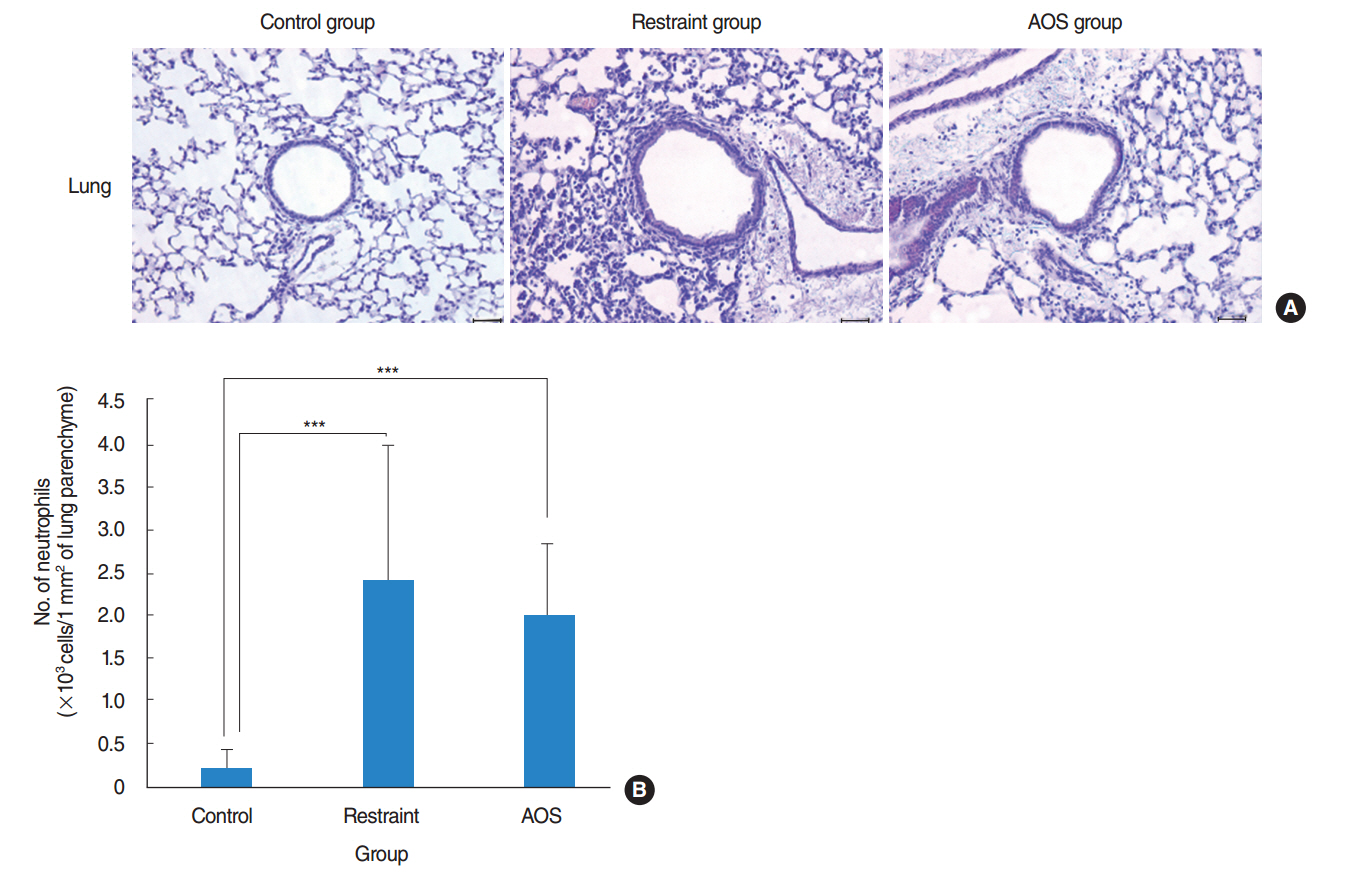
AOS group had significant weight loss compared to control and restraint group (P<0.001). AOS group also showed significantly decreased lymphocytes (P=0.023) compared to control group. In AOS group, titer for IL-1β in BAL fluid was significantly lower than restraint group (P=0.049). Titer for serum MPO was significantly decreased in AOS group compared to restraint group (P=0.004). However, there was no significant difference of MPO titers in lung tissue between groups. Histopathologic examination of lung tissue revealed no significant difference in the degree of pulmonary infiltration between restraint and AOS group.
CONCLUSION
In spite of modest anti-inflammatory effect, prolonged AOS caused no significant change in LPS-induced pulmonary inflammation.
MeSH Terms
Figure
Reference
-
1. Gaignier F, Schenten V, De Carvalho Bittencourt M, Gauquelin-Koch G, Frippiat JP, Legrand-Frossi C. Three weeks of murine hindlimb unloading induces shifts from B to T and from th to tc splenic lymphocytes in absence of stress and differentially reduces cell-specific mitogenic responses. PLoS One. 2014; Mar. 9(3):e92664.
Article2. Felix K, Wise K, Manna S, Yamauchi K, Wilson BL, Thomas RL, et al. Altered cytokine expression in tissues of mice subjected to simulated microgravity. Mol Cell Biochem. 2004; Nov. 266(1-2):79–85.
Article3. Morey-Holton E, Globus RK, Kaplansky A, Durnova G. The hindlimb unloading rat model: literature overview, technique update and comparison with space flight data. Adv Space Biol Med. 2005; Oct. 10:7–40.
Article4. Sonnenfeld G, Morey ER, Williams JA, Mandel AD. Effect of a simulated weightlessness model on the production of rat interferon. J Interferon Res. 2009; May. 2(4):467–70.
Article5. Bigatello LM, Zapol WM. New approaches to acute lung injury. Br J Anaesth. 1996; Jul. 77(1):99–109.
Article6. Fang L, Gao Y, Liu F, Hou R, Cai RL, Qi Y. Shuang-huang-lian attenuates lipopolysaccharide-induced acute lung injury in mice involving anti-inflammatory and antioxidative activities. Evid Based Complement Alternat Med. 2015; 2015:283939.
Article7. Lew PS, Wong D, Yamaguchi T, Leckstrom A, Schwartz J, Dodd JG, et al. Tail suspension increases energy expenditure independently of the melanocortin system in mice. Can J Physiol Pharmacol. 2009; Oct. 87(10):839–49.8. Amblard D, Lafage-Proust MH, Laib A, Thomas T, Ruegsegger P, Alexandre C, et al. Tail suspension induces bone loss in skeletally mature mice in the C57BL/6J strain but not in the C3H/HeJ strain. J Bone Miner Res. 2003; Mar. 18(3):561–9.
Article9. Da Silva MS, Zimmerman PM, Meguid MM, Nandi J, Ohinata K, Xu Y, et al. Anorexia in space and possible etiologies: an overview. Nutrition. 2002; Oct. 18(10):805–13.10. Ferrando AA, Paddon-Jones D, Wolfe RR. Alterations in protein metabolism during space flight and inactivity. Nutrition. 2002; Oct. 18(10):837–41.
Article11. LeBlanc A, Rowe R, Schneider V, Evans H, Hedrick T. Regional muscle loss after short duration spaceflight. Aviat Space Environ Med. 1995; Dec. 66(12):1151–4.12. Lane HW, Feeback DL. Water and energy dietary requirements and endocrinology of human space flight. Nutrition. 2002; Oct. 18(10):820–8.
Article13. Rambaut PC, Leach CS, Leonard JI. Observations in energy balance in man during spaceflight. Am J Physiol. 1977; Nov. 233(5):R208–12.
Article14. Stein TP, Schluter MD. Excretion of IL-6 by astronauts during space-flight. Am J Physiol. 1994; Mar. 266(3 Pt 1):E448–52.
Article15. Durnova GN, Kaplansky AS, Portugalov VV. Effect of a 22-day space flight on the lymphoid organs of rats. Aviat Space Environ Med. 1976; Jun. 47(6):588–91.16. Taylor GR, Neale LS, Dardano JR. Immunological analyses of U.S. Space Shuttle crewmembers. Aviat Space Environ Med. 1986; Mar. 57(3):213–7.17. Sonnenfeld G, Mandel AD, Konstantinova IV, Taylor GR, Berry WD, Wellhausen SR, et al. Effects of spaceflight on levels and activity of immune cells. Aviat Space Environ Med. 1990; Jul. 61(7):648–53.18. Murdaca G, Setti M, Brenci S, Fenoglio D, Lantieri P, Indiveri F, et al. Modifications of immunological and neuro-endocrine parameters induced by antiorthostatic bed-rest in human healthy volunteers. Minerva Med. 2003; Dec. 94(6):363–78.19. Miller ES, Bates RA, Koebel DA, Sonnenfeld G. Antiorthostatic suspension stimulates profiles of macrophage activation in mice. Neuroimmunomodulation. 1999; May-Jun. 6(3):160–7.
Article20. Limouse M, Manie S, Konstantinova I, Ferrua B, Schaffar L. Inhibition of phorbol ester-induced cell activation in microgravity. Exp Cell Res. 1991; Nov. 197(1):82–6.
Article21. Schmitt DA, Hatton JP, Emond C, Chaput D, Paris H, Levade T, et al. The distribution of protein kinase C in human leukocytes is altered in microgravity. FASEB J. 1996; Dec. 10(14):1627–34.
Article22. Wang C, Luo H, Zhu L, Yang F, Chu Z, Tian H, et al. Microgravity inhibition of lipopolysaccharide-induced tumor necrosis factor-α expression in macrophage cells. Inflamm Res. 2014; Jan. 63(1):91–8.
Article23. Hsieh CL, Chao PD, Fang SH. Morin sulphates/glucuronides enhance macrophage function in microgravity culture system. Eur J Clin Invest. 2005; Sep. 35(9):591–6.
Article24. Silva MT. Neutrophils and macrophages work in concert as inducers and effectors of adaptive immunity against extracellular and intracellular microbial pathogens. J Leukoc Biol. 2010; May. 87(5):805–13.
Article25. Kwon T, Sparks JA, Nakashima J, Allen SN, Tang Y, Blancaflor EB. Transcriptional response of Arabidopsis seedlings during spaceflight reveals peroxidase and cell wall remodeling genes associated with root hair development. Am J Bot. 2015; Jan. 102(1):21–35.
Article26. Guha M, Mackman N. LPS induction of gene expression in human monocytes. Cell Signal. 2001; Feb. 13(2):85–94.
Article27. Nikaido H. Molecular basis of bacterial outer membrane permeability revisited. Microbiol Mol Biol Rev. 2003; Dec. 67(4):593–656.
Article28. Polumuri SK, Jayakar GG, Shirey KA, Roberts ZJ, Perkins DJ, Pitha PM, et al. Transcriptional regulation of murine IL-33 by TLR and non-TLR agonists. J Immunol. 2012; Jul. 189(1):50–60.
Article29. Belay T, Aviles H, Vance M, Fountain K, Sonnenfeld G. Effects of the hindlimb-unloading model of spaceflight conditions on resistance of mice to infection with Klebsiella pneumoniae. J Allergy Clin Immunol. 2002; Aug. 110(2):262–8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Establishment of a murine model based on chronic exposure to domestic house dust mites in Korea
- Lung repair after acute lung injury
- Nonventilatory medical treatments for acute respiratory distress syndrome and acute lung injury
- Long-term Results of the Gittes Procedure for Treatment of Stress Urinary Incontinence
- An animal model of severe acute respiratory distress syndrome for translational research