J Korean Med Sci.
2016 Jan;31(1):18-24. 10.3346/jkms.2016.31.1.18.
Allergenic Characterization of 27-kDa Glycoprotein, a Novel Heat Stable Allergen, from the Pupa of Silkworm, Bombyx mori
- Affiliations
-
- 1Department of Internal Medicine, Institute of Allergy, Yonsei University College of Medicine, Seoul, Korea. parkjw@yuhs.ac
- 2Brain Korea 21 PLUS Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2359987
- DOI: http://doi.org/10.3346/jkms.2016.31.1.18
Abstract
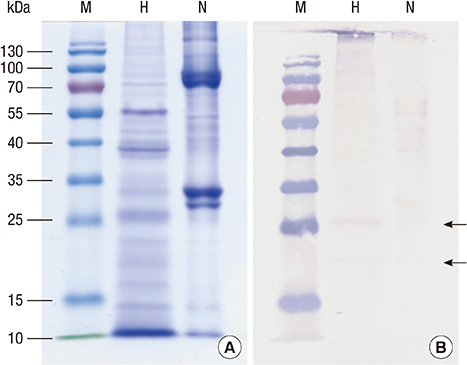
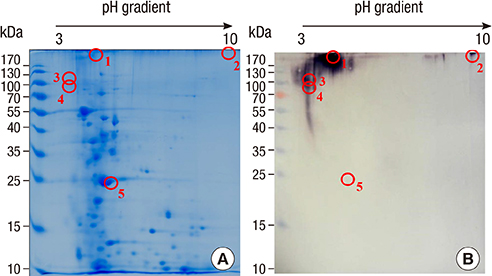
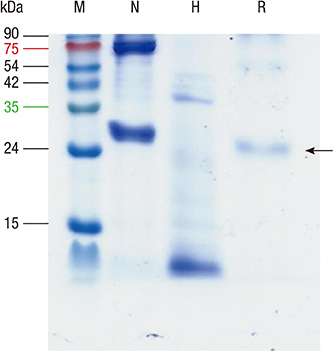
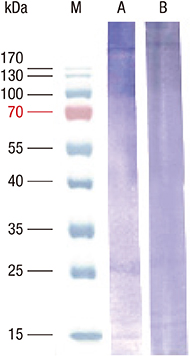
- Boiled silkworm pupa is a traditional food in Asia, and patients with silkworm pupa food allergy are common in these regions. Still now only one allergen from silkworm, arginine kinase, has been identified. The purpose of this study was to identify novel food allergens in silkworm pupa by analyzing a protein extract after heat treatment. Heat treated extracts were examined by proteomic analysis. A 27-kDa glycoprotein was identified, expressed in Escherichia coli, and purified. IgE reactivity of the recombinant protein was investigated by ELISA. High molecular weight proteins (above 100 kDa) elicited increased IgE binding after heat treatment compared to that before heat treatment. The molecular identities of these proteins, however, could not be determined. IgE reactivity toward a 27-kDa glycoprotein was also increased after heating the protein extract. The recombinant protein was recognized by IgE antibodies from allergic subjects (33.3%). Glycation or aggregation of protein by heating may create new IgE binding epitopes. Heat stable allergens are shown to be important in silkworm allergy. Sensitization to the 27-kDa glycoprotein from silkworm may contribute to elevation of IgE to silkworm.
Keyword
MeSH Terms
-
Adolescent
Adult
Allergens/*chemistry/*immunology
Amino Acid Sequence
Animals
Bombyx/*chemistry/genetics/growth & development/*immunology
Epitopes/immunology
Female
Food Hypersensitivity/etiology
Glycoproteins/*chemistry/genetics/*immunology
Hot Temperature
Humans
Immunoglobulin E/immunology
Male
Molecular Sequence Data
Molecular Weight
Proteomics
Pupa/chemistry/immunology
Recombinant Proteins/biosynthesis/chemistry/immunology
Sequence Alignment
Allergens
Epitopes
Glycoproteins
Immunoglobulin E
Recombinant Proteins
Figure
Cited by 1 articles
-
Physical and biochemical characteristics of allergens
Kyoung Yong Jeong
Allergy Asthma Respir Dis. 2016;4(3):157-166. doi: 10.4168/aard.2016.4.3.157.
Reference
-
1. Harindranath N, Prakash O, Subba Rao PV. Prevalence of occupational asthma in silk filatures. Ann Allergy. 1985; 55:511–515.2. Johansson SG, Wüthrich B, Zortea-Caflisch C. Nightly asthma caused by allergens in silk-filled bed quilts: clinical and immunologic studies. J Allergy Clin Immunol. 1985; 75:452–459.3. Celedón JC, Palmer LJ, Xu X, Wang B, Fang Z, Weiss ST. Sensitization to silk and childhood asthma in rural China. Pediatrics. 2001; 107:E80.4. Wen CM, Ye ST, Zhou LX, Yu Y. Silk-induced asthma in children: a report of 64 cases. Ann Allergy. 1990; 65:375–378.5. Suzuki M, Itoh H, Sugiyama K, Takagi I, Nishimura J, Kato K, Mamiya S, Baba S, Ohya Y, Itoh H, et al. Causative allergens of allergic rhinitis in Japan with special reference to silkworm moth allergen. Allergy. 1995; 50:23–27.6. Dewair M, Baur X, Ziegler K. Use of immunoblot technique for detection of human IgE and IgG antibodies to individual silk proteins. J Allergy Clin Immunol. 1985; 76:537–542.7. Celedón JC, Palmer LJ, Weiss ST, Wang B, Fang Z, Xu X. Asthma, rhinitis, and skin test reactivity to aeroallergens in families of asthmatic subjects in Anqing, China. Am J Respir Crit Care Med. 2001; 163:1108–1112.8. Kim SA, Kim KM, Oh BJ. Current status and perspective of the insect industry in Korea. Entomol Res. 2008; 38:S79–S85.9. MacEvilly C. Bugs in the system. Nutr Bull. 2000; 25:267–268.10. Yang Y, Tang L, Tong L, Liu H. Silkworms culture as a source of protein for humans in space. Adv Space Res. 2009; 43:1236–1242.11. Ji KM, Zhan ZK, Chen JJ, Liu ZG. Anaphylactic shock caused by silkworm pupa consumption in China. Allergy. 2008; 63:1407–1408.12. Kim SH, Kang HR, Kim KM, Kim TB, Kim SS, Chang YS, Kim CW, Bahn JW, Kim YK, Cho SH, et al. The sensitization rates of food allergens in a Korean population: a multi-center study. J Asthma Allergy Clin Immunol. 2003; 23:502–514.13. van der Poel L, Chen J. Food allergy epidemic - is it only a Western phenomenon? Curr Allergy Clin Immunol. 2009; 22:121–126.14. Liu Z, Xia L, Wu Y, Xia Q, Chen J, Roux KH. Identification and characterization of an arginine kinase as a major allergen from silkworm (Bombyx mori) larvae. Int Arch Allergy Immunol. 2009; 150:8–14.15. Binder M, Mahler V, Hayek B, Sperr WR, Schöller M, Prozell S, Wiedermann G, Valent P, Valenta R, Duchêne M. Molecular and immunological characterization of arginine kinase from the Indianmeal moth, Plodia interpunctella, a novel cross-reactive invertebrate pan-allergen. J Immunol. 2001; 167:5470–5477.16. Bannon GA. What makes a food protein an allergen? Curr Allergy Asthma Rep. 2004; 4:43–46.17. Samaraweera P, Law JH. Isolation, cloning and deduced amino acid sequence of a novel glycoprotein from the haemolymph of the hawkmoth Manduca sexta. Insect Mol Biol. 1995; 4:7–13.18. Madero MF, Enríquez-Matas A, Fernández-Nieto M, Sastre B, Del Pozo V, Pastor C, Quirce S, Sastre J. Characterization of allergens from the fish bait Galleria mellonella. J Allergy Clin Immunol. 2007; 119:1021–1022.19. Hales BJ, Laing IA, Pearce LJ, Hazell LA, Mills KL, Chua KY, Thornton RB, Richmond P, Musk AW, James AL, et al. Distinctive immunoglobulin E anti-house dust allergen-binding specificities in a tropical Australian Aboriginal community. Clin Exp Allergy. 2007; 37:1357–1363.20. Jeong KY, Lee JY, Son M, Yi MH, Yong TS, Shin JU, Lee KH, Kim YJ, Park KH, Park HJ, et al. Profiles of IgE sensitization to Der f 1, Der f 2, Der f 6, Der f 8, Der f 10, and Der f 20 in Korean house dust mite allergy patients. Allergy Asthma Immunol Res. 2015; 7:483–488.21. Poulsen MW, Hedegaard RV, Andersen JM, de Courten B, Bügel S, Nielsen J, Skibsted LH, Dragsted LO. Advanced glycation endproducts in food and their effects on health. Food Chem Toxicol. 2013; 60:10–37.22. Jiménez-Saiz R, Benedé S, Molina E, López-Expósito I. Effect of processing technologies on the allergenicity of food products. Crit Rev Food Sci Nutr. 2015; 55:1902–1917.23. Choi GS, Shin YS, Kim JE, Ye YM, Park HS. Five cases of food allergy to vegetable worm (Cordyceps sinensis) showing cross-reactivity with silkworm pupae. Allergy. 2010; 65:1196–1197.24. Liu ZG, Zhang J, Lin G. Analysis, purification and identification of the 30 kD specific allergen of Bombyx mori using mass spectrometry. Acta Entomol Sin. 2007; 50:101–105.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Mycoparasitic Ascomycete Syspastospora parasitica on the Entomopathogenic Fungus Paecilomyces tenuipes Growing in Bombyx mori
- Fruit Body Formation on Silkworm by Cordyceps militaris
- Breeding of a Silkworm Variety for Synnemata Production of Isaria tenuipes
- Red ginseng extract increases the functional components of silkworm Bombyx mori L. used as an anti-diabetes medicinal resource
- Synnemata Production Using Silkworm Variety, Female Yangwonjam by Isaria tenuipes