World J Mens Health.
2013 Dec;31(3):247-253.
Expression of Heat Shock Protein 27 in Prostate Cancer Cell Lines According to the Extent of Malignancy and Doxazosin Treatment
- Affiliations
-
- 1Department of Urology, Hanyang University College of Medicine, Seoul, Korea. swleepark@hanyang.ac.kr
- 2Department of Urology, Hanyang University Guri Hospital, Guri, Korea.
Abstract
- PURPOSE
Heat shock protein 27 (HSP27) is known as the material that plays a role in apoptosis control in tumor and cell protection including the immune response, drug tolerance, and so on. In this study, HSP27 expression according to the prostate cancer malignancy level was evaluated, and HSP27 expression was also analyzed after inducing apoptosis by doxazosin treatment of the prostate cancer cell lines.
MATERIALS AND METHODS
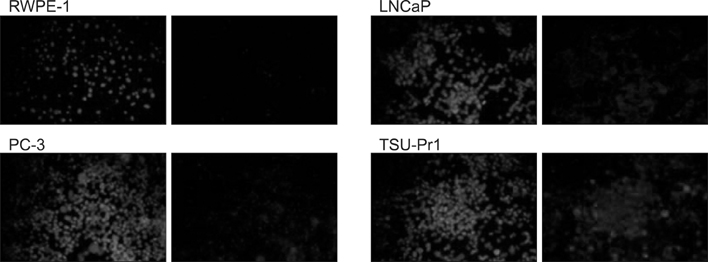
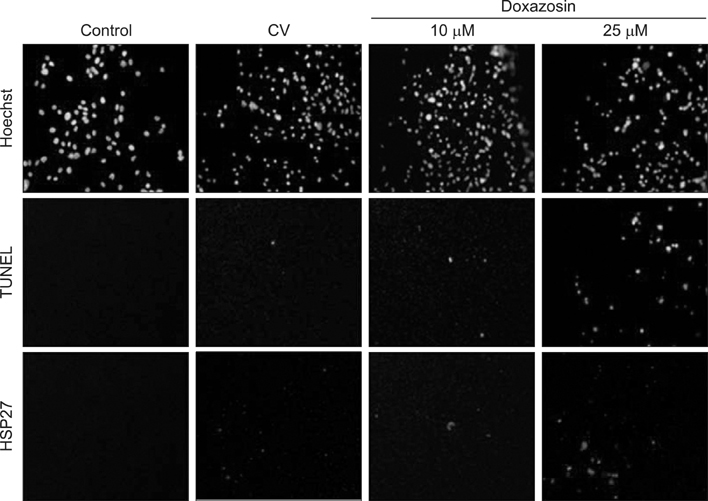
Reverse transcription polymerase chain reaction (RT-PCR) and immunofluorescence staining of the HSP27 was implemented by the culture of RWPE-1, LNCaP, androgen-independent human prostate cancer cells (PC-3), and TSU-Pr1 cell lines. Analysis was separately conducted in the control group, control vector group treated by dimethyl sulfoxide, and groups treated with 10 microM or 25 microM doxazosin. The expression of HSP27 in RT-PCR and immunofluorescence staining was observed and evaluated after conversion to numerical values.
RESULTS
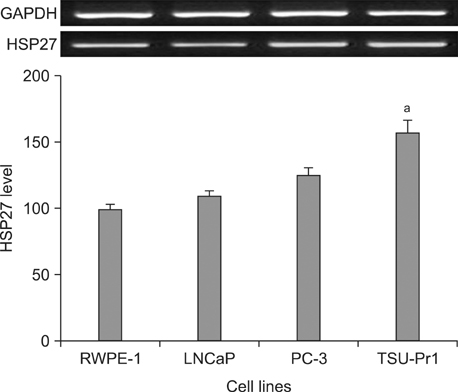
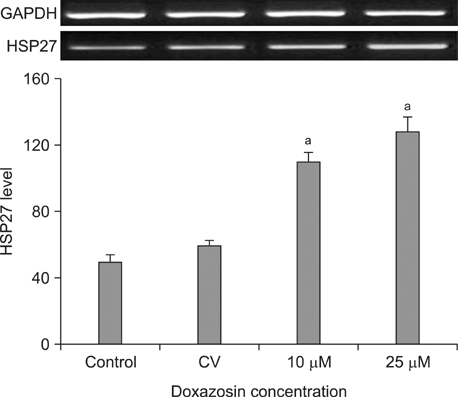
In the RT-PCR results, depending on the cell type, LNCaP, TSU-Pr1 showed the highest HSP27 expression followed by PC-3, LNCaP and RWPE-1 in sequence. After doxazosin treatment, the expression detected by RT-PCR was stronger at a 25-microM doxazosin concentration compared to that at a 10-microM concentration, and the result was similar by immunofluorescence staining.
CONCLUSIONS
HSP27 expression increased depending on the prostate cancer cell line. This meant that HSP27 expression was related to the prostate cancer malignancy level. Additionally, the higher the treatment concentration in PC-3 was, the higher the HSP27 expression was. This result showed that doxazosin induced apoptosis of prostate cancer.
Keyword
MeSH Terms
-
Apoptosis
Cell Line*
Control Groups
Cytoprotection
Dimethyl Sulfoxide
Doxazosin*
Drug Tolerance
Fluorescent Antibody Technique
Heat-Shock Proteins*
Hot Temperature*
HSP27 Heat-Shock Proteins*
Humans
Polymerase Chain Reaction
Prostatic Neoplasms
Reverse Transcription
Dimethyl Sulfoxide
Doxazosin
HSP27 Heat-Shock Proteins
Heat-Shock Proteins
Figure
Reference
-
1. Kim JM, Kim HM, Jung BY, Park EC, Cho WH, Lee SG. The association between cancer incidence and family income: analysis of Korean National Health Insurance cancer registration data. Asian Pac J Cancer Prev. 2012; 13:1371–1376.
Article2. Rocchi P, Beraldi E, Ettinger S, Fazli L, Vessella RL, Nelson C, et al. Increased Hsp27 after androgen ablation facilitates androgen-independent progression in prostate cancer via signal transducers and activators of transcription 3-mediated suppression of apoptosis. Cancer Res. 2005; 65:11083–11093.
Article3. Hessenkemper W, Baniahmad A. Targeting heat shock proteins in prostate cancer. Curr Med Chem. 2013; 20:2731–2740.
Article4. Atkins D, Lichtenfels R, Seliger B. Heat shock proteins in renal cell carcinomas. Contrib Nephrol. 2005; 148:35–56.
Article5. Rocchi P, So A, Kojima S, Signaevsky M, Beraldi E, Fazli L, et al. Heat shock protein 27 increases after androgen ablation and plays a cytoprotective role in hormone-refractory prostate cancer. Cancer Res. 2004; 64:6595–6602.
Article6. Youm YH, Kim S, Bahk YY, Yoo TK. Proteomic analysis of androgen-independent growth in low and high passage human LNCaP prostatic adenocarcinoma cells. BMB Rep. 2008; 41:722–727.
Article7. Kyprianou N, Benning CM. Suppression of human prostate cancer cell growth by alpha1-adrenoceptor antagonists doxazosin and terazosin via induction of apoptosis. Cancer Res. 2000; 60:4550–4555.8. Giardinà D, Martarelli D, Sagratini G, Angeli P, Ballinari D, Gulini U, et al. Doxazosin-related alpha1-adrenoceptor antagonists with prostate antitumor activity. J Med Chem. 2009; 52:4951–4954.9. Garrido C, Gurbuxani S, Ravagnan L, Kroemer G. Heat shock proteins: endogenous modulators of apoptotic cell death. Biochem Biophys Res Commun. 2001; 286:433–442.
Article10. Sarto C, Valsecchi C, Magni F, Tremolada L, Arizzi C, Cordani N, et al. Expression of heat shock protein 27 in human renal cell carcinoma. Proteomics. 2004; 4:2252–2260.
Article11. Concannon CG, Orrenius S, Samali A. Hsp27 inhibits cytochrome c-mediated caspase activation by sequestering both pro-caspase-3 and cytochrome c. Gene Expr. 2001; 9:195–201.
Article12. Erkizan O, Kirkali G, Yörükoğlu K, Kirkali Z. Significance of heat shock protein-27 expression in patients with renal cell carcinoma. Urology. 2004; 64:474–478.
Article13. Miyake H, Muramaki M, Kurahashi T, Takenaka A, Fujisawa M. Expression of potential molecular markers in prostate cancer: correlation with clinicopathological outcomes in patients undergoing radical prostatectomy. Urol Oncol. 2010; 28:145–151.
Article14. Teimourian S, Jalal R, Sohrabpour M, Goliaei B. Down-regulation of Hsp27 radiosensitizes human prostate cancer cells. Int J Urol. 2006; 13:1221–1225.
Article15. Dowling AJ, Tannock IF. Systemic treatment for prostate cancer. Cancer Treat Rev. 1998; 24:283–301.
Article16. Denmeade SR, Lin XS, Isaacs JT. Role of programmed (apoptotic) cell death during the progression and therapy for prostate cancer. Prostate. 1996; 28:251–265.
Article17. Landry J, Chrétien P, Laszlo A, Lambert H. Phosphorylation of HSP27 during development and decay of thermotolerance in Chinese hamster cells. J Cell Physiol. 1991; 147:93–101.
Article18. Huot J, Lambert H, Lavoie JN, Guimond A, Houle F, Landry J. Characterization of 45-kDa/54-kDa HSP27 kinase, a stress-sensitive kinase which may activate the phosphorylation-dependent protective function of mammalian 27-kDa heat-shock protein HSP27. Eur J Biochem. 1995; 227:416–427.
Article19. Mehlen P, Arrigo AP. The serum-induced phosphorylation of mammalian hsp27 correlates with changes in its intracellular localization and levels of oligomerization. Eur J Biochem. 1994; 221:327–334.
Article20. Thomas SA, Brown IL, Hollins GW, Hocken A, Kirk D, King RJ, et al. Detection and distribution of heat shock proteins 27 and 90 in human benign and malignant prostatic tissue. Br J Urol. 1996; 77:367–372.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Silencing of Heat Shock Protein 27 Expression Accelerates Doxazosin-induced Apoptosis in Prostate Cancer Cell Line PC-3
- The Protective Effect of Induced Heat Shock Protein in Human Corneal Epithelial Cells
- Environmental factors regulating the expression of Porphyromonas gingivalis heat shock protein
- The Expression of Heat Shock Protein 60 kDa in Tissues and Cell Lines of Breast Cancer
- Expression of Heat Shock Protein 27 and Apoptosis in Renal Cell Carcinomas