Nutr Res Pract.
2015 Dec;9(6):579-585. 10.4162/nrp.2015.9.6.579.
Sonchus asper extract inhibits LPS-induced oxidative stress and pro-inflammatory cytokine production in RAW264.7 macrophages
- Affiliations
-
- 1College of Life Science, Henan Normal University, Xinxiang, Henan Province 453003, China.
- 2Henan Institute of Science and Technology, Henan Province, Xinxiang 453003, China.
- 3Department of Medical Biotechnology, College of Biomedical Science, Kangwon National University, 1 Gangwondaehak-gil, Chuncheon, Gangwon 24341, Korea. mhwang@kangwon.ac.kr
- KMID: 2313880
- DOI: http://doi.org/10.4162/nrp.2015.9.6.579
Abstract
- BACKGROUND/OBJECTIVES
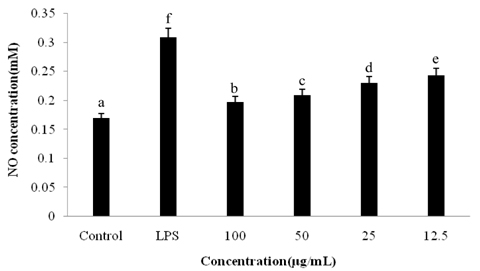
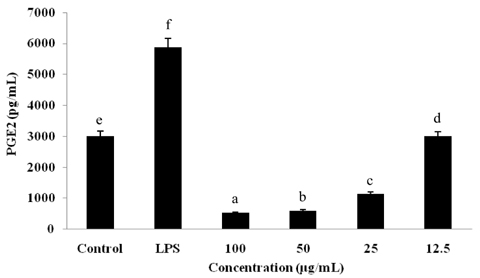
Sonchus asper is used extensively as an herbal anti-inflammatory for treatment of bronchitis, asthma, wounds, burns, and cough; however, further investigation is needed in order to understand the underlying mechanism. To determine its mechanism of action, we examined the effects of an ethyl acetate fraction (EAF) of S. asper on nitric oxide (NO) production and prostaglandin-E2 levels in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages.
MATERIALS/METHODS
An in vitro culture of RAW264.7 macrophages was treated with LPS to induce inflammation.
RESULTS
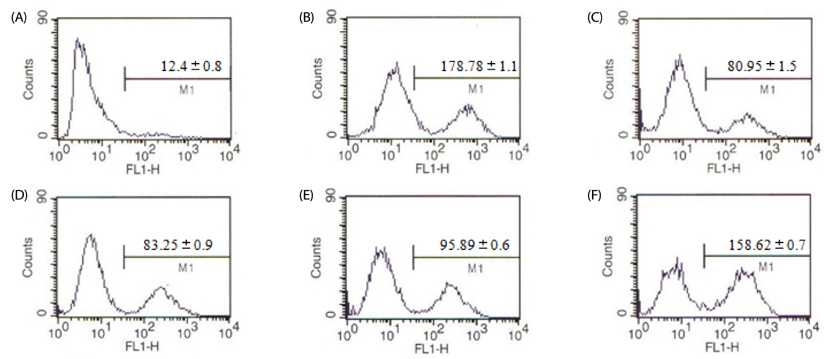
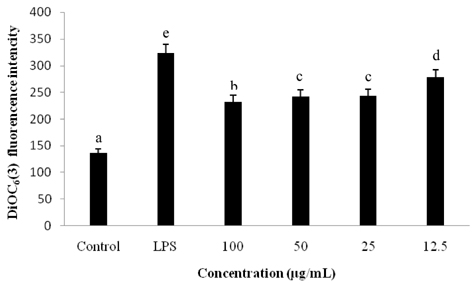
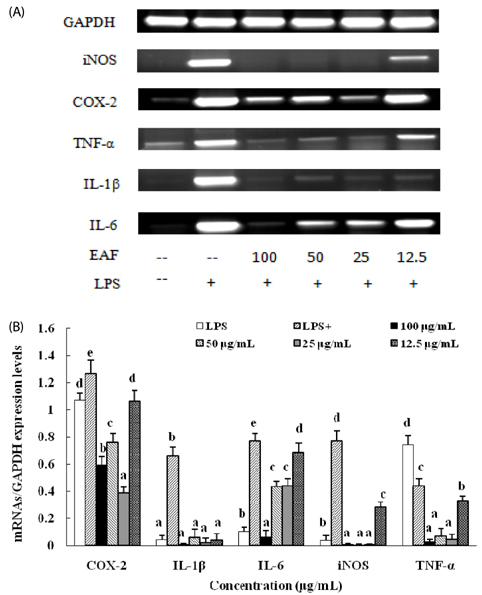
Treatment with EAF resulted in significant suppression of oxidative stress in RAW264.7 macrophages as demonstrated by increased endogenous superoxide dismutase (SOD) activity and intracellular glutathione levels, decreased generation of reactive oxygen species and lipid peroxidation, and restoration of the mitochondrial membrane potential. To confirm its anti-inflammatory effects, analysis of expression of inducible NO synthase, cyclooxygenase-2, tumor necrosis factor-alpha, and the anti-inflammatory cytokines IL-1beta and IL-6 was performed using semi-quantitative RT-PCR. EAF treatment resulted in significantly reduced dose-dependent expression of all of these factors, and enhanced expression of the antioxidants MnSOD and heme oxygenase-1. In addition, HPLC fingerprint results suggest that rutin, caffeic acid, and quercetin may be the active ingredients in EAF.
CONCLUSIONS
Taken together, findings of this study imply that the anti-inflammatory effect of EAF on LPS-stimulated RAW264.7 cells is mediated by suppression of oxidative stress.
MeSH Terms
-
Antioxidants
Asthma
Bronchitis
Burns
Chromatography, High Pressure Liquid
Cough
Cyclooxygenase 2
Cytokines
Dermatoglyphics
Glutathione
Heme Oxygenase-1
Inflammation
Interleukin-6
Lipid Peroxidation
Macrophages*
Membrane Potential, Mitochondrial
Nitric Oxide
Nitric Oxide Synthase
Oxidative Stress*
Quercetin
Reactive Oxygen Species
Rutin
Sonchus*
Superoxide Dismutase
Tumor Necrosis Factor-alpha
Wounds and Injuries
Antioxidants
Cyclooxygenase 2
Cytokines
Glutathione
Heme Oxygenase-1
Interleukin-6
Nitric Oxide
Nitric Oxide Synthase
Quercetin
Reactive Oxygen Species
Rutin
Superoxide Dismutase
Tumor Necrosis Factor-alpha
Figure
Reference
-
1. Leopold JA, Loscalzo J. Oxidative mechanisms and atherothrombotic cardiovascular disease. Drug Discov Today Ther Strateg. 2008; 5:5–13.
Article2. Turrens JF, Boveris A. Generation of superoxide anion by the NADH dehydrogenase of bovine heart mitochondria. Biochem J. 1980; 191:421–427.
Article3. Griendling KK, Sorescu D, Ushio-Fukai M. NAD(P)H oxidase: role in cardiovascular biology and disease. Circ Res. 2000; 86:494–501.4. Romano M, Claria J. Cyclooxygenase-2 and 5-lipoxygenase converging functions on cell proliferation and tumor angiogenesis: implications for cancer therapy. FASEB J. 2003; 17:1986–1995.
Article5. Kinnula VL, Crapo JD. Superoxide dismutases in the lung and human lung diseases. Am J Respir Crit Care Med. 2003; 167:1600–1619.
Article6. Cai H, Harrison DG. Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ Res. 2000; 87:840–844.
Article7. Lakshmi SV, Padmaja G, Kuppusamy P, Kutala VK. Oxidative stress in cardiovascular disease. Indian J Biochem Biophys. 2009; 46:421–440.8. Benz CC, Yau C. Ageing, oxidative stress and cancer: paradigms in parallax. Nat Rev Cancer. 2008; 8:875–879.
Article9. Yung LM, Leung FP, Yao X, Chen ZY, Huang Y. Reactive oxygen species in vascular wall. Cardiovasc Hematol Disord Drug Targets. 2006; 6:1–19.
Article10. Reuter S, Gupta SC, Chaturvedi MM, Aggarwal BB. Oxidative stress, inflammation, and cancer: how are they linked? Free Radic Biol Med. 2010; 49:1603–1616.
Article11. Balasundram N, Sundram K, Samman S. Phenolic compounds in plants and agro industrial by-products: antioxidant activity, occurrence, and potential uses. Food Chem. 2006; 99:191–203.
Article12. Jeruto P, Lukhoba C, Ouma G, Otieno D, Mutai C. An ethnobotanical study of medicinal plants used by the Nandi people in Kenya. J Ethnopharmacol. 2008; 116:370–376.
Article13. Sabeen M, Ahmad SS. Exploring the folk medicinal flora of Abbotabad City, Pakistan. Ethnobot Leafl. 2009; 13:810–833.14. Khan RA, Khan MR, Sahreen S, Bokhari J. Prevention of CCl4-induced nephrotoxicity with Sonchus asper in rat. Food Chem Toxicol. 2010; 48:2469–2476.15. Wang L, Xu ML, Hu JH, Rasmussen SK, Wang MH. Codonopsis lanceolata extract induces G0/G1 arrest and apoptosis in human colon tumor HT-29 cells--involvement of ROS generation and polyamine depletion. Food Chem Toxicol. 2011; 49:149–154.
Article16. Tiwari M, Dwivedi UN, Kakkar P. Suppression of oxidative stress and pro-inflammatory mediators by Cymbopogon citratus D. Stapf extract in lipopolysaccharide stimulated murine alveolar macrophages. Food Chem Toxicol. 2010; 48:2913–2919.
Article17. Kakkar P, Das B, Viswanathan PN. A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys. 1984; 21:130–132.18. Cho JY, Baik KU, Jung JH, Park MH. In vitro anti-inflammatory effects of cynaropicrin, a sesquiterpene lactone, from Saussurea lappa. Eur J Pharmacol. 2000; 398:399–407.
Article19. Lee HJ, Hyun EA, Yoon WJ, Kim BH, Rhee MH, Kang HK, Cho JY, Yoo ES. In vitro anti-inflammatory and anti-oxidative effects of Cinnamomum camphora extracts. J Ethnopharmacol. 2006; 103:208–216.
Article20. Enayat S, Banerjee S. Comparative antioxidant activity of extracts from leaves, bark and catkins of Salix aegyptiaca sp. Food Chem. 2009; 116:23–28.
Article21. Rahman I, MacNee W. Regulation of redox glutathione levels and gene transcription in lung inflammation: therapeutic approaches. Free Radic Biol Med. 2000; 28:1405–1420.
Article22. Kleinert H, Euchenhofer C, Ihrig-Biedert I, Förstermann U. Glucocorticoids inhibit the induction of nitric oxide synthase II by down-regulating cytokine-induced activity of transcription factor nuclear factor-kappa B. Mol Pharmacol. 1996; 49:15–21.23. Hennebert O, Pelissier MA, Le Mee S, Wülfert E, Morfin R. Anti-inflammatory effects and changes in prostaglandin patterns induced by 7beta-hydroxy-epiandrosterone in rats with colitis. J Steroid Biochem Mol Biol. 2008; 110:255–262.
Article24. Afolayan AJ, Jimoh FO. Nutritional quality of some wild leafy vegetables in South Africa. Int J Food Sci Nutr. 2009; 60:424–431.
Article25. Helal AM, Nakamura N, El-Askary H, Hattori M. Sesquiterpene lactone glucosides from Sonchus asper. Phytochemistry. 2000; 53:473–477.
Article26. Kim KM, Kwon YG, Chung HT, Yun YG, Pae HO, Han JA, Ha KS, Kim TW, Kim YM. Methanol extract of Cordyceps pruinosa inhibits in vitro and in vivo inflammatory mediators by suppressing NF-kappaB activation. Toxicol Appl Pharmacol. 2003; 190:1–8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibitory Effects of Paeonia Suffruticosa on the Expression of Inducible Nitric Oxide Synthase Gene in RAW264.7 Macrophages
- Acrosorium polyneurum Extract Inhibits the LPS-Induced Inflammatory Response by Impairing the MAPK and NF-κB Pathways
- Inhibitory Effects on Oral Microbial Activity and Production of Lipopolysaccharides-Induced Pro-Inflammatory Mediators in Raw264.7 Macrophages of Ethanol Extract of Perilla flutescens (L.) Britton
- Coptis chinensis Extract Inhibits the Production of Inflammatory Mediators and Delayed Type Hypersensitivity in Mice
- Rhamnazin inhibits LPS-induced inflammation and ROS/RNS in raw macrophages