Nutr Res Pract.
2014 Oct;8(5):509-515. 10.4162/nrp.2014.8.5.509.
Vitis amurensis Ruprecht root inhibited alpha-melanocyte stimulating hormone-induced melanogenesis in B16F10 cells
- Affiliations
-
- 1Blue-Bio Industry Regional Innovation Center, Dong-Eui University, 176 Eomgwangno, Busanjin-gu, Busan 614-714, Korea. bwkim@deu.ac.kr
- 2Department of Food Science and Nutrition, College of Natural Science, Dong-Eui University, Busan 614-714, Korea.
- 3Department of Life Science and Biotechnology, College of Natural Science, Dong-Eui University, Busan 614-714, Korea.
- KMID: 2313771
- DOI: http://doi.org/10.4162/nrp.2014.8.5.509
Abstract
- BACKGROUND/OBJECTIVES
The root of Vitis amurensis Ruprecht, a sort of wild-growing grape, has been used in oriental medicine for treatment of skin ailments; however, its dermatological activity is not sufficiently understood. The aim of this study was to investigate tyrosinase inhibitory and anti-melanogenic activities of V. amurensis Ruprecht root methanol extract (VARM) in B16F10 mouse melanoma cells and to attempt to isolate and identify the active compound issued from VARM.
MATERIALS/METHODS
Anti-melanogenic activity of VARM was analyzed in alpha-melanocyte stimulating hormone (MSH)-stimulated B16F10 cells through evaluation of antioxidative activity as well as inhibited tyrosinase activity and melanin contents compared with those of kojic acid and arbutin. After anti-melanogenic analysis of VARM, serial fractionation, nuclear magnetic resonance (NMR), and thin layer chromatorgraphy (TLC) were applied for identification of active compounds contained in VARM.
RESULTS
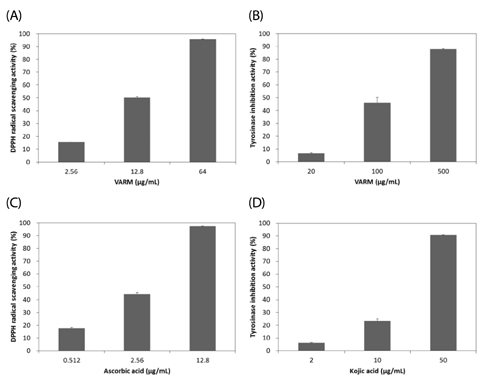
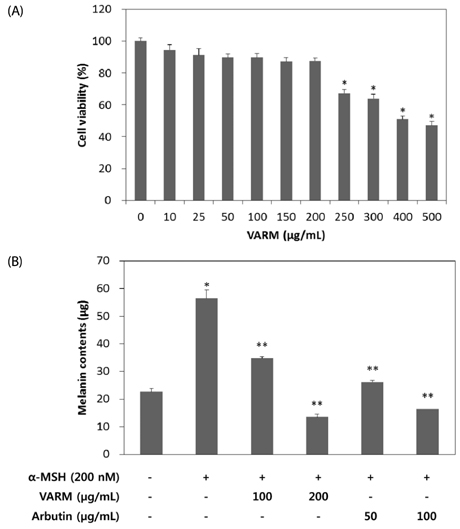
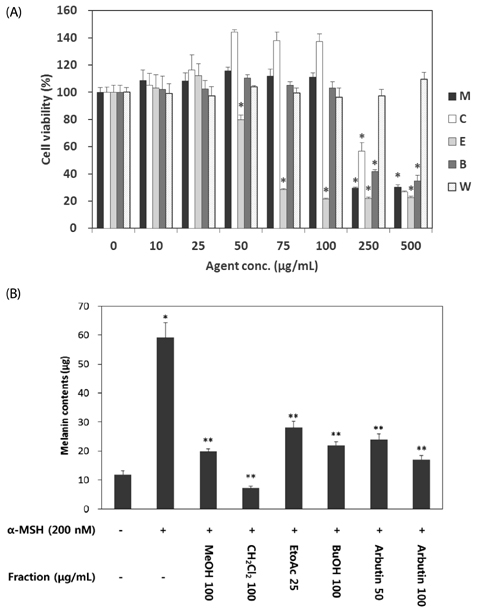
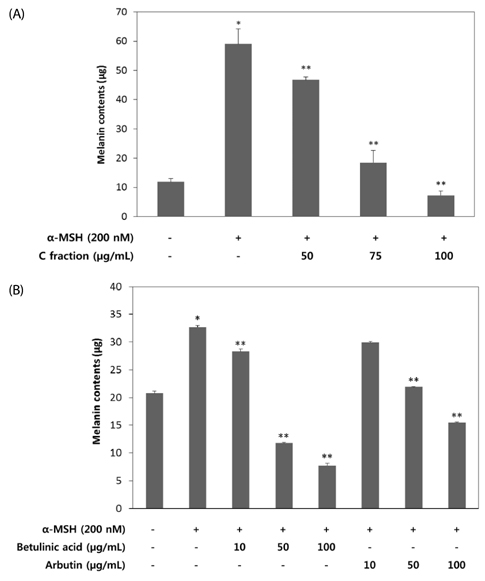
VARM significantly inhibited oxidative stress and tyrosinase activity and attenuated alpha-MSH-induced melanin production in B16F10 cells. For isolation of active compounds, VARM was fractionated using a series of organic solvents, including dichloromethane (CH2Cl2), ethyl acetate (EtOAc), and n-butanol (n-BuOH). Among fractions showing anti-melanogenic activity, the CH2Cl2 fraction induced the most potent attenuation of melanogenesis without cytotoxicity and the major compound in the CH2Cl2 fraction was identified as betulinic acid. Betulinic acid isolated from the CH2Cl2 fraction of VARM significantly attenuated alpha-MSH-induced melanogenesis in a dose dependent manner, which was stronger than that of arbutin used as a positive control.
CONCLUSIONS
These results indicate that VARM inhibits oxidative stress, tyrosinase activity, and alpha-MSH-induced melanogenesis in B16F10 cells, due primarily to the active compound, betulinic acid, in the CH2Cl2 fraction.
MeSH Terms
Figure
Reference
-
1. Yamaguchi Y, Brenner M, Hearing VJ. The regulation of skin pigmentation. J Biol Chem. 2007; 282:27557–27561.
Article2. Slominski A, Tobin DJ, Shibahara S, Wortsman J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol Rev. 2004; 84:1155–1228.
Article3. Briganti S, Camera E, Picardo M. Chemical and instrumental approaches to treat hyperpigmentation. Pigment Cell Res. 2003; 16:101–110.
Article4. Costin GE, Hearing VJ. Human skin pigmentation: melanocytes modulate skin color in response to stress. FASEB J. 2007; 21:976–994.
Article5. Kondo T, Hearing VJ. Update on the regulation of mammalian melanocyte function and skin pigmentation. Expert Rev Dermatol. 2011; 6:97–108.
Article6. Cheli Y, Luciani F, Khaled M, Beuret L, Bille K, Gounon P, Ortonne JP, Bertolotto C, Ballotti R. αMSH and cyclic AMP elevating agents control melanosome pH through a protein kinase A-independent mechanism. J Biol Chem. 2009; 284:18699–18706.
Article7. Passeron T, Namiki T, Passeron HJ, Le Pape E, Hearing VJ. Forskolin protects keratinocytes from UVB-induced apoptosis and increases DNA repair independent of its effects on melanogenesis. J Invest Dermatol. 2009; 129:162–166.
Article8. Lee J, Jung E, Park J, Jung K, Park E, Kim J, Hong S, Park J, Park S, Lee S, Park D. Glycyrrhizin induces melanogenesis by elevating a cAMP level in b16 melanoma cells. J Invest Dermatol. 2005; 124:405–411.
Article9. Gibbs S, Murli S, De Boer G, Mulder A, Mommaas AM, Ponec M. Melanosome capping of keratinocytes in pigmented reconstructed epidermis--effect of ultraviolet radiation and 3-isobutyl-1-methylxanthine on melanogenesis. Pigment Cell Res. 2000; 13:458–466.
Article10. Agar N, Young AR. Melanogenesis: a photoprotective response to DNA damage? Mutat Res. 2005; 571:121–132.
Article11. Wen KC, Chang CS, Chien YC, Wang HW, Wu WC, Wu CS, Chiang HM. Tyrosol and its analogues inhibit alpha-melanocyte-stimulating hormone induced melanogenesis. Int J Mol Sci. 2013; 14:23420–23440.
Article12. Lee JY, Choi HJ, Chung TW, Kim CH, Jeong HS, Ha KT. Caffeic acid phenethyl ester inhibits alpha-melanocyte stimulating hormone-induced melanin synthesis through suppressing transactivation activity of microphthalmia-associated transcription factor. J Nat Prod. 2013; 76:1399–1405.
Article13. Eves PC, MacNeil S, Haycock JW. α-Melanocyte stimulating hormone, inflammation and human melanoma. Peptides. 2006; 27:444–452.
Article14. Mastore M, Kohler L, Nappi AJ. Production and utilization of hydrogen peroxide associated with melanogenesis and tyrosinase-mediated oxidations of DOPA and dopamine. FEBS J. 2005; 272:2407–2415.
Article15. Chaubal VA, Nair SS, Ito S, Wakamatsu K, Mojamdar MV. γ-Glutamyl transpeptidase and its role in melanogenesis: redox reactions and regulation of tyrosinase. Pigment Cell Res. 2002; 15:420–425.
Article16. Ha do T, Kim H, Thuong PT, Ngoc TM, Lee I, Hung ND, Bae K. Antioxidant and lipoxygenase inhibitory activity of oligostilbenes from the leaf and stem of Vitis amurensis. J Ethnopharmacol. 2009; 125:304–309.
Article17. Weidner S, Powałka A, Karamać M, Amarowicz R. Extracts of phenolic compounds from seeds of three wild grapevines-comparison of their antioxidant activities and the content of phenolic compounds. Int J Mol Sci. 2012; 13:3444–3457.
Article18. Wang JN, Chen YJ, Hano Y, Nomura T, Tan RX. Antioxidant activity of polyphenols from seeds of Vitis amurensis in vitro. Acta Pharmacol Sin. 2000; 21:633–636.19. Jang MH, Piao XL, Kim JM, Kwon SW, Park JH. Inhibition of cholinesterase and amyloid-beta aggregation by resveratrol oligomers from Vitis amurensis. Phytother Res. 2008; 22:544–549.
Article20. Kim JY, Jeong HY, Lee HK, Kim S, Hwang BY, Bae K, Seong YH. Neuroprotection of the leaf and stem of Vitis amurensis and their active compounds against ischemic brain damage in rats and excitotoxicity in cultured neurons. Phytomedicine. 2012; 19:150–159.
Article21. Jeong HY, Kim JY, Lee HK, Ha do T, Song KS, Bae K, Seong YH. Leaf and stem of Vitis amurensis and its active components protect against amyloid beta protein (25-35)-induced neurotoxicity. Arch Pharm Res. 2010; 33:1655–1664.
Article22. Lee EO, Lee HJ, Hwang HS, Ahn KS, Chae C, Kang KS, Lu J, Kim SH. Potent inhibition of Lewis lung cancer growth by heyneanol A from the roots of Vitis amurensis through apoptotic and anti-angiogenic activities. Carcinogenesis. 2006; 27:2059–2069.
Article23. Kim JA, Kim MR, Kim O, Phuong NT, Yun J, Oh WK, Bae K, Kang KW. Amurensin G inhibits angiogenesis and tumor growth of tamoxifen-resistant breast cancer via Pin1 inhibition. Food Chem Toxicol. 2012; 50:3625–3634.
Article24. Yim N, Ha do T, Trung TN, Kim JP, Lee S, Na M, Jung H, Kim HS, Kim YH, Bae K. The antimicrobial activity of compounds from the leaf and stem of Vitis amurensis against two oral pathogens. Bioorg Med Chem Lett. 2010; 20:1165–1168.
Article25. Shin TY, Kim SH. Meoruh wine suppresses mast cell-mediated allergic inflammation. Immunopharmacol Immunotoxicol. 2011; 33:271–278.
Article26. Kim SH, Kwon TK, Shin TY. Antiallergic effects of Vitis amurensis on mast cell-mediated allergy model. Exp Biol Med (Maywood). 2008; 233:192–199.
Article27. Zhu L, Zhang Y, Lu J. Phenolic contents and compositions in skins of red wine grape cultivars among various genetic backgrounds and originations. Int J Mol Sci. 2012; 13:3492–3510.
Article28. Bak MJ, Jun M, Jeong WS. Procyanidins from wild grape (Vitis amurensis) seeds regulate ARE-mediated enzyme expression via Nrf2 coupled with p38 and PI3K/Akt pathway in HepG2 cells. Int J Mol Sci. 2012; 13:801–818.
Article29. Huang KS, Lin M. Oligostilbenes from the roots of Vitis amurensis. J Asian Nat Prod Res. 1999; 2:21–28.30. Oh WK, Cho KB, Hien TT, Kim TH, Kim HS, Dao TT, Han HK, Kwon SM, Ahn SG, Yoon JH, Kim TH, Kim YG, Kang KW. Amurensin G, a potent natural SIRT1 inhibitor, rescues doxorubicin responsiveness via down-regulation of multidrug resistance 1. Mol Pharmacol. 2010; 78:855–864.
Article31. Lee EO, Kwon BM, Song GY, Chae CH, Kim HM, Shim IS, Ahn KS, Kim SH. Heyneanol A induces apoptosis via cytochrome c release and caspase activation in human leukemic U937 cells. Life Sci. 2004; 74:2313–2326.
Article32. Huang KS, Lin M, Cheng GF. Anti-inflammatory tetramers of resveratrol from the roots of Vitis amurensis and the conformations of the seven-membered ring in some oligostilbenes. Phytochemistry. 2001; 58:357–362.
Article33. Kedare SB, Singh RP. Genesis and development of DPPH method of antioxidant assay. J Food Sci Technol. 2011; 48:412–422.
Article34. Piao LZ, Park HR, Park YK, Lee SK, Park JH, Park MK. Mushroom tyrosinase inhibition activity of some chromones. Chem Pharm Bull (Tokyo). 2002; 50:309–311.
Article35. Ngamwongsatit P, Banada PP, Panbangred W, Bhunia AK. WST-1-based cell cytotoxicity assay as a substitute for MTT-based assay for rapid detection of toxigenic Bacillus species using CHO cell line. J Microbiol Methods. 2008; 73:211–215.
Article36. Chung KW, Park YJ, Choi YJ, Park MH, Ha YM, Uehara Y, Yoon JH, Chun P, Moon HR, Chung HY. Evaluation of in vitro and in vivo anti-melanogenic activity of a newly synthesized strong tyrosinase inhibitor (E)-3-(2,4 dihydroxybenzylidene)pyrrolidine-2,5-dione (3-DBP). Biochim Biophys Acta. 2012; 1820:962–969.
Article37. Schwahn DJ, Xu W, Herrin AB, Bales ES, Medrano EE. Tyrosine levels regulate the melanogenic response to alpha-melanocyte-stimulating hormone in human melanocytes: implications for pigmentation and proliferation. Pigment Cell Res. 2001; 14:32–39.
Article38. Park HY, Wu C, Yaar M, Stachur CM, Kosmadaki M, Gilchrest BA. Role of BMP-4 and its signaling pathways in cultured human melanocytes. Int J Cell Biol. 2009; 2009:750482.
Article39. Ding HY, Lin HC, Chang TS. Tyrosinase inhibitors isolated from the roots of Paeonia suffruticosa. J Cosmet Sci. 2009; 60:347–352.40. Zi SX, Ma HJ, Li Y, Liu W, Yang QQ, Zhao G, Lian S. Oligomeric proanthocyanidins from grape seeds effectively inhibit ultraviolet-induced melanogenesis of human melanocytes in vitro. Int J Mol Med. 2009; 23:197–204.
Article41. Tan Y, Yu R, Pezzuto JM. Betulinic acid-induced programmed cell death in human melanoma cells involves mitogen-activated protein kinase activation. Clin Cancer Res. 2003; 9:2866–2875.42. Chowdhury AR, Mandal S, Mittra B, Sharma S, Mukhopadhyay S, Majumder HK. Betulinic acid, a potent inhibitor of eukaryotic topoisomerase I: identification of the inhibitory step, the major functional group responsible and development of more potent derivatives. Med Sci Monit. 2002; 8:BR254–BR265.43. Zuco V, Supino R, Righetti SC, Cleris L, Marchesi E, Gambacorti-Passerini C, Formelli F. Selective cytotoxicity of betulinic acid on tumor cell lines, but not on normal cells. Cancer Lett. 2002; 175:17–25.
Article44. Yogeeswari P, Sriram D. Betulinic acid and its derivatives: a review on their biological properties. Curr Med Chem. 2005; 12:657–666.
Article45. Choi JY, Choi EH, Jung HW, Oh JS, Lee WH, Lee JG, Son JK, Kim Y, Lee SH. Melanogenesis inhibitory compounds from Saussureae radix. Arch Pharm Res. 2008; 31:294–299.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Baicalein Inhibits αα-Melanocyte-stimulating Hormonestimulated Melanogenesis via p38 Mitogen-activated Protein Kinase Pathway in B16F10 Mouse Melanoma Cells
- Superoxide Dismutase 1 Inhibits Alpha-Melanocyte Stimulating Hormone and Ultraviolet B-Induced Melanogenesis in Murine Skin
- Inhibitory effect of Gastrodia elata Blume extract on alpha-melanocyte stimulating hormone-induced melanogenesis in murine B16F10 melanoma
- Diarylpropionitrile inhibits melanogenesis via protein kinase A/cAMP-response element-binding protein/microphthalmiaassociated transcription factor signaling pathway in α-MSHstimulated B16F10 melanoma cells
- Induction of Melanogenesis by Rapamycin in Human MNT-1 Melanoma Cells