Exendin-4 Improves Nonalcoholic Fatty Liver Disease by Regulating Glucose Transporter 4 Expression in ob/ob Mice
- Affiliations
-
- 1Department of Anatomy and Convergence Medical Science, Institutes of Health Science, Gyeongsnag National University School of Medicine, Jinju 660-751, Korea. anaroh@gnu.ac.kr
- 2Department of Internal Medicine, Institutes of Health Science, Gyeongsnag National University School of Medicine, Jinju 660-751, Korea.
- 3Department of Thoracic and Cardiovascular Surgery, Institutes of Health Science, Gyeongsnag National University School of Medicine, Jinju 660-751, Korea.
- 4Department of Neurologic Surgery, Biochemistry and Molecular Biology, Mayo Clinic College of Medicine, Rochester, MN 55905, USA.
- 5Division of Metabolic Diseases, Center for Biomedical Sciences, National Institutes of Health, Cheongwon-gun 363-700, Korea.
- KMID: 2285528
- DOI: http://doi.org/10.4196/kjpp.2014.18.4.333
Abstract
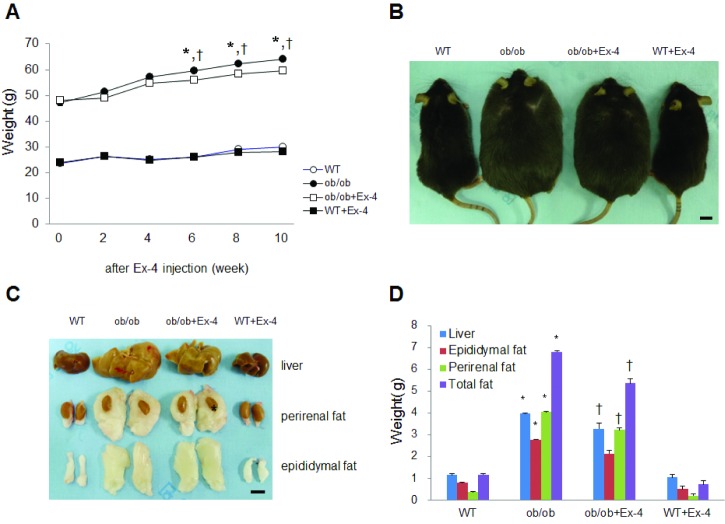
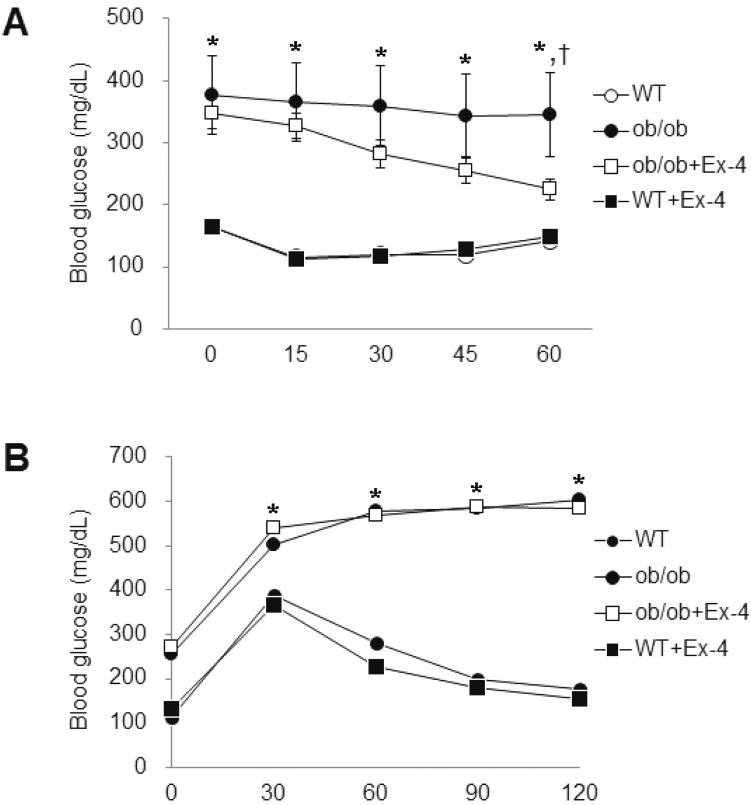
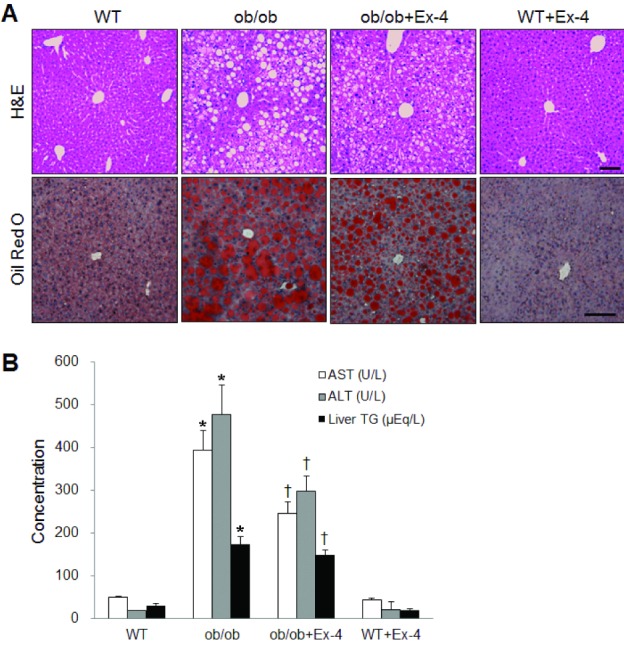
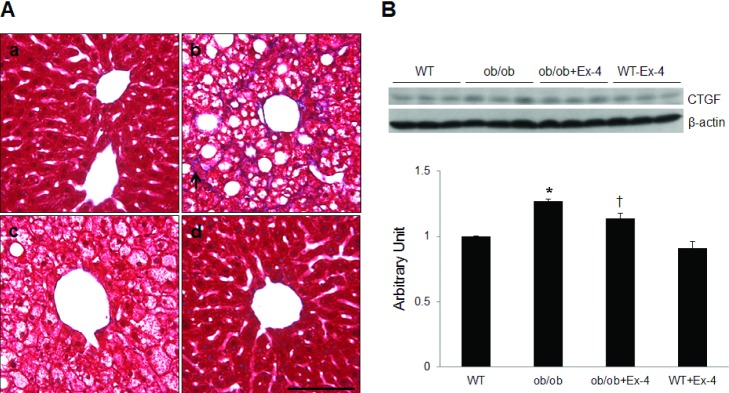
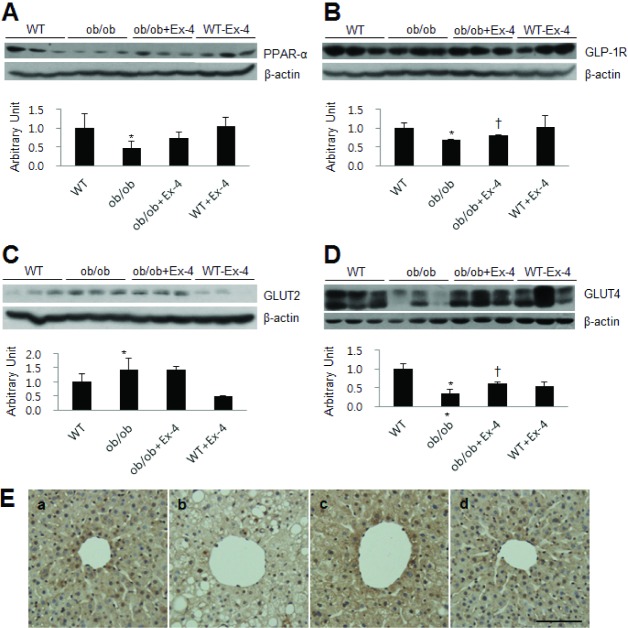
- Exendin-4 (Ex-4), a glucagon-like peptide-1 receptor (GLP-1R) agonist, has been known to reverse hepatic steatosis in ob/ob mice. Although many studies have evaluated molecular targets of Ex-4, its mechanism of action on hepatic steatosis and fibrosis has not fully been determined. In the liver, glucose transporter 4 (GLUT4) is mainly expressed in hepatocytes, endothelial cells and hepatic stellate cells (HSCs). In the present study, the effects of Ex-4 on GLUT4 expression were determined in the liver of ob/ob mice. Ob/ob mice were treated with Ex-4 for 10 weeks. Serum metabolic parameters, hepatic triglyceride levels, and liver tissues were evaluated for hepatic steatosis. The weights of the whole body and liver in ob/ob mice were reduced by long-term Ex-4 treatment. Serum metabolic parameters, hepatic steatosis, and hepatic fibrosis in ob/ob mice were reduced by Ex-4. Particularly, Ex-4 improved hepatic steatosis by enhancing GLUT4 via GLP-1R activation in ob/ob mice. Ex-4 treatment also inhibited hepatic fibrosis by decreasing expression of connective tissue growth factor in HSCs of ob/ob mice. Our data suggest that GLP-1 agonists exert a protective effect on hepatic steatosis and fibrosis in obesity and type 2 diabetes.
MeSH Terms
-
Animals
Connective Tissue Growth Factor
Endothelial Cells
Fatty Liver*
Fibrosis
Glucagon-Like Peptide 1
Glucagon-Like Peptide-1 Receptor
Glucose Transport Proteins, Facilitative*
Hepatic Stellate Cells
Hepatocytes
Liver
Mice*
Obesity
Triglycerides
Weights and Measures
Connective Tissue Growth Factor
Glucagon-Like Peptide 1
Glucose Transport Proteins, Facilitative
Figure
Cited by 3 articles
-
EGCG Blocked Phenylephrin-Induced Hypertrophy in H9C2 Cardiomyocytes, by Activating AMPK-Dependent Pathway
Yi Cai, Li Zhao, Yuan Qin, Xiao-Qian Wu
Korean J Physiol Pharmacol. 2015;19(3):203-210. doi: 10.4196/kjpp.2015.19.3.203.Myeloid-specific SIRT1 Deletion Aggravates Hepatic Inflammation and Steatosis in High-fat Diet-fed Mice
Kyung Eun Kim, Hwajin Kim, Rok Won Heo, Hyun Joo Shin, Chin-ok Yi, Dong Hoon Lee, Hyun Joon Kim, Sang Soo Kang, Gyeong Jae Cho, Wan Sung Choi, Gu Seob Roh
Korean J Physiol Pharmacol. 2015;19(5):451-460. doi: 10.4196/kjpp.2015.19.5.451.Anti-diabetic activities of catalpol in db/db mice
Qinwen Bao, Xiaozhu Shen, Li Qian, Chen Gong, Maoxiao Nie, Yan Dong
Korean J Physiol Pharmacol. 2016;20(2):153-160. doi: 10.4196/kjpp.2016.20.2.153.
Reference
-
1. Park SH, Jeon WK, Kim SH, Kim HJ, Park DI, Cho YK, Sung IK, Sohn CI, Keum DK, Kim BI. Prevalence and risk factors of non-alcoholic fatty liver disease among Korean adults. J Gastroenterol Hepatol. 2006; 21:138–143. PMID: 16706825.
Article2. Malhi H, Gores GJ. Molecular mechanisms of lipotoxicity in nonalcoholic fatty liver disease. Semin Liver Dis. 2008; 28:360–369. PMID: 18956292.
Article3. Sheth SG, Gordon FD, Chopra S. Nonalcoholic steatohepatitis. Ann Intern Med. 1997; 126:137–145. PMID: 9005748.
Article4. Adams LA, Lymp JF, St Sauver J, Sanderson SO, Lindor KD, Feldstein A, Angulo P. The natural history of nonalcoholic fatty liver disease: a population-based cohort study. Gastroenterology. 2005; 129:113–121. PMID: 16012941.
Article5. Postic C, Girard J. The role of the lipogenic pathway in the development of hepatic steatosis. Diabetes Metab. 2008; 34:643–648. PMID: 19195625.
Article6. Browning JD, Horton JD. Molecular mediators of hepatic steatosis and liver injury. J Clin Invest. 2004; 114:147–152. PMID: 15254578.
Article7. Frühbeck G, Gómez-Ambrosi J. Modulation of the leptin-induced white adipose tissue lipolysis by nitric oxide. Cell Signal. 2001; 13:827–833. PMID: 11583918.
Article8. Wang Y, Kole HK, Montrose-Rafizadeh C, Perfetti R, Bernier M, Egan JM. Regulation of glucose transporters and hexose uptake in 3T3-L1 adipocytes: glucagon-like peptide-1 and insulin interactions. J Mol Endocrinol. 1997; 19:241–248. PMID: 9460645.
Article9. Villanueva-Peñacarrillo ML, Puente J, Redondo A, Clemente F, Valverde I. Effect of GLP-1 treatment on GLUT2 and GLUT4 expression in type 1 and type 2 rat diabetic models. Endocrine. 2001; 15:241–248. PMID: 11720253.10. Ding X, Guo L, Zhang Y, Fan S, Gu M, Lu Y, Jiang D, Li Y, Huang C, Zhou Z. Extracts of pomelo peels prevent high-fat diet-induced metabolic disorders in c57bl/6 mice through activating the PPARα and GLUT4 pathway. PLoS One. 2013; 8:e77915. PMID: 24147098.
Article11. Karim S, Adams DH, Lalor PF. Hepatic expression and cellular distribution of the glucose transporter family. World J Gastroenterol. 2012; 18:6771–6781. PMID: 23239915.
Article12. Tang Y, Chen A. Curcumin prevents leptin raising glucose levels in hepatic stellate cells by blocking translocation of glucose transporter-4 and increasing glucokinase. Br J Pharmacol. 2010; 161:1137–1149. PMID: 20977462.
Article13. Eng J, Kleinman WA, Singh L, Singh G, Raufman JP. Isolation and characterization of exendin-4, an exendin-3 analogue, from Heloderma suspectum venom. Further evidence for an exendin receptor on dispersed acini from guinea pig pancreas. J Biol Chem. 1992; 267:7402–7405. PMID: 1313797.
Article14. Schepp W, Schmidtler J, Riedel T, Dehne K, Schusdziarra V, Holst JJ, Eng J, Raufman JP, Classen M. Exendin-4 and exendin-(9-39)NH2: agonist and antagonist, respectively, at the rat parietal cell receptor for glucagon-like peptide-1-(7-36)NH2. Eur J Pharmacol. 1994; 269:183–191. PMID: 7851494.
Article15. Fehmann HC, Jiang J, Schweinfurth J, Wheeler MB, Boyd AE 3rd, Göke B. Stable expression of the rat GLP-I receptor in CHO cells: activation and binding characteristics utilizing GLP-I(7-36)-amide, oxyntomodulin, exendin-4, and exendin (9-39). Peptides. 1994; 15:453–456. PMID: 7937318.16. Göke R, Fehmann HC, Linn T, Schmidt H, Krause M, Eng J, Göke B. Exendin-4 is a high potency agonist and truncated exendin-(9-39)-amide an antagonist at the glucagon-like peptide 1-(7-36)-amide receptor of insulin-secreting beta-cells. J Biol Chem. 1993; 268:19650–19655. PMID: 8396143.
Article17. Shirazi R, Palsdottir V, Collander J, Anesten F, Vogel H, Langlet F, Jaschke A, Schürmann A, Prévot V, Shao R, Jansson JO, Skibicka KP. Glucagon-like peptide 1 receptor induced suppression of food intake, and body weight is mediated by central IL-1 and IL-6. Proc Natl Acad Sci U S A. 2013; 110:16199–16204. PMID: 24048027.
Article18. Vilsbøll T, Holst JJ. Incretins, insulin secretion and Type 2 diabetes mellitus. Diabetologia. 2004; 47:357–366. PMID: 14968296.
Article19. Creutzfeldt WO, Kleine N, Willms B, Orskov C, Holst JJ, Nauck MA. Glucagonostatic actions and reduction of fasting hyperglycemia by exogenous glucagon-like peptide I(7-36) amide in type I diabetic patients. Diabetes Care. 1996; 19:580–586. PMID: 8725855.
Article20. Vilsbøll T, Toft-Nielsen MB, Krarup T, Madsbad S, Dinesen B, Holst JJ. Evaluation of beta-cell secretory capacity using glucagon-like peptide 1. Diabetes Care. 2000; 23:807–812. PMID: 10841001.
Article21. Nauck MA, Niedereichholz U, Ettler R, Holst JJ, Orskov C, Ritzel R, Schmiegel WH. Glucagon-like peptide 1 inhibition of gastric emptying outweighs its insulinotropic effects in healthy humans. Am J Physiol. 1997; 273:E981–E988. PMID: 9374685.22. Ding X, Saxena NK, Lin S, Gupta NA, Anania FA. Exendin-4, a glucagon-like protein-1 (GLP-1) receptor agonist, reverses hepatic steatosis in ob/ob mice. Hepatology. 2006; 43:173–181. PMID: 16374859.23. Van Wagner LB, Rinella ME. The role of insulin-sensitizing agents in the treatment of nonalcoholic steatohepatitis. Therap Adv Gastroenterol. 2011; 4:249–263.
Article24. Gupta NA, Mells J, Dunham RM, Grakoui A, Handy J, Saxena NK, Anania FA. Glucagon-like peptide-1 receptor is present on human hepatocytes and has a direct role in decreasing hepatic steatosis in vitro by modulating elements of the insulin signaling pathway. Hepatology. 2010; 51:1584–1592. PMID: 20225248.
Article25. Ben-Shlomo S, Zvibel I, Shnell M, Shlomai A, Chepurko E, Halpern Z, Barzilai N, Oren R, Fishman S. Glucagon-like peptide-1 reduces hepatic lipogenesis via activation of AMP-activated protein kinase. J Hepatol. 2011; 54:1214–1223. PMID: 21145820.
Article26. Gupta NA, Kolachala VL, Jiang R, Abramowsky C, Romero R, Fifadara N, Anania F, Knechtle S, Kirk A. The glucagon-like peptide-1 receptor agonist Exendin 4 has a protective role in ischemic injury of lean and steatotic liver by inhibiting cell death and stimulating lipolysis. Am J Pathol. 2012; 181:1693–1701. PMID: 22960075.
Article27. Trevaskis JL, Griffin PS, Wittmer C, Neuschwander-Tetri BA, Brunt EM, Dolman CS, Erickson MR, Napora J, Parkes DG, Roth JD. Glucagon-like peptide-1 receptor agonism improves metabolic, biochemical, and histopathological indices of nonalcoholic steatohepatitis in mice. Am J Physiol Gastrointest Liver Physiol. 2012; 302:G762–G772. PMID: 22268099.
Article28. Lee J, Hong SW, Chae SW, Kim DH, Choi JH, Bae JC, Park SE, Rhee EJ, Park CY, Oh KW, Park SW, Kim SW, Lee WY. Exendin-4 improves steatohepatitis by increasing Sirt1 expression in high-fat diet-induced obese C57BL/6J mice. PLoS One. 2012; 7:e31394. PMID: 22363635.
Article29. Krawczyk M, Bonfrate L, Portincasa P. Nonalcoholic fatty liver disease. Best Pract Res Clin Gastroenterol. 2010; 24:695–708. PMID: 20955971.
Article30. Svegliati-Baroni G, Saccomanno S, Rychlicki C, Agostinelli L, De Minicis S, Candelaresi C, Faraci G, Pacetti D, Vivarelli M, Nicolini D, Garelli P, Casini A, Manco M, Mingrone G, Risaliti A, Frega GN, Benedetti A, Gastaldelli A. Glucagon-like peptide-1 receptor activation stimulates hepatic lipid oxidation and restores hepatic signalling alteration induced by a high-fat diet in nonalcoholic steatohepatitis. Liver Int. 2011; 31:1285–1297. PMID: 21745271.
Article31. Kota BP, Huang TH, Roufogalis BD. An overview on biological mechanisms of PPARs. Pharmacol Res. 2005; 51:85–94. PMID: 15629253.
Article32. Brumbaugh DE, Friedman JE. Developmental origins of nonalcoholic fatty liver disease. Pediatr Res. 2014; 75:140–147. PMID: 24192698.
Article33. Zhao FQ, Keating AF. Functional properties and genomics of glucose transporters. Curr Genomics. 2007; 8:113–128. PMID: 18660845.
Article34. González-Périz A, Horrillo R, Ferré N, Gronert K, Dong B, Morán-Salvador E, Titos E, Martínez-Clemente M, López-Parra M, Arroyo V, Clària J. Obesity-induced insulin resistance and hepatic steatosis are alleviated by omega-3 fatty acids: a role for resolvins and protectins. FASEB J. 2009; 23:1946–1957. PMID: 19211925.35. Gao H, Wang X, Zhang Z, Yang Y, Yang J, Li X, Ning G. GLP-1 amplifies insulin signaling by up-regulation of IRbeta, IRS-1 and Glut4 in 3T3-L1 adipocytes. Endocrine. 2007; 32:90–95. PMID: 17992607.36. Jung TS, Kim SK, Shin HJ, Jeon BT, Hahm JR, Roh GS. α-lipoic acid prevents non-alcoholic fatty liver disease in OLETF rats. Liver Int. 2012; 32:1565–1573. PMID: 22863080.
Article37. Friedman SL. Hepatic stellate cells: protean, multifunctional, and enigmatic cells of the liver. Physiol Rev. 2008; 88:125–172. PMID: 18195085.
Article38. Kisseleva T, Brenner DA. Role of hepatic stellate cells in fibrogenesis and the reversal of fibrosis. J Gastroenterol Hepatol. 2007; 22(Suppl 1):S73–S78. PMID: 17567473.
Article39. Williams EJ, Gaça MD, Brigstock DR, Arthur MJ, Benyon RC. Increased expression of connective tissue growth factor in fibrotic human liver and in activated hepatic stellate cells. J Hepatol. 2000; 32:754–761. PMID: 10845662.
Article40. Paradis V, Perlemuter G, Bonvoust F, Dargere D, Parfait B, Vidaud M, Conti M, Huet S, Ba N, Buffet C, Bedossa P. High glucose and hyperinsulinemia stimulate connective tissue growth factor expression: a potential mechanism involved in progression to fibrosis in nonalcoholic steatohepatitis. Hepatology. 2001; 34:738–744. PMID: 11584370.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Increased Plasma Dipeptidyl Peptidase IV Activities in ob/ob Mice
- Hog millet (Panicum miliaceum L.)-supplemented diet ameliorates hyperlipidemia and hepatic lipid accumulation in C57BL/6J-ob/ob mice
- Glucose-Lowering Agents in the Management of Nonalcoholic Fatty Liver Disease
- Cordyceps militaris alleviates non-alcoholic fatty liver disease in ob/ob mice
- Effects of Cyclo-His-Pro-enriched yeast hydrolysate on blood glucose levels and lipid metabolism in obese diabetic ob/ob mice