Ann Dermatol.
2015 Feb;27(1):32-39. 10.5021/ad.2015.27.1.32.
Synergistic Inhibition of Tumor Necrosis Factor-Alpha-Stimulated Pro-Inflammatory Cytokine Expression in HaCaT Cells by a Combination of Rapamycin and Mycophenolic Acid
- Affiliations
-
- 1Department of Dermatology, Chung-Ang University College of Medicine, Seoul, Korea. beomjoon@unitel.co.kr
- 2Department of Dermatology, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 3Department of Dermatology, St. Paul's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 2264834
- DOI: http://doi.org/10.5021/ad.2015.27.1.32
Abstract
- BACKGROUND
Keratinocytes release various pro-inflammatory cytokines, chemokines, and adhesion molecules such as intercellular adhesion molecule 1 (ICAM-1) in response to cytokines such as tumor necrosis factor (TNF)-alpha and interferon (IFN)-gamma. Rapamycin and mycophenolic acid (MPA) have potent immunosuppressive activity because they inhibit lymphocyte proliferation.
OBJECTIVE
We investigated the effects of rapamycin and MPA on the expression of inflammation-related factors such as ICAM-1 and inducible nitric oxide synthase (iNOS), pro-inflammatory cytokines and chemokines, and related signaling pathways in TNF-alpha-stimulated HaCaT cells.
METHODS
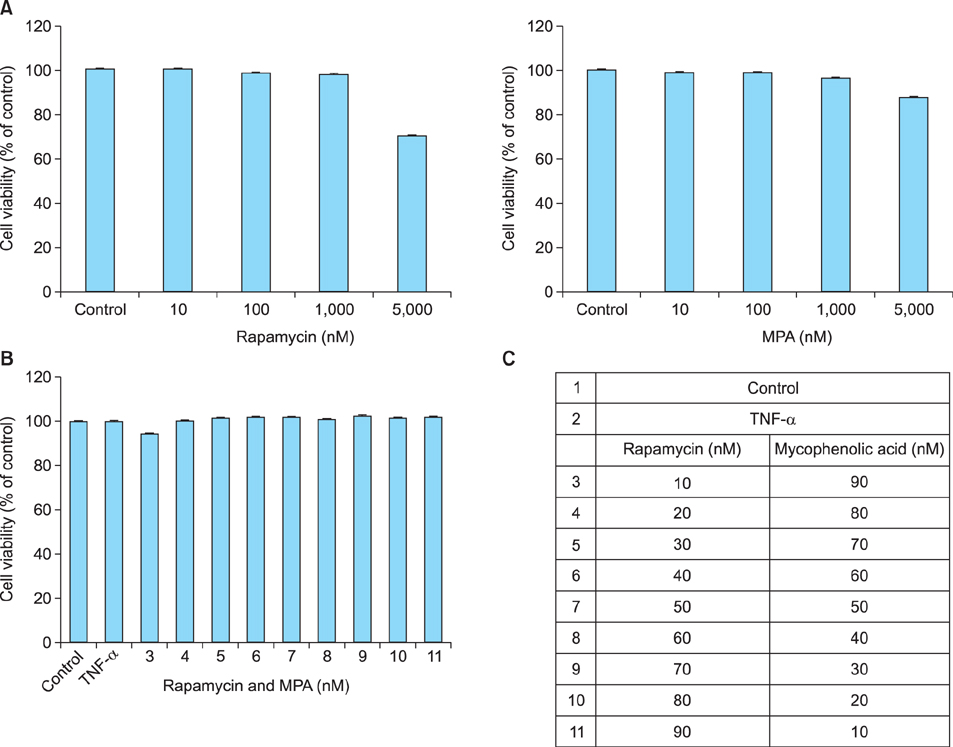
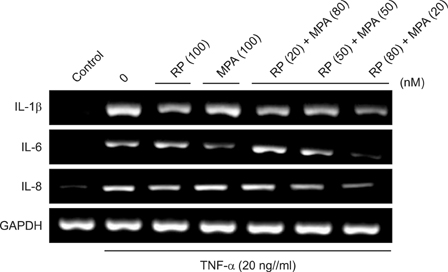
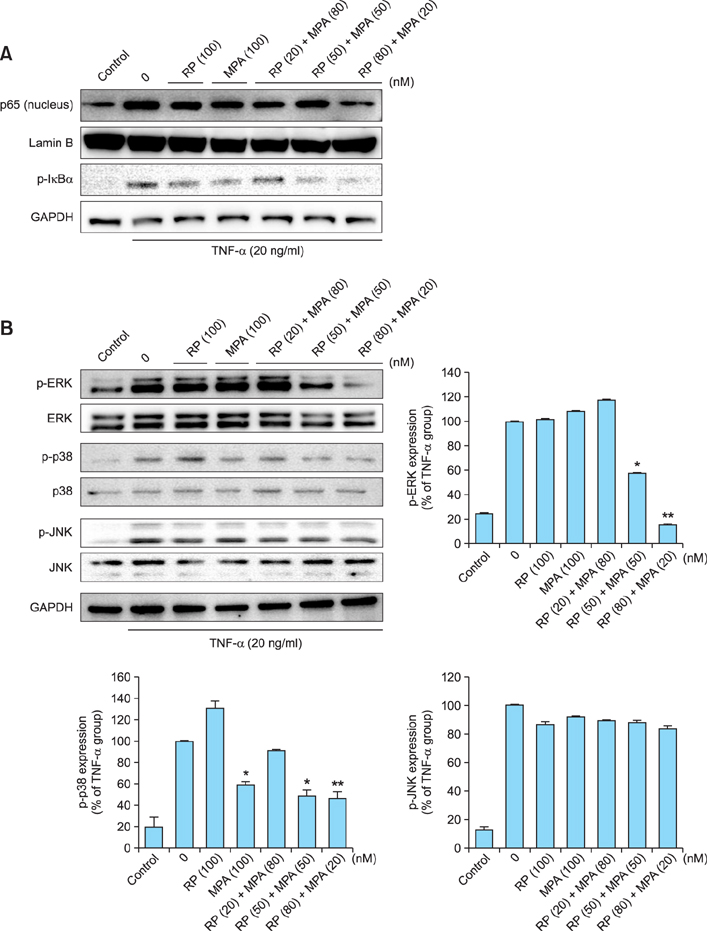
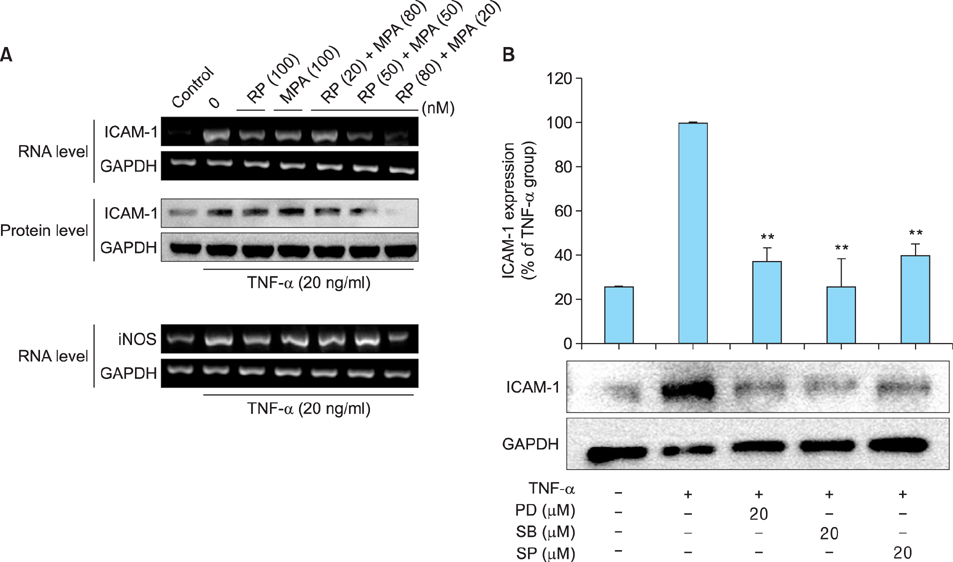
The viability of HaCaT cells treated with rapamycin and MPA was confirmed using MTT assay. The expression of various cytokines such as interleukin (IL)-1beta, IL-6, and IL-8; inflammation-related factors such as ICAM-1 and iNOS; and the activation of mitogen activated protein kinase (MAPK) signaling pathways mediated by extracellular signal-related kinases (ERK), p38, and c-Jun N-terminal kinases (JNK) in TNF-alpha-stimulated HaCaT cells were confirmed using reverse transcription-polymerase chain reaction and western blotting.
RESULTS
Combined treatment of TNF-alpha-induced HaCaT cells with rapamycin and MPA decreased ICAM-1 and iNOS expression and ERK and p38 activation more than treatment with either drug alone. The most significant decrease was observed with a combination of rapamycin (80 nM) and MPA (20 nM). These results show that co-treatment with these agents has a synergistic anti-inflammatory effect by blocking the activation of the ERK/p38 MAPK signaling pathway and thus suppressing the TNF-alpha-induced expression of ICAM-1 and iNOS.
CONCLUSION
The combination of rapamycin and MPA could potentially be used as a therapeutic approach in inflammatory skin diseases.
MeSH Terms
-
Blotting, Western
Chemokines
Cytokines
Intercellular Adhesion Molecule-1
Interferons
Interleukin-6
Interleukin-8
Interleukins
Keratinocytes
Lymphocytes
Mycophenolic Acid*
Necrosis*
Nitric Oxide Synthase Type II
Phosphotransferases
Protein Kinases
Sirolimus*
Skin Diseases
Tumor Necrosis Factor-alpha
Chemokines
Cytokines
Intercellular Adhesion Molecule-1
Interferons
Interleukin-6
Interleukin-8
Interleukins
Mycophenolic Acid
Nitric Oxide Synthase Type II
Phosphotransferases
Protein Kinases
Sirolimus
Tumor Necrosis Factor-alpha
Figure
Reference
-
1. Bos JD, Kapsenberg ML. The skin immune system: progress in cutaneous biology. Immunol Today. 1993; 14:75–78.
Article2. Kupper TS. Mechanisms of cutaneous inflammation. Interactions between epidermal cytokines, adhesion molecules, and leukocytes. Arch Dermatol. 1989; 125:1406–1412.
Article3. Barker JN, Mitra RS, Griffiths CE, Dixit VM, Nickoloff BJ. Keratinocytes as initiators of inflammation. Lancet. 1991; 337:211–214.
Article4. Cho JW, Lee KS, Kim CW. Curcumin attenuates the expression of IL-1beta, IL-6, and TNF-alpha as well as cyclin E in TNF-alpha-treated HaCaT cells; NF-kappaB and MAPKs as potential upstream targets. Int J Mol Med. 2007; 19:469–474.5. Albanesi C, Cavani A, Girolomoni G. Interferon-gamma-stimulated human keratinocytes express the genes necessary for the production of peptide-loaded MHC class II molecules. J Invest Dermatol. 1998; 110:138–142.6. Trefzer U, Brockhaus M, Loetscher H, Parlow F, Kapp A, Schöpf E, et al. 55-kd tumor necrosis factor receptor is expressed by human keratinocytes and plays a pivotal role in regulation of human keratinocyte ICAM-1 expression. J Invest Dermatol. 1991; 97:911–916.
Article7. Arnott CH, Scott KA, Moore RJ, Robinson SC, Thompson RG, Balkwill FR. Expression of both TNF-alpha receptor subtypes is essential for optimal skin tumour development. Oncogene. 2004; 23:1902–1910.
Article8. Bachelez H. Immunopathogenesis of psoriasis: recent insights on the role of adaptive and innate immunity. J Autoimmun. 2005; 25:Suppl. 69–73.
Article9. Bahar-Shany K, Ravid A, Koren R. Upregulation of MMP-9 production by TNFalpha in keratinocytes and its attenuation by vitamin D. J Cell Physiol. 2010; 222:729–737.10. Banno T, Gazel A, Blumenberg M. Effects of tumor necrosis factor-alpha (TNF alpha) in epidermal keratinocytes revealed using global transcriptional profiling. J Biol Chem. 2004; 279:32633–32642.
Article11. Chen G, Goeddel DV. TNF-R1 signaling: a beautiful pathway. Science. 2002; 296:1634–1635.
Article12. Mercurio F, Young DB, Manning AM. Detection and purification of a multiprotein kinase complex from mammalian cells. IKK signalsome. Methods Mol Biol. 2000; 99:109–125.
Article13. Lawrence T, Gilroy DW, Colville-Nash PR, Willoughby DA. Possible new role for NF-kappaB in the resolution of inflammation. Nat Med. 2001; 7:1291–1297.
Article14. Baldwin AS Jr. The NF-kappa B and I kappa B proteins: new discoveries and insights. Annu Rev Immunol. 1996; 14:649–683.15. Baeuerle PA, Baltimore D. NF-kappa B: ten years after. Cell. 1996; 87:13–20.16. Young CN, Koepke JI, Terlecky LJ, Borkin MS, Boyd Savoy L, Terlecky SR. Reactive oxygen species in tumor necrosis factor-alpha-activated primary human keratinocytes: implications for psoriasis and inflammatory skin disease. J Invest Dermatol. 2008; 128:2606–2614.
Article17. Mason J. Pharmacology of cyclosporine (sandimmune). VII. Pathophysiology and toxicology of cyclosporine in humans and animals. Pharmacol Rev. 1990; 41:423–434.18. Lipsky JJ. Mycophenolate mofetil. Lancet. 1996; 348:1357–1359.
Article19. Huang S, Houghton PJ. Inhibitors of mammalian target of rapamycin as novel antitumor agents: from bench to clinic. Curr Opin Investig Drugs. 2002; 3:295–304.20. Sabatini DM, Pierchala BA, Barrow RK, Schell MJ, Snyder SH. The rapamycin and FKBP12 target (RAFT) displays phosphatidylinositol 4-kinase activity. J Biol Chem. 1995; 270:20875–20878.
Article21. Lynch WS, Roenigk HH Jr. Mycophenolic acid for psoriasis. Arch Dermatol. 1977; 113:1203–1208.
Article22. Epinette WW, Parker CM, Jones EL, Greist MC. Mycophenolic acid for psoriasis. A review of pharmacology, long-term efficacy, and safety. J Am Acad Dermatol. 1987; 17:962–971.23. Ransom JT. Mechanism of action of mycophenolate mofetil. Ther Drug Monit. 1995; 17:681–684.
Article24. Allison AC, Eugui EM. Immunosuppressive and other effects of mycophenolic acid and an ester prodrug, mycophenolate mofetil. Immunol Rev. 1993; 136:5–28.
Article25. Langford CA, Klippel JH, Balow JE, James SP, Sneller MC. Use of cytotoxic agents and cyclosporine in the treatment of autoimmune disease. Part 2: Inflammatory bowel disease, systemic vasculitis, and therapeutic toxicity. Ann Intern Med. 1998; 129:49–58.
Article26. Taylor AC, Connell WR, Elliott R, d'Apice AJ. Oral cyclosporin in refractory inflammatory bowel disease. Aust N Z J Med. 1998; 28:179–183.
Article27. Bobadilla NA, Gamba G, Tapia E, García-Torres R, Bolio A, López-Zetina P, et al. Role of NO in cyclosporin nephrotoxicity: effects of chronic NO inhibition and NO synthases gene expression. Am J Physiol. 1998; 274:F791–F798.28. Gerber DA, Bonham CA, Thomson AW. Immunosuppressive agents: recent developments in molecular action and clinical application. Transplant Proc. 1998; 30:1573–1579.
Article29. Hutcheson AE, Rao MR, Olinde KD, Markov AK. Myocardial toxicity of cyclosporin A: inhibition of calcium ATPase and nitric oxide synthase activities and attenuation by fructose-1,6-diphosphate in vitro. Res Commun Mol Pathol Pharmacol. 1995; 89:17–26.30. Stein CM, Pincus T, Yocum D, Tugwell P, Wells G, Gluck O, et al. The Methotrexate-Cyclosporine Combination Study Group. Combination treatment of severe rheumatoid arthritis with cyclosporine and methotrexate for forty-eight weeks: an open-label extension study. Arthritis Rheum. 1997; 40:1843–1851.
Article31. Cohn RG, Mirkovich A, Dunlap B, Burton P, Chiu SH, Eugui E, et al. Mycophenolic acid increases apoptosis, lysosomes and lipid droplets in human lymphoid and monocytic cell lines. Transplantation. 1999; 68:411–418.
Article32. Blaheta RA, Leckel K, Wittig B, Zenker D, Oppermann E, Harder S, et al. Mycophenolate mofetil impairs transendothelial migration of allogeneic CD4 and CD8 T-cells. Transplant Proc. 1999; 31:1250–1252.
Article33. Govindarajan B, Willoughby L, Band H, Curatolo AS, Veledar E, Chen S, et al. Cooperative benefit for the combination of rapamycin and imatinib in tuberous sclerosis complex neoplasia. Vasc Cell. 2012; 4:11.
Article34. Werzowa J, Cejka D, Fuereder T, Dekrout B, Thallinger C, Pehamberger H, et al. Suppression of mTOR complex 2-dependent AKT phosphorylation in melanoma cells by combined treatment with rapamycin and LY294002. Br J Dermatol. 2009; 160:955–964.
Article35. Martin DF, DeBarge LR, Nussenblatt RB, Chan CC, Roberge FG. Synergistic effect of rapamycin and cyclosporin A in the treatment of experimental autoimmune uveoretinitis. J Immunol. 1995; 154:922–927.36. Krutmann J, Czech W, Parlow F, Trefzer U, Kapp A, Schöpf E, et al. Ultraviolet radiation effects on human keratinocyte ICAM-1 expression: UV-induced inhibition of cytokine-induced ICAM-1 mRNA expression is transient, differentially restored for IFN gamma versus TNF alpha, and followed by ICAM-1 induction via a TNF alpha-like pathway. J Invest Dermatol. 1992; 98:923–928.
Article37. Winiski AP, Foster CA. ICAM-1 expression in a spontaneously transformed human keratinocyte cell line: characterization by a simple cell-ELISA assay. J Invest Dermatol. 1992; 99:48–52.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Role of Gallic Acid in Inflammatory Allergic Process
- The Effect of Blue Light on Propionibacterium acnes and the Expression of Inflammatory Cytokines in HaCaT Cells
- A Mechanism for the Up-regulation of the IL-8 Gene Expression in Keratinocytes by All-trans Retinoic Acid
- Pro-inflammatory cytokine expression through NF-kappaB/IkappaB pathway in lung epithelial cells
- The Expression of ICAM-1 by Cytokines and the Effect of Dexamethasone on ICAM-1 Expression in Cultured Keratocytes