Cancer Res Treat.
2004 Aug;36(4):214-221.
DNA Damage Response Mediated through BRCA1
- Affiliations
-
- 1Research Institute, National Cancer Center, Goyang, Gyeonggi, Korea. leejs@ncc.re.kr
- 2College of Natural Sciences, Seoul National University, Seoul, Korea.
Abstract
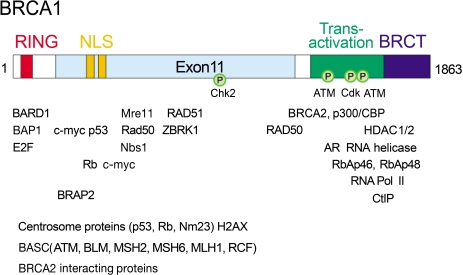
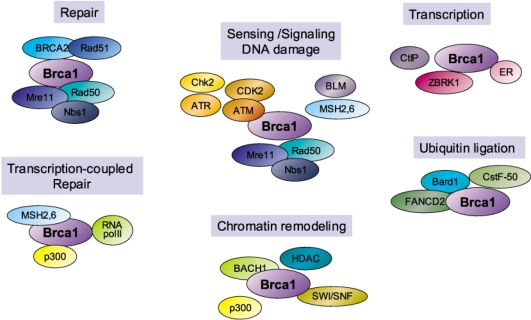
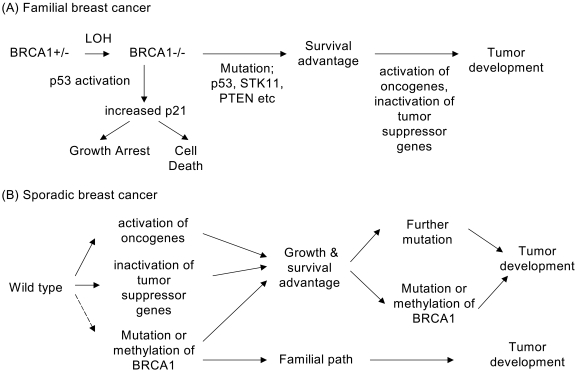
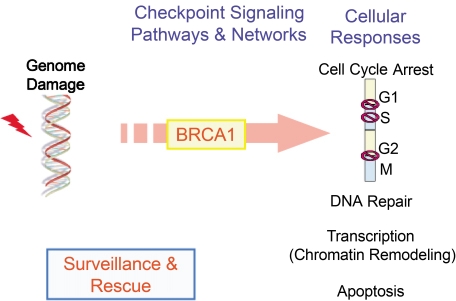
- The BRCA1 gene was identified and cloned in 1994 based on its linkage to early onset breast and ovarian cancer syndromes in women. The tumor suppressor, BRCA1 is known as a major player in the DNA damage response. These are evident from its loss, which causes malignant transformation in breast and ovary, and renders cells to become sensitive to a wide variety of DNA damaging agents. Here, we have implications on functional coupling of the pleiotropic roles of BRCA1, including DNA damage signal networking, DNA repair, transcription, and checkpoint of cell cycle, to tumor suppression by examining the molecular mechanisms and functions of BRCA1. The breast cancer susceptibility 1 (BRCA1) gene was identified and mapped to chromosome 17q21 by analyzing families at high risk from breast and ovarian cancer, and was first cloned in 1994 (1). The BRCA1 gene encodes a large nuclear protein that is ubiquitously expressed in a number of tissues. BRCA1 shares little structural resemblance to the majority of other known proteins (Fig. 1). Its ortholog is only found in mammals but not in yeast, fly, worm, or zebra fish, indicating that BRCA1 may come later in evolution and it may have more specialized and tissue-specific functions in mammalian cells. Although a number of studies delineating and deciphering the real biological roles of BRCA1 have accumulated, understanding these BRCA1 unique features still remains to be challengingly elucidated.
MeSH Terms
Figure
Reference
-
1. Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S, et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science. 1994; 266:66–71. PMID: 7545954.2. Neuhausen SL, Marshall CJ. Loss of heterozygosity in familial tumors from three BRCA1-linked kindreds. Cancer Res. 1994; 54:6069–6072. PMID: 7954448.3. Kinzler KW, Vogelstein B. Cancer-susceptibility genes. Gatekeepers and caretakers. Nature. 1997; 386:761–763. PMID: 9126728.4. Xu X, Weaver Z, Linke SP, Li C, Gotay J, Wang XW, Harris CC, Ried T, Deng CX. Centrosome amplification and a defective G2-M cell cycle checkpoint induce genetic instability in BRCA1 exon 11 isoform-deficient cells. Mol Cell. 1999; 3:389–395. PMID: 10198641.
Article5. Khanna KK, Jackson SP. DNA double-strand breaks: signaling, repair and the cancer connection. Nat Genet. 2001; 27:247–254. PMID: 11242102.
Article6. Shen SX, Weaver Z, Xu X, Li C, Weinstein M, Chen L, Guan XY, Ried T, Deng CX. A targeted disruption of the murine Brca1 gene causes gamma-irradiation hypersensitivity and genetic instability. Oncogene. 1998; 17:3115–3124. PMID: 9872327.7. Scully R, Chen J, Plug A, Xiao Y, Weaver D, Feunteun J, Ashley T, Livingston DM. Association of BRCA1 with Rad51 in mitotic and meiotic cells. Cell. 1997; 88:265–275. PMID: 9008167.
Article8. Zhong Q, Chen CF, Li S, Chen Y, Wang CC, Xiao J, Chen PL, Sharp ZD, Lee WH. Association of BRCA1 with the hRad50-hMre11-p95 complex and the DNA damage response. Science. 1999; 285:747–750. PMID: 10426999.
Article9. Wang Y, Cortez D, Yazdi P, Neff N, Elledge SJ, Qin J. BASC, a super complex of BRCA1-associated proteins involved in the recognition and repair of aberrant DNA structures. Genes Dev. 2000; 14:927–939. PMID: 10783165.
Article10. Le Page F, Randrianarison V, Marot D, Cabannes J, Perricaudet M, Feunteun J, Sarasin A. BRCA1 and BRCA2 are necessary for the transcription-coupled repair of the oxidative 8-oxoguanine lesion in human cells. Cancer Res. 2000; 60:5548–5552. PMID: 11034101.11. Scully R, Chen J, Ochs RL, Keegan K, Hoekstra M, Feunteun J, Livingston DM. Dynamic changes of BRCA1 subnuclear location and phosphorylation state are initiated by DNA damage. Cell. 1997; 90:425–435. PMID: 9267023.
Article12. Paull TT, Rogakou EP, Yamazaki V, Kirchgessner CU, Gellert M, Bonner WM. A critical role for histone H2AX in recruitment of repair factors to nuclear foci after DNA damage. Curr Biol. 2000; 10:886–895. PMID: 10959836.
Article13. Zhong Q, Boyer TG, Chen PL, Lee WH. Deficient nonhomologous end-joining activity in cell-free extracts from Brca1-null fibroblasts. Cancer Res. 2002; 62:3966–3970. PMID: 12124328.14. Ganesan S, Silver DP, Greenberg RA, Avni D, Drapkin R, Miron A, Mok SC, Randrianarison V, Brodie S, Salstrom J, Rasmussen TP, Klimke A, Marrese C, Marahrens Y, Deng CX, Feunteun J, Livingston DM. BRCA1 supports XIST RNA concentration on the inactive X chromosome. Cell. 2002; 111:393–405. PMID: 12419249.
Article15. Somasundaram K, Zhang H, Zeng YX, Houvras Y, Peng Y, Zhang H, Wu GS, Licht JD, Weber BL, El-Deiry WS. Arrest of the cell cycle by the tumour-suppressor BRCA1 requires the CDK-inhibitor p21WAF1/CiP1. Nature. 1997; 389:187–190. PMID: 9296497.16. Williamson EA, Dadmanesh F, Koeffler HP. BRCA1 transactivates the cyclin-dependent kinase inhibitor p27(Kip1). Oncogene. 2002; 21:3199–3206. PMID: 12082635.
Article17. Deng CX, Brodie SG. Roles of BRCA1 and its interacting proteins. Bioessays. 2000; 22:728–737. PMID: 10918303.
Article18. Yu X, Wu LC, Bowcock AM, Aronheim A, Baer R. The C-terminal (BRCT) domains of BRCA1 interact in vivo with CtIP, a protein implicated in the CtBP pathway of transcriptional repression. J Biol Chem. 1998; 273:25388–25392. PMID: 9738006.
Article19. Fan S, Wang J, Yuan R, Ma Y, Meng Q, Erdos MR, Pestell RG, Yuan F, Auborn KJ, Goldberg ID, Rosen EM. BRCA1 inhibition of estrogen receptor signaling in transfected cells. Science. 1999; 284:1354–1356. PMID: 10334989.
Article20. Hakem R, de la Pompa JL, Elia A, Potter J, Mak TW. Partial rescue of Brca1 (5-6) early embryonic lethality by p53 or p21 null mutation. Nat Genet. 1997; 16:298–302. PMID: 9207798.21. Xu X, Wagner KU, Larson D, Weaver Z, Li C, Ried T, Hennighausen L, Wynshaw-Boris A, Deng CX. Conditional mutation of Brca1 in mammary epithelial cells results in blunted ductal morphogenesis and tumour formation. Nat Genet. 1999; 22:37–43. PMID: 10319859.
Article22. Cortez D, Wang Y, Qin J, Elledge SJ. Requirement of ATM-dependent phosphorylation of brca1 in the DNA damage response to double-strand breaks. Science. 1999; 286:1162–1166. PMID: 10550055.
Article23. Lee JS, Collins KM, Brown AL, Lee CH, Chung JH. hCds1-mediated phosphorylation of BRCA1 regulates the DNA damage response. Nature. 2000; 404:201–204. PMID: 10724175.
Article24. Zhang J, Willers H, Feng Z, Ghosh JC, Kim S, Weaver DT, Chung JH, Powell SN, Xia F. Chk2 phosphorylation of BRCA1 regulates DNA double-strand break repair. Mol Cell Biol. 2004; 24:708–718. PMID: 14701743.
Article25. Starita LM, Parvin JD. The multiple nuclear functions of BRCA1: transcription, ubiquitination and DNA repair. Curr Opin Cell Biol. 2003; 15:345–350. PMID: 12787778.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of DNA Damage Response Proteins and Associations with Clinicopathologic Characteristics in Chinese Familial Breast Cancer Patients with BRCA1/2 Mutations
- Synergistic Effect of Trabectedin and Olaparib Combination Regimen in Breast Cancer Cell Lines
- Ser1778 of 53BP1 Plays a Role in DNA Double-strand Break Repairs
- Mad2B forms a complex with Cdc20, Cdc27, Rev3 and Rev1 in response to cisplatin-induced DNA damage
- Trophoblast Apoptosis and BRCA1 Expression in Fetal Growth Restriction Pregnancy