J Vet Sci.
2015 Jun;16(2):145-150. 10.4142/jvs.2015.16.2.145.
Evaluation of adjuvant effects of fucoidan for improving vaccine efficacy
- Affiliations
-
- 1Laboratory of Veterinary Pharmacology, College of Veterinary Medicine, Jeju National University, Jeju 690-756, Korea. jooh@jejunu.ac.kr
- KMID: 2164522
- DOI: http://doi.org/10.4142/jvs.2015.16.2.145
Abstract
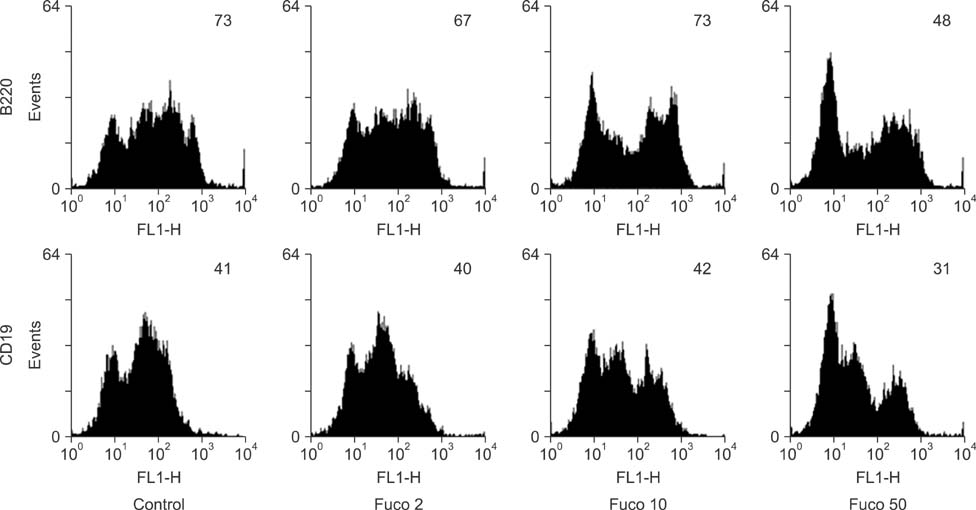
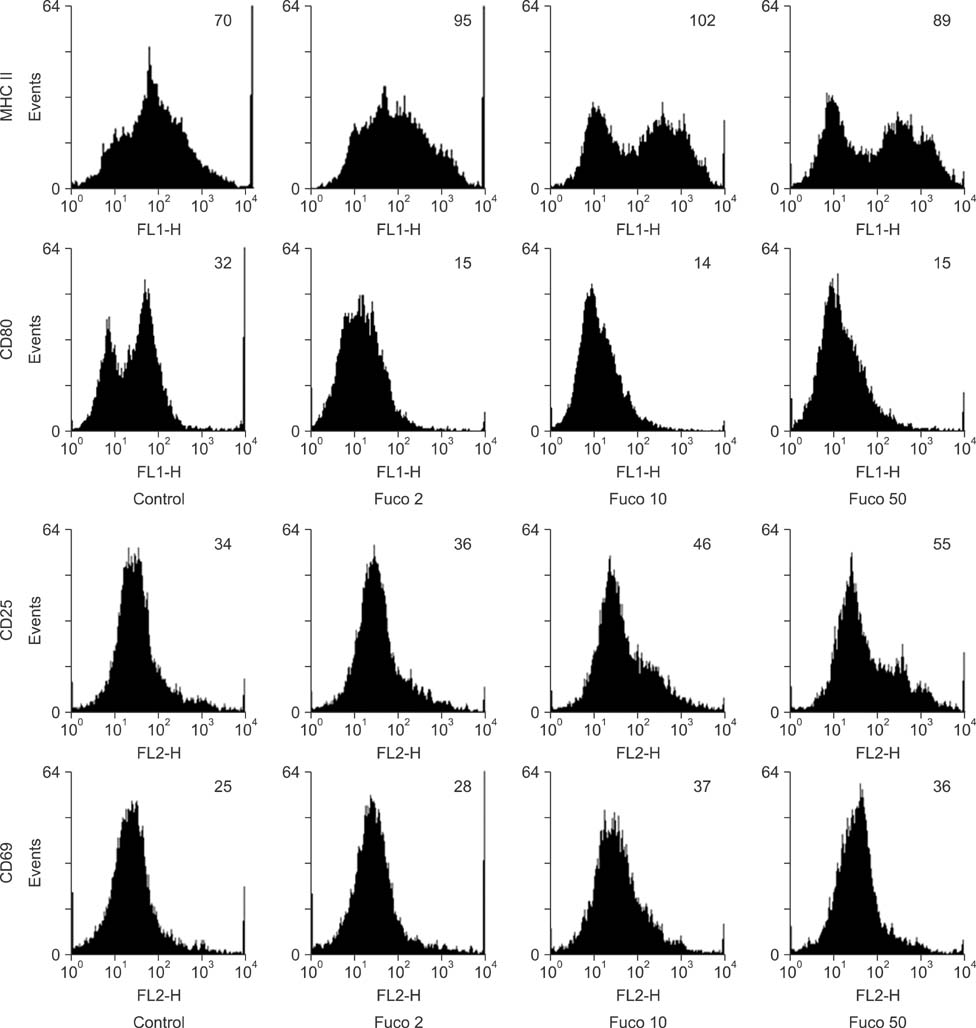
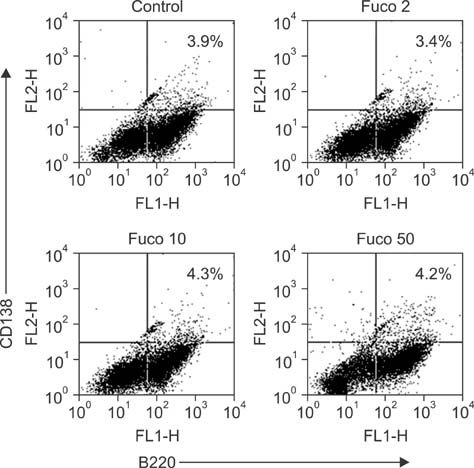
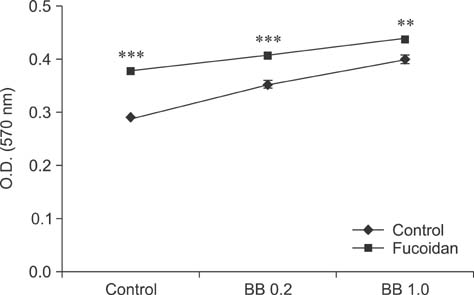
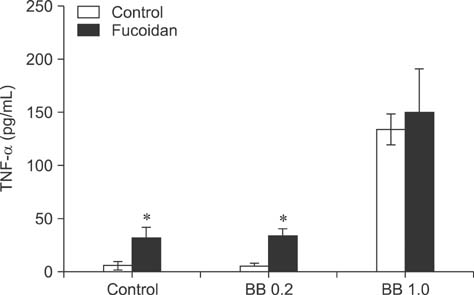
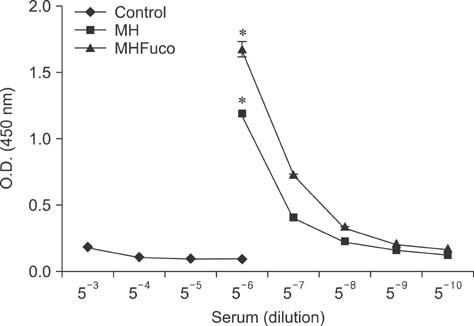
- Fucoidan is a sulfated polysaccharide derived from brown seaweed, including Fucus vesiculosus. This compound is known to have immunostimulatory effects on various types of immune cells including macrophages and dendritic cells. A recent study described the application of fucoidan as a vaccine adjuvant. Vaccination is regarded as the most efficient prophylactic method for preventing harmful or epidemic diseases. To increase vaccine efficacy, effective adjuvants are needed. In the present study, we determined whether fucoidan can function as an adjuvant using vaccine antigens. Flow cytometric analysis revealed that fucoidan increases the expression of the activation markers major histocompatibility complex class II, cluster of differentiation (CD)25, and CD69 in spleen cells. In combination with Bordetella bronchiseptica antigen, fucoidan increased the viability and tumor necrosis factor-alpha production of spleen cells. Furthermore, fucoidan increased the in vivo production of antigen-specific antibodies in mice inoculated with Mycoplasma hyopneumoniae antigen. Overall, this study has provided valuable information about the use of fucoidan as a vaccine adjuvant.
MeSH Terms
-
Adjuvants, Immunologic/pharmacology
Animals
Antigens, Bacterial/*immunology
Bacterial Vaccines/administration & dosage/*immunology
Biomarkers/metabolism
Bordetella bronchiseptica/*immunology
Cells, Cultured
Cytokines/*metabolism
Female
Flow Cytometry
Fucus/*chemistry
Gene Expression Regulation/drug effects
Mice
Mice, Inbred BALB C
Mycoplasma hyopneumoniae/*immunology
Polysaccharides/*pharmacology
Spleen/metabolism
Adjuvants, Immunologic
Antigens, Bacterial
Bacterial Vaccines
Biomarkers
Cytokines
Polysaccharides
Figure
Reference
-
1. Ale MT, Maruyama H, Tamauchi H, Mikkelsen JD, Meyer AS. Fucoidan from Sargassum sp. and Fucus vesiculosus reduces cell viability of lung carcinoma and melanoma cells in vitro and activates natural killer cells in mice in vivo. Int J Biol Macromol. 2011; 49:331–336.
Article2. Amanna IJ, Slifka MK. Contributions of humoral and cellular immunity to vaccine-induced protection in humans. Virology. 2011; 411:206–215.
Article3. Choi EM, Kim AJ, Kim YO, Hwang JK. Immunomodulating activity of arabinogalactan and fucoidan in vitro. J Med Food. 2005; 8:446–453.4. Coffman RL, Sher A, Seder RA. Vaccine adjuvants: putting innate immunity to work. Immunity. 2010; 33:492–503.
Article5. Horiguchi Y. Swine atrophic rhinitis caused by Pasteurella multocida toxin and Bordetella dermonecrotic toxin. Curr Top Microbiol Immunol. 2012; 361:113–129.6. Irhimeh MR, Fitton JH, Lowenthal RM. Fucoidan ingestion increases the expression of CXCR4 on human CD34+ cells. Exp Hematol. 2007; 35:989–994.
Article7. Jefferson T, Rudin M, DiPietrantonj C. Systematic review of the effects of pertussis vaccines in children. Vaccine. 2003; 21:2003–2014.
Article8. Jeong BE, Ko EJ, Joo HG. Cytoprotective effects of fucoidan, an algae-derived polysaccharide on 5-fluorouracil-treated dendritic cells. Food Chem Toxicol. 2012; 50:1480–1484.
Article9. Jin JO, Zhang W, Du JY, Wong KW, Oda T, Yu Q. Fucoidan can function as an adjuvant in vivo to enhance dendritic cell maturation and function and promote antigen-specific T cell immune responses. PLoS One. 2014; 9:e99396.
Article10. Kim JM, Joo HG. Immunostimulatory effects of β-glucan purified from Paenibacillus polymyxa JB115 on mouse splenocytes. Korean J Physiol Pharmacol. 2012; 16:225–230.
Article11. Kim MH, Joo HG. Immunostimulatory effects of fucoidan on bone marrow-derived dendritic cells. Immunol Lett. 2008; 115:138–143.
Article12. Ko EJ, Joo HG. Fucoidan enhances the survival and sustains the number of splenic dendritic cells in mouse endotoxemia. Korean J Physiol Pharmacol. 2011; 15:89–94.
Article13. Kwak JY. Fucoidan as a marine anticancer agent in preclinical development. Mar Drugs. 2014; 12:851–870.
Article14. Maes D, Segales J, Meyns T, Sibila M, Pieters M, Haesebrouck F. Control of Mycoplasma hyopneumoniae infections in pigs. Vet Microbiol. 2008; 126:297–309.15. Oomizu S, Yanase Y, Suzuki H, Kameyoshi Y, Hide M. Fucoidan prevents Cε transcription and NFκB p52 translocation for IgE production in B cells. Biochem Biophys Res Commun. 2006; 350:501–507.
Article16. Pulendran B, Ahmed R. Translating innate immunity into immunological memory: implications for vaccine development. Cell. 2006; 124:849–863.
Article17. Reed SG, Orr MT, Fox CB. Key roles of adjuvants in modern vaccines. Nat Med. 2013; 19:1597–1608.
Article18. Simionatto S, Marchioro SB, Maes D, Dellagostin OA. Mycoplasma hyopneumoniae: from disease to vaccine development. Vet Microbiol. 2013; 165:234–242.19. Yang JW, Yoon SY, Oh SJ, Kim SK, Kang KW. Bifunctional effects of fucoidan on the expression of inducible nitric oxide synthase. Biochem Biophys Res Commun. 2006; 346:345–350.
Article20. Yang M, Ma C, Sun J, Shao Q, Gao W, Zhang Y, Li Z, Xie Q, Dong Z, Qu X. Fucoidan stimulation induces a functional maturation of human monocyte-derived dendritic cells. Int Immunopharmacol. 2008; 8:1754–1760.
Article21. Zhao Z, Wang C, Xue Y, Tang X, Wu B, Cheng X, He Q, Chen H. The occurrence of Bordetella bronchiseptica in pigs with clinical respiratory disease. Vet J. 2011; 188:337–340.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Microbiota Influences Vaccine and Mucosal Adjuvant Efficacy
- Fucoidan Enhances the Survival and Sustains the Number of Splenic Dendritic Cells in Mouse Endotoxemia
- Biological effects of fucoidan isolated from Fucus vesiculosus on thrombosis and vascular cells
- Vaccine adjuvant materials for cancer immunotherapy and control of infectious disease
- Antitumor Effects of Fucoidan on Human Colon Cancer Cells via Activation of Akt Signaling