Korean J Physiol Pharmacol.
2009 Oct;13(5):401-408. 10.4196/kjpp.2009.13.5.401.
Arachidonic Acid Activates K+-Cl--cotransport in HepG2 Human Hepatoblastoma Cells
- Affiliations
-
- 1College of Pharmacy, Duksung Women's University, Seoul 132-714, Korea. yongslee@duksung.ac.kr
- KMID: 1982575
- DOI: http://doi.org/10.4196/kjpp.2009.13.5.401
Abstract
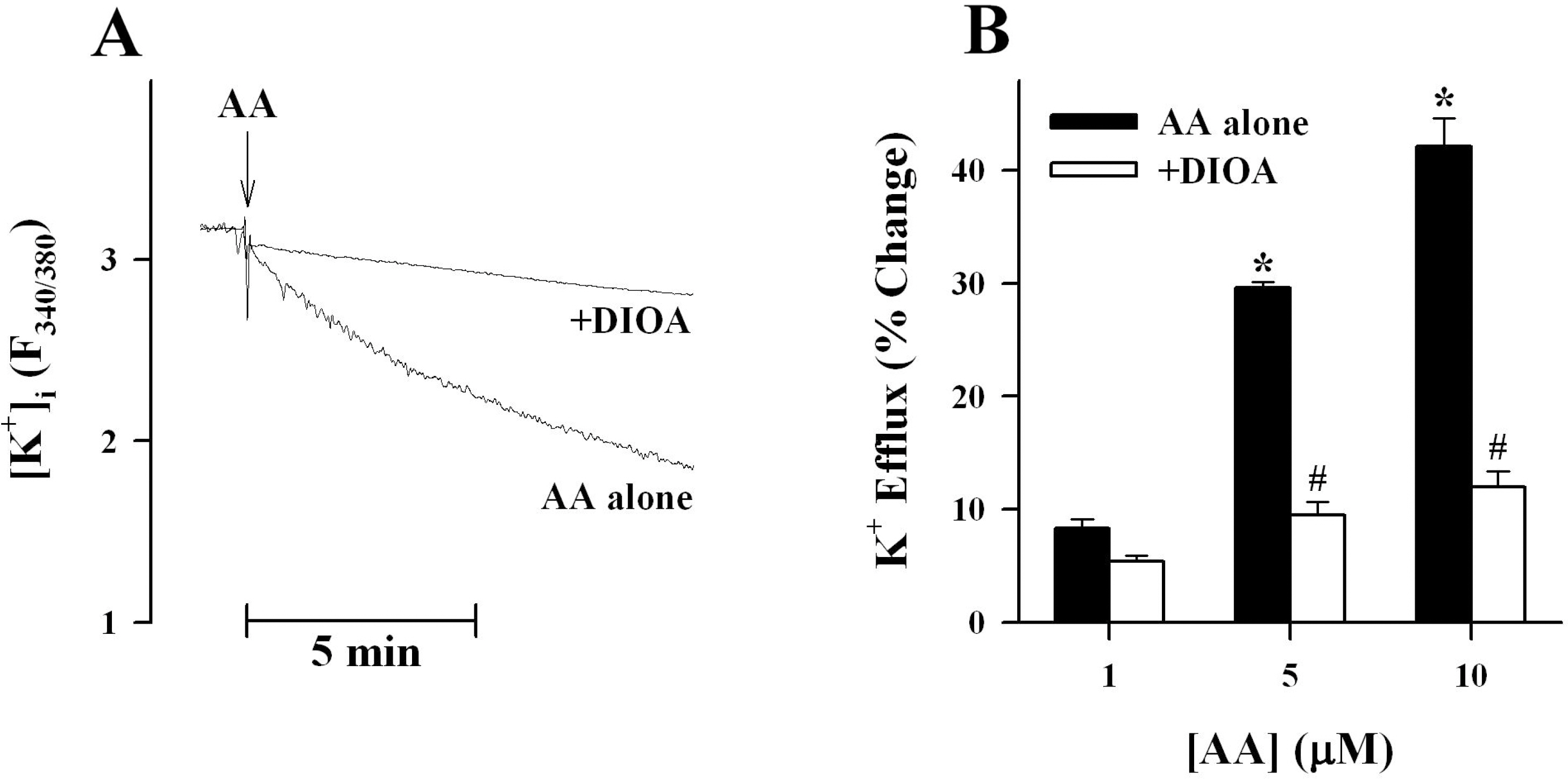
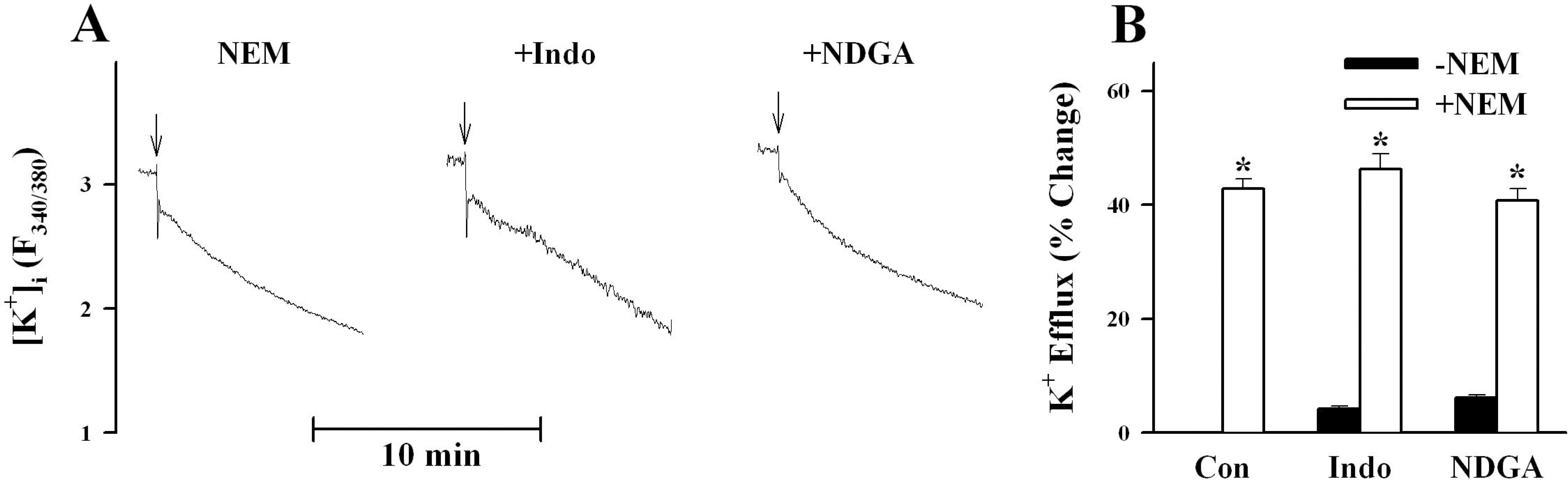
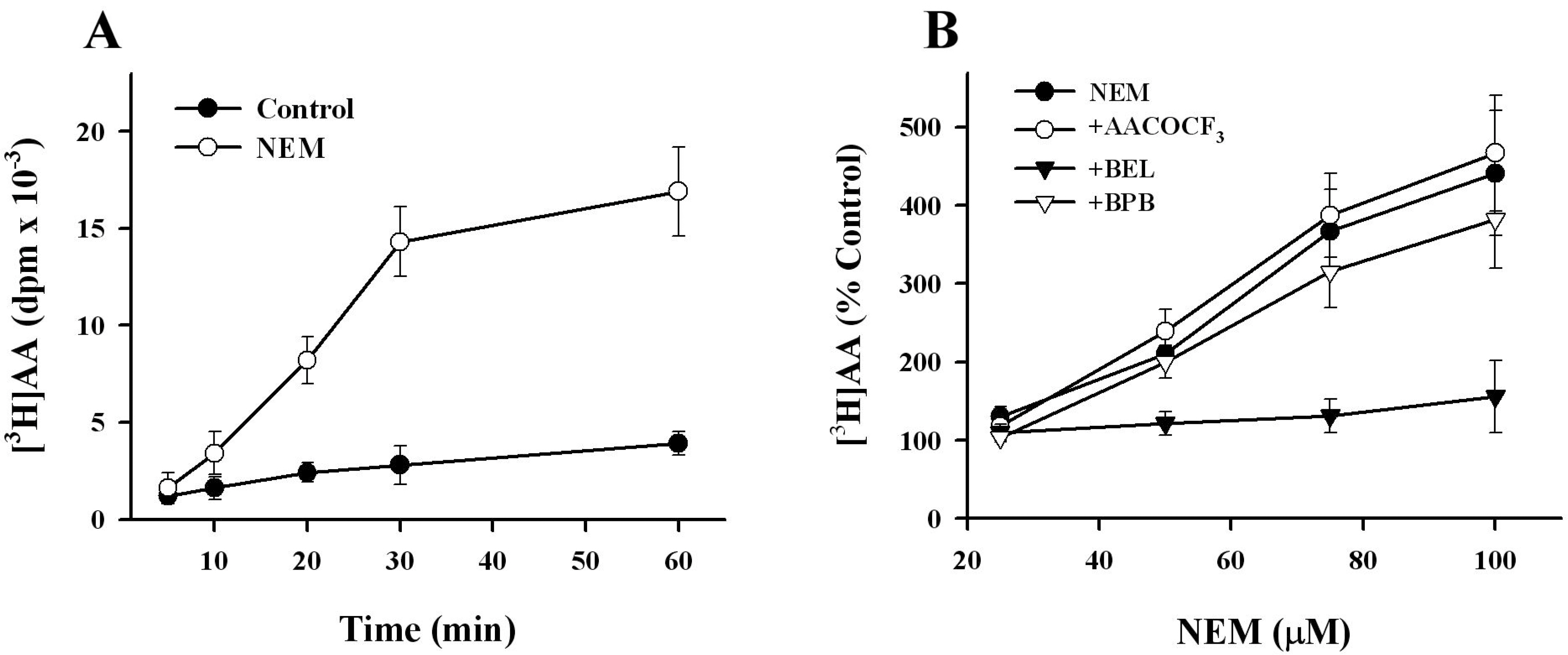
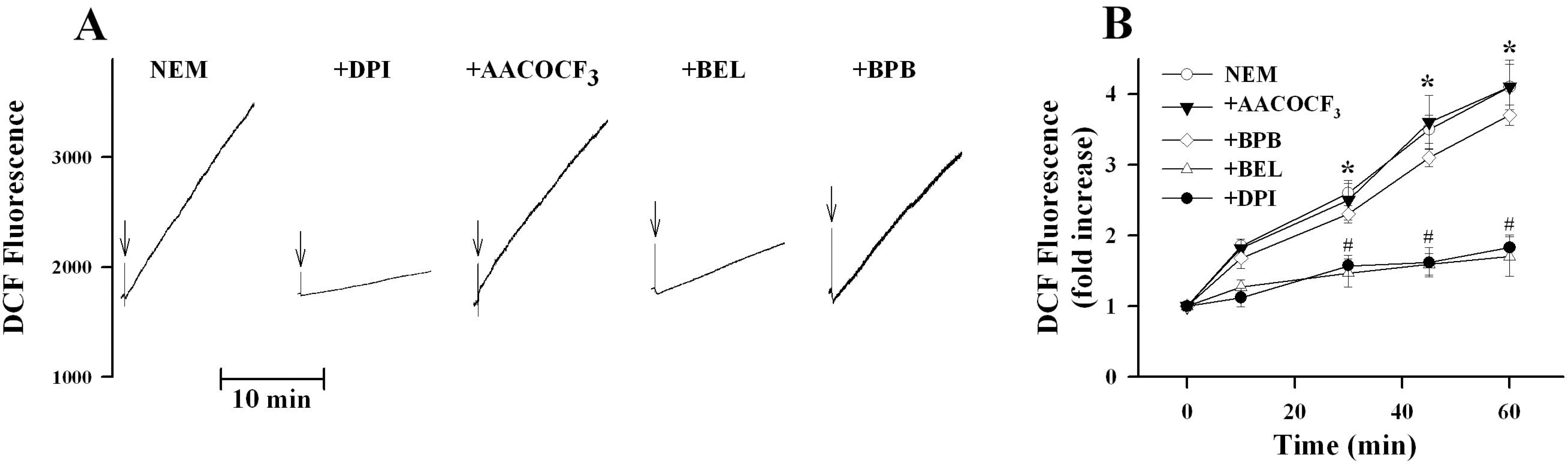
- K+-Cl--cotransport (KCC) has been reported to have various cellular functions, including proliferation and apoptosis of human cancer cells. However, the signal transduction pathways that control the activity of KCC are currently not well understood. In this study we investigated the possible role of phospholipase A2 (PLA2)-arachidonic acid (AA) signal in the regulatory mechanism of KCC activity. Exogenous application of AA significantly induced K+ efflux in a dose-dependent manner, which was completely blocked by R-(+)-[2-n-butyl-6,7-dichloro-2-cyclopentyl-2,3-dihydro-1-oxo-1H-inden-5-yl]oxy]acetic acid (DIOA), a specific KCC inhibitor. N-Ethylmaleimide (NEM), a KCC activator-induced K+ efflux was significantly suppressed by bromoenol lactone (BEL), an inhibitor of the calcium-independent PLA2 (iPLA2), whereas it was not significantly altered by arachidonyl trifluoromethylketone (AACOCF3) and p-bromophenacyl bromide (BPB), inhibitors of the calcium-dependent cytosolic PLA2 (cPLA2) and the secretory PLA2 (sPLA2), respectively. NEM increased AA liberation in a dose- and time-dependent manner, which was markedly prevented only by BEL. In addition, the NEM-induced ROS generation was significantly reduced by DPI and BEL, whereas AACOCF3 and BPB did not have an influence. The NEM-induced KCC activation and ROS production was not significantly affected by treatment with indomethacin (Indo) and nordihydroguaiaretic acid (NDGA), selective inhibitors of cyclooxygenase (COX) and lipoxygenase (LOX), respectively. Treatment with 5,8,11,14-eicosatetraynoic acid (ETYA), a non-metabolizable analogue of AA, markedly produced ROS and activated the KCC. Collectively, these results suggest that iPLA2-AA signal may be essentially involved in the mechanism of ROS-mediated KCC activation in HepG2 cells.
Keyword
MeSH Terms
-
5,8,11,14-Eicosatetraynoic Acid
Acetophenones
Apoptosis
Arachidonic Acid
Arachidonic Acids
Cytosol
Ethylmaleimide
Hep G2 Cells
Hepatoblastoma
Humans
Indomethacin
Lipoxygenase
Naphthalenes
Nordihydroguaiaretic Acid
Phospholipases A2
Prostaglandin-Endoperoxide Synthases
Pyrones
Reactive Oxygen Species
Signal Transduction
5,8,11,14-Eicosatetraynoic Acid
Acetophenones
Arachidonic Acid
Arachidonic Acids
Ethylmaleimide
Indomethacin
Lipoxygenase
Naphthalenes
Nordihydroguaiaretic Acid
Phospholipases A2
Prostaglandin-Endoperoxide Synthases
Pyrones
Reactive Oxygen Species
Figure
Reference
-
References
Adragna NC., Di Fulvio M., Lauf PK. Regulation of K-Cl cotransport: from function to genes. J Membr Biol. 201:109–137. 2004.
ArticleAdragna NC., Ferrell CM., Zhang J., Di Fulvio M., Temprana CF., Sharma A., Fyffe RE., Cool DR., Lauf PK. Signal transduction mechanisms of K+-Cl− cotransport regulation and relationship to disease. Acta Physiol (Oxf). 187:125–139. 2006.Adragna NC., White RE., Orlov SN., Lauf PK. K-Cl cotransport in vascular smooth muscle and erythrocytes: possible implication in vasodilation. Am J Physiol. 278:C381–C390. 2000.Amlal H., Paillard M., Bichara M. Cl−dependent NH4+ transport mechanisms in medullary thick ascending limb cells. Am J Physiol. 267:C1607–C1615. 1994.Bakalova R., Matsura T., Kanno I. The cyclooxygenase inhibitors indomethacin and rofecoxib reduce regional cerebral blood flow evoked by somatosensory stimulation in rats. Exp Biol Med. 227:465–473. 2002.
ArticleBize I., Munoz P., Canessa M., Dunham PB. Stimulation of membrane serine-threonine phosphatase in erythrocytes by hydrogen peroxide and staurosporine. Am J Physiol. 274:C440–C446. 1998.Block K., Ricono JM., Lee DY., Bhandari B., Choudhury GG., Abboud HE., Gorin Y. Arachidonic acid-dependent activation of a p22phox-based NAD(P)H oxidase mediates angiotensin II-induced mesangial cell protein synthesis and fibronectin expression via Akt/PKB. Antioxid Redox Signal. 8:1497–1508. 2006.Choi SH., Langenbach R., Bosetti F. Genetic deletion or pharmacological inhibition of cyclooxygenase-1 attenuate lipopolysaccharide-induced inflammatory response and brain injury. FASEB J. 22:1491–1501. 2008.Cool RH., Merten E., Theiss C., Acker H. Rac1, and not Rac2, is involved in the regulation of the intracellular hydrogen peroxide level in HepG2 cells. Biochem J. 332:5–8. 1998.
ArticleCossins AR., Gibson JS. Volume-sensitive transport systems and volume homeostasis in vertebrate red blood cells. J Exp Biol. 200:343–352. 1997.
ArticleCurnutte JT. Activation of human neutrophil nicotinamide adenine dinucleotide phosphate, reduced (triphosphopyridine nucleotide, reduced) oxidase by arachidonic acid in a cell-free system. J Clin Invest. 75:1740–1743. 1985.
ArticleEhleben W., Porwol T., Fandrey J., Kummer W., Acker H. Cobalt and desferrioxamine reveal crucial members of the oxygen sensing pathway in HepG2 cells. Kidney Int. 51:483–491. 1997.
ArticleEllison DH., Velazquez H., Wright FS. Stimulation of distal potassium secretion by low lumen chloride in the presence of barium. Am J Physiol. 248:F638–F649. 1985.
ArticleFlatman PW., Adragna NC., Lauf PK. Role of protein kinases in regulating sheep erythrocyte K-Cl cotransport. Am J Physiol. 271:C255–C263. 1996.
ArticleGaray RP., Nazaret C., Hannaert PA., Cragoe Jr. EJ. Demonstration of a [K+,Cl−]-cotransport system in human red cells by its sensitivity to [(dihydroindenyl)oxy]alkanoic acids: regulation of cell swelling and distinction from the bumetanide-sensitive [Na+, K+,Cl−]-cotransport system. Mol Pharmacol. 33:696–701. 1988.Gibson JS., Muzyamba MC., Ellory CJ. Effect of phenazine methosulphate on K+ transport in human red cells. Cell Physiol Biochem. 13:329–336. 2003.
ArticleGreger R., Schlatter E. Properties of the basolateral membrane of the cortical thick ascending limb of Henle's loop of rabbit kidney. A model for secondary active chloride transport. Pflugers Archiv. 396:325–334. 1983.Harizi H., Corcuff JB., Gualde N. Arachidonic-acid-derived eicosanoids: roles in biology and immunopathology. Trends Mol Med. 14:461–469. 2008.
ArticleHii CS., Ferrante A. Regulation of the NADPH oxidase activity and anti-microbial function of neutrophils by arachidonic acid. Arch Immunol Ther Exp (Warsz). 55:99–110. 2007.
ArticleHoshino S., Kikkawa S., Takahashi K., Itoh H., Kaziro Y., Kawasaki H., Suzuki K., Katada T., Ui M. Identification of sites for alkylation by N-ethylmaleimide and pertussis toxin-catalyzed ADP-ribosylation on GTP-binding proteins. FEBS Lett. 276:227–231. 1990.Hsu YM., Chen YF., Chou CY., Tang MJ., Chen JH., Wilkins RJ., Ellory JC., Shen MR. KCl cotransporter-3 down-regulates E-cadherin/beta-catenin complex to promote epithelial-mesenchymal transition. Cancer Res. 67:11064–11073. 2007a.Hsu YM., Chou CY., Chen HH., Lee WY., Chen YF., Lin PW., Alper SL., Ellory JC., Shen MR. IGF-1 upregulates electroneutral K-Cl cotransporter KCC3 and KCC4 which are differentially required for breast cancer cell proliferation and invasiveness. J Cell Physiol. 210:626–636. 2007b.
ArticleJoiner CH. Cation transport and volume regulation in sickle red blood cells. Am J Physiol. 264:C251–C270. 1993.
ArticleKaji DM., Tsukitani Y. Role of protein phosphatase in activation of KCl cotransport in human erythrocytes. Am J Physiol. 260:C176–C180. 1991.
ArticleKehl SJ. Eicosatetraynoic acid (ETYA), a non-metabolizable analogue of arachidonic acid, blocks the fast-inactivating potassium current of rat pituitary melanotrophs. Can J Physiol Pharmacol. 79:338–345. 2001.
ArticleKim C., Dinauer MC. Impaired NADPH oxidase activity in Rac2-deficient murine neutrophils does not result from defective translocation of p47phox and p67phox and can be rescued by exogenous arachidonic acid. J Biol Chem. 79:223–234. 2006.Kim C., Kim JY., Kim JH. Cytosolic phospholipase A2, lipoxygenase metabolites, and reactive oxygen species. BMB Rep. 41:555–559. 2008.Kim D., Nakamura A., Okamoto T., Komatsu N., Oda T., Iida T., Ishimatsu A., Muramatsu T. Mechanism of superoxide anion generation in the toxic red tide phytoplankton Chattonella marina: possible involvement of NAD(P)H oxidase. Biochim Biophys Acta. 1524:220–227. 2000.
ArticleKim JA., Kang YS., Lee YS. Involvement of K+-Cl−cotransport in the apoptosis induced by N-ethylmaleimide in HepG2 human hepatoblastoma cells. Eur J Pharmacol. 418:1–5. 2001.Kim JA., Lee YS. Role of reactive oxygen species generated by NADPH oxidase in the mechanism of activation of K+-Cl−cotransport by N-ethylmaleimide in HepG2 human hepatoma cells. Free Radic Res. 35:43–53. 2001.LaBel CP., Ischiopoulos H., Bondy SC. Evaluation of the probe 2′,7′-dichlorofluorescin as indicator of reactive oxygen species formation and oxidative stress. Chem Res Toxicol. 5:227–231. 1992.Lauf PK., Bauer J., Adragna NC., Fujise H., Zade-Oppen AMM., Ryu KH., Delpire E. Erythrocyte K-Cl cotransport: properties and regulation. Am J Physiol. 263:C917–C932. 1992.
ArticleLeoncini G., Signorello MG. N-ethylmaleimide inhibition of thrombin-induced platelet aggregation. Biochem Pharmacol. 58:1293–1299. 1999a.
ArticleLeoncini G., Signorello MG. N-ethylmaleimide-stimulated arachidonic acid release in human platelets. Biochem Pharmacol. 57:785–791. 1999b.
ArticleLeslie CC. Regulation of arachidonic acid availability for eicosanoid production. Biochem Cell Biol. 82:1–17. 2004.
ArticleLuchtefeld M., Drexler H., Schieffer B. 5-Lipoxygenase is involved in the angiotensin II-induced NAD(P)H-oxidase activation. Biochem Biophys Res Commun. 308:668–672. 2003.
ArticleMinta A., Tsien RY. Fluorescent indicators for cytosolic sodium. J Biol Chem. 264:19449–19457. 1989.
ArticleMuzyamba MC., Speake PF., Gibson JS. Oxidants and regulation of K+-Cl− cotransport in equine red blood cells. Am J Physiol. 271:C981–C989. 2000.Narendra Sharath Chandra JN., Ponnappa KC., Sadashiva CT., Priya BS., Nanda BL., Gowda TV., Vishwanath BS., Rangappa KS. Chemistry and structural evaluation of different phospholipase A2 inhibitors in arachidonic acid pathway mediated inflammation and snake venom toxicity. Curr Top Med Chem. 7:787–800. 2007.Olivieri O., Vitoux D., Galacteros F., Bachir D., Blouquit Y., Beuzard Y., Brugnara C. Hemoglobin variants and activity of (K+Cl−) cotransport system in human erythrocytes. Blood. 79:793–797. 1992.Perry PB., O'Neill WC. Swelling-activated K fluxes in vascular endothelial cells: volume regulation via K-Cl cotransport and K channels. Am J Physiol. 265:C763–C769. 1993.
ArticleRamanadham S., Song H., Bao S., Hsu FF., Zhang S., Ma Z., Jin C., Turk J. Islet complex lipids: involvement in the actions of group VIA calcium-independent phospholipase A2 in beta-cells. Diabetes. 53(Suppl 1):S179–185. 2004.Rivera C., Voipio J., Payne JA., Ruusuvuori E., Latineen H., Lamsa K., Pirvola U., Saarma M., Kaila K. The K+/Cl− co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature. 397:251–255. 1999.Shen HM., Shi CY., Ong CN. Detection of elevated reactive oxygen species level in cultured rat hepatocytes treated with aflatoxin B1. Free Rad Biol Med. 21:139–146. 1996.
ArticleShen MR., Lin AC., Hsu YM., Chang TJ., Tang MJ., Alper SL., Ellory JC., Chou CY. Insulin-like growth factor 1 stimulates KCl cotransport, which is necessary for invasion and proliferation of cervical cancer and ovarian cancer cells. J Biol Chem. 279:40017–40025. 2004.
ArticleShiose A., Sumimoto H. Arachidonic acid and phosphorylation synergistically induce a conformational change of p47phox to activate the phagocyte NADPH oxidase. J Biol Chem. 275:13793–13801. 2000.Starke LC., Jennings ML. K-Cl cotransport in rabbit red cells: further evidence for regulation by protein phosphatase type 1. Am J Physiol. 264:C118–C124. 1993.
ArticleStuart J., Ellory JC. Rheological consequences of erythrocyte dehydration. Br J Haematol. 69:1–4. 1988.
ArticleTang DG., Chen YQ., Honn KV. Arachidonate lipoxygenases as essential regulators of cell survival and apoptosis. Proc Natl Acad Sci USA. 93:5241–5246. 1996.
ArticleVan Der Zee L., Nelemans A., Den Hertog A. Arachidonic acid is functioning as a second messenger in activating the Ca2+ entry process on H1-histaminoceptor stimulation in DDT1 MF-2 cells. Biochem J. 305:859–864. 1995.Waite M. Phospholipases. Vance DE, Vance JE, editors. ed,. Biochemistry of Lipids, Lipoproteins and Membranes. 1st ed.Elsevier Science Publishing Co;New York: p. p. 211–236. 1996.
ArticleWeaver YR., Cossins AR. Protein tyrosine phosphorylation and the regulation of KCl cotransport in trout erythrocytes. Pflugers Archiv. 432:727–734. 1996.
ArticleWeil-Maslansky E., Gutman Y., Sasson S. Insulin activates furosemide-sensitive K+ and Cl− uptake system in BC3H1 cells. Am J Physiol. 267:C932–C939. 1994.Wolf MJ., Wang J., Turk J., Gross RW. Depletion of intracellular calcium stores activates smooth muscle cell calcium-independent phospholipase A2. A novel mechanism underlying arachidonic acid mobilization. J Biol Chem. 272:1522–1526. 1997.Wong RK., Pettit AI., Quinn PA., Jennings SC., Davies JE., Ng LL. Advanced glycation end products stimulate an enhanced neutrophil respiratory burst mediated through the activation of cytosolic phospholipase A2 and generation of arachidonic acid. Circulation. 108:1858–1864. 2003.Wurster S., Nakov R., Allgaier C., Hertting G. Involvement of N-ethylmaleimide-sensitive G proteins in the modulation of evoked [3H]noradrenaline release from rabbit hippocampus synaptosomes. Neurochem Int. 17:149–155. 1990.Yan GX., Chen J., Yamada KA., Kleber AG., Corr PB. Contribution of shrinkage of extracellular space to extracellular K+ accumulation in myocardial ischaemia of the rabbit. J Physiol (London). 490:215–228. 1996.Yellaturu CR., Rao GN. A requirement for calcium-independent phospholipase A2 in thrombin-induced arachidonic acid release and growth in vascular smooth muscle cells. J Biol Chem. 278:43831–43837. 2003.Zhang W., Wang Y., Chen CW., Xing K., Vivekanandan S., Lou MF. The positive feedback role of arachidonic acid in the platelet-derived growth factor-induced signaling in lens epithelial cells. Mol Vis. 12:821–831. 2006.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Role of Ca2+ influx in the tert-butyl hydroperoxide-induced apoptosis of HepG2 human hepatoblastoma cellse
- Effect of CLA (Conjugated Linoleic Acid) on the Anti-Atherosclerotic Factors in Human Hepatoma HepG2 Cells
- Changes of arachidonic acid metabolites in silica-expossed alveolar macrophage of rats
- Profile of arachidonic acid metabolites and platelet-activatingfactors in human middle ear effusion
- Expression and Intracellular Localization of Hepatitis C Viral Core Protein in Human Hepatoma Cell Line Transfected with Viral cDNA