J Korean Med Sci.
2010 Mar;25(3):440-448. 10.3346/jkms.2010.25.3.440.
Neurotoxicity Screening in a Multipotent Neural Stem Cell Line Established from the Mouse Brain
- Affiliations
-
- 1Department of Pathology, Chonnam National University Medical School, Gwangju, Korea. mclee@jnu.ac.kr
- 2Department of Forensic Medicine and Pathology, Chonnam National University Medical School, Gwangju, Korea.
- 3Center for Biomedical Human Resources (BK 21), Chonnam National University Medical School, Gwangju, Korea.
- 4Department of Pathology, Seonam University College of Medicine, Namwon, Korea.
- 5Department of Ophthalmology, Chonnam National University Medical School, Gwangju, Korea.
- 6Department of Physiology, Chonnam National University Medical School, Gwangju, Korea.
- 7Department of Neurology, Chonnam National University Medical School, Gwangju, Korea.
- 8Department of Pediatrics, Chonnam National University Medical School, Gwangju, Korea.
- 9Department of Neurobiology, Chung-Ang University Medical Center, Seoul, Korea.
- 10Department of Medicine, University of British Columbia, Vancouver, BC, Canada.
- KMID: 1778041
- DOI: http://doi.org/10.3346/jkms.2010.25.3.440
Abstract
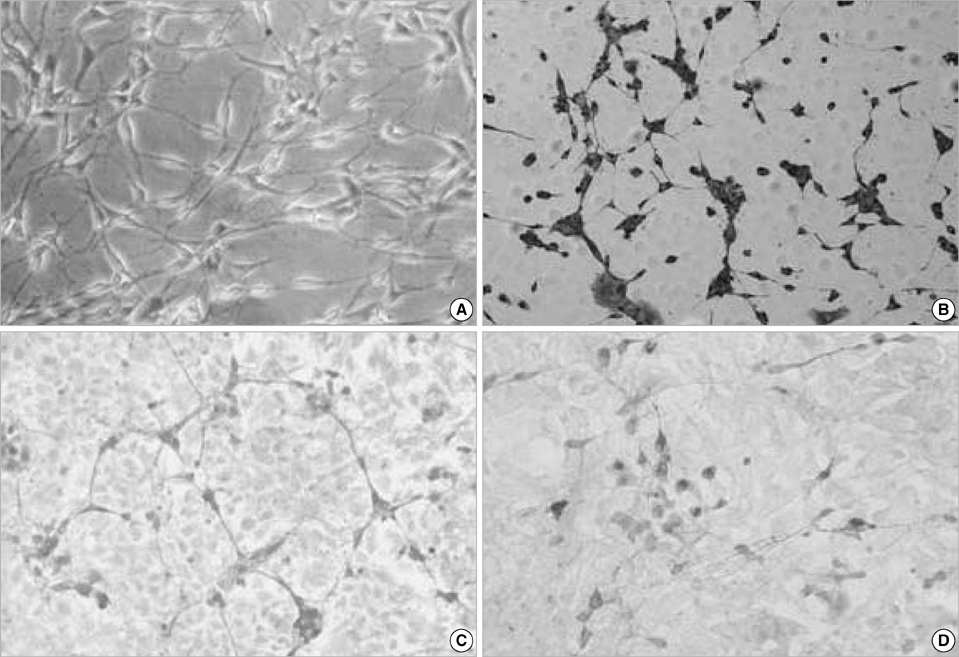
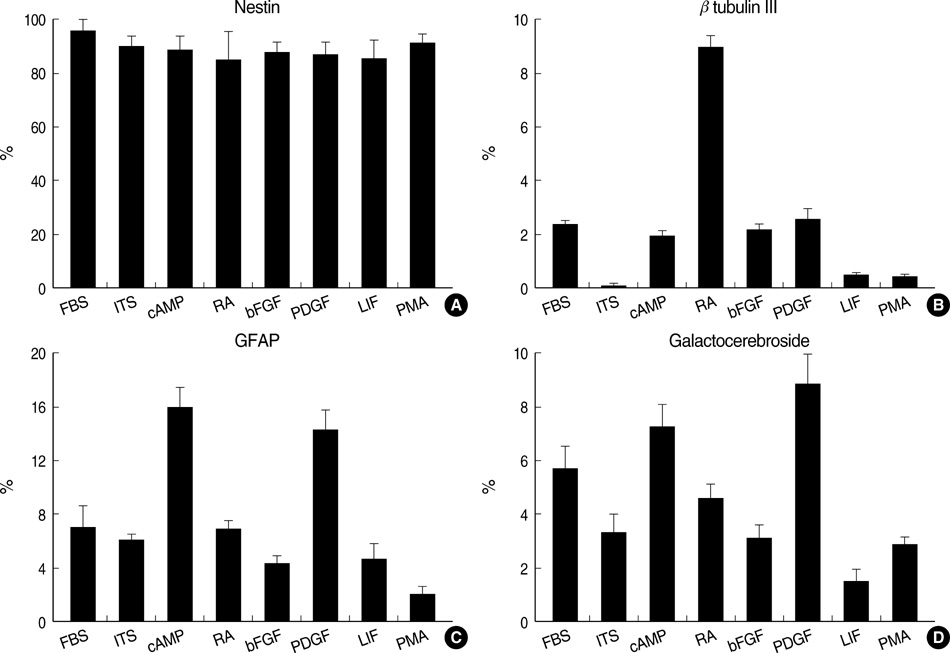
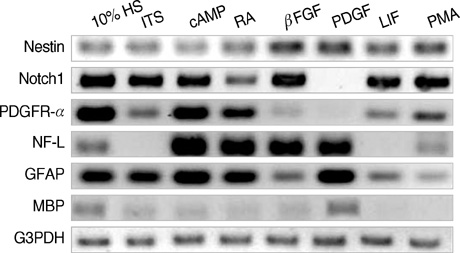
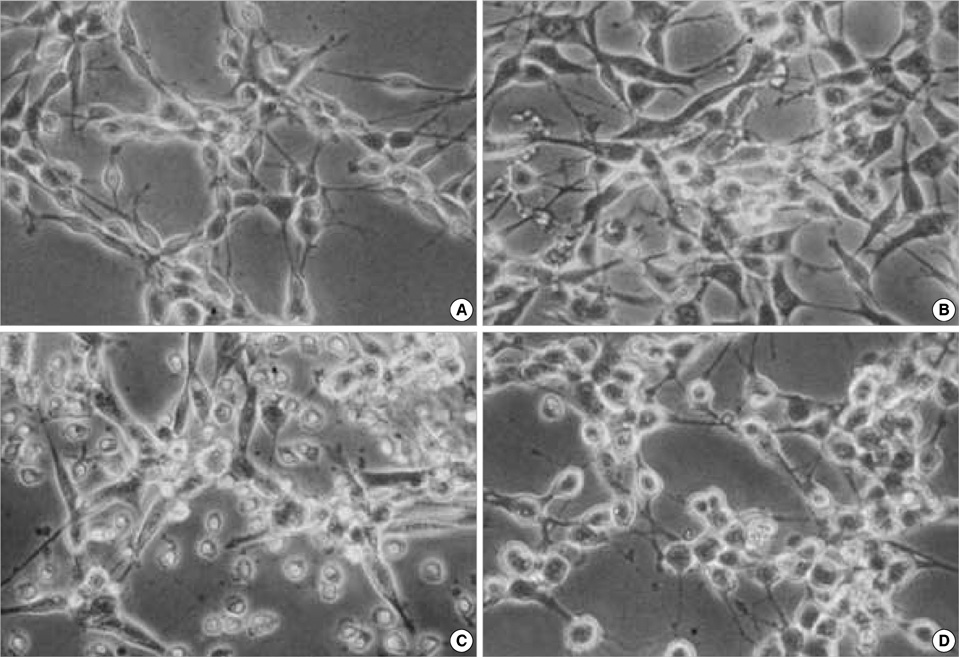
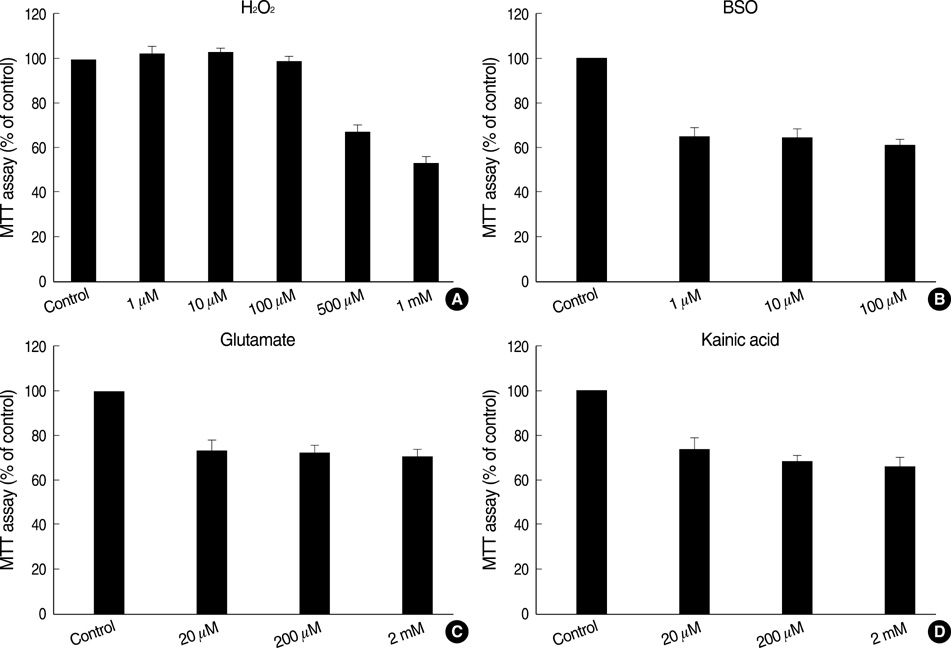
- Neural stem cells (NSCs) have mainly been applied to neurodegeneration in some medically intractable neurologic diseases. In this study, we established a novel NSC line and investigated the cytotoxic responses of NSCs to exogenous neurotoxicants, glutamates and reactive oxygen species (ROS). A multipotent NSC line, B2A1 cells, was established from long-term primary cultures of oligodendrocyte-enriched cells from an adult BALB/c mouse brain. B2A1 cells could be differentiated into neuronal, astrocytic and oligodendroglial lineages. The cells also expressed genotypic mRNA messages for both neural progenitor cells and differentiated neuronoglial cells. B2A1 cells treated with hydrogen peroxide and L-buthionine-(S,R)-sulfoximine underwent 30-40% cell death, while B2A1 cells treated with glutamate and kainate showed 25-35% cell death. Cytopathologic changes consisting of swollen cell bodies, loss of cytoplasmic processes, and nuclear chromatin disintegration, developed after exposure to both ROS and excitotoxic chemicals. These results suggest that B2A1 cells may be useful in the study of NSC biology and may constitute an effective neurotoxicity screening system for ROS and excitotoxic chemicals.
Keyword
MeSH Terms
-
Animals
Brain/*cytology
Buthionine Sulfoximine/pharmacology
Cell Differentiation
Cell Line
Cell Lineage
Cytokines/pharmacology
Enzyme Inhibitors/pharmacology
Excitatory Amino Acid Agonists/pharmacology
Glutamic Acid/pharmacology
Humans
Hydrogen Peroxide/pharmacology
Intercellular Signaling Peptides and Proteins/pharmacology
Kainic Acid/pharmacology
Mice
Mice, Inbred BALB C
Multipotent Stem Cells/cytology/*drug effects/physiology
Neuroglia/cytology/drug effects/physiology
Neurons/cytology/*drug effects/physiology
Neurotoxins/*pharmacology
Oxidants/pharmacology
Phenotype
Reactive Oxygen Species/metabolism
Cytokines
Enzyme Inhibitors
Excitatory Amino Acid Agonists
Intercellular Signaling Peptides and Proteins
Neurotoxins
Oxidants
Reactive Oxygen Species
Kainic Acid
Buthionine Sulfoximine
Glutamic Acid
Hydrogen Peroxide
Figure
Reference
-
1. Metcalf D. Concise review: hematopoietic stem cells and tissue stem cells: current concepts and unanswered questions. Stem Cells. 2007. 25:2390–2395.
Article2. Flax JD, Aurora S, Yang C, Simonin C, Wills AM, Billinghurst LL, Jendoubi M, Sidman RL, Wolfe JH, Kim SU, Snyder EY. Engraftable human neural stem cells respond to developmental cues, replace neurons, and express foreign genes. Nat Biotechnol. 1998. 16:1033–1039.3. Mimeault M, Batra SK. Recent progress on tissue-resident adult stem cell biology and their therapeutic implications. Stem Cell Rev. 2008. 4:27–49.
Article4. Davis AA, Temple S. A self-renewing multipotential stem cell in embryonic rat cerebral cortex. Nature. 1994. 372:263–266.
Article5. Kukekov VG, Laywell ED, Suslov O, Davies K, Scheffler B, Thomas LB, O'Brien TF, Kusakabe M, Steindler DA. Multipotent stem/progenitor cells with similar properties arise from two neurogenic regions of adult human brain. Exp Neurol. 1999. 156:333–344.
Article6. Anderson DJ. Stem cells and pattern formation in the nervous system: the possible versus the actual. Neuron. 2001. 30:19–35.7. Ourednik J, Ourednik V, Lynch WP, Schachner M, Snyder EY. Neural stem cells display an inherent mechanism for rescuing dysfunctional neurons. Nat Biotechnol. 2002. 20:1103–1110.
Article8. Ryu JK, Kim J, Cho SJ, Hatori K, Nagai A, Choi HB, Lee MC, Mc-Larnon JG, Kim SU. Proactive transplantation of human neural stem cells prevents degeneration of striatal neurons in a rat model of Huntington disease. Neurobiol Dis. 2004. 16:68–77.
Article9. Davidovics Z, DiCicco-Bloom E. Moderate lead exposure elicits neurotrophic effects in cerebral cortical precursor cells in culture. J Neurosci Res. 2005. 80:817–825.
Article10. Breier JM, Radio NM, Mundy WR, Shafer TJ. Development of a high-throughput screening assay for chemical effects on proliferation and viability of immortalized human neural progenitor cells. Toxicol Sci. 2008. 105:119–133.
Article11. Satoh J, Tabira T, Kim SU. Rapidly proliferating glial cells isolated from adult mouse brain have a differentiative capacity in response to cyclic AMP. Neurosci Res. 1994. 20:175–184.
Article12. Rey JA, Bello MJ, De Campos JM, Kusak ME, Moreno S. Cytogenetic analysis in human neurinomas. Cancer Genet Cytogenet. 1987. 28:187–188.
Article13. Kim SU, Stern J, Kim MW, Pleasure DE. Culture of purified rat astrocytes in serum-free medium supplemented with mitogen. Brain Res. 1983. 274:79–86.
Article14. Cai J, Wu Y, Mirua T, Pierce JL, Lucero MT, Albertine KH, Spangrude GJ, Rao MS. Properties of a fetal multipotent neural stem cell (NEP cell). Dev Biol. 2002. 251:221–240.
Article15. Nagai A, Suzuki Y, Baek SY, Lee KS, Lee MC, McLarnon JG, Kim SU. Generation and characterization of human hybrid neurons produced between embryonic CNS neurons and neuroblastoma cells. Neurobiol Dis. 2002. 11:184–198.
Article16. Lee MC, Chung YT, Lee JH, Jung JJ, Kim HS, Kim SU. Antioxidant effect of melatonin in human retinal neuron cultures. Exp Neurol. 2001. 172:407–415.
Article17. Bignami A, Raju T, Dahl D. Localization of vimentin, the nonspecific intermediate filament protein, in embryonal glia and in early differentiating neurons. In vivo and in vitro immunofluorescence study of the rat embryo with vimentin and neurofilament antisera. Dev Biol. 1982. 91:286–295.18. Lendahl U, Zimmerman LB, McKay RD. CNS stem cells express a new class of intermediate filament protein. Cell. 1990. 60:585–595.
Article19. Zhong W, Jiang MM, Weinmaster G, Jan LY, Jan YN. Differential expression of mammalian Numb, Numblike and Notch1 suggests distinct roles during mouse cortical neurogenesis. Development. 1997. 124:1887–1897.
Article20. Andrae J, Hansson I, Afink GB, Nister M. Platelet-derived growth factor receptor-alpha in ventricular zone cells and in developing neurons. Mol Cell Neurosci. 2001. 17:1001–1013.21. Kageyama R, Ohtsuka T. The Notch-Hes pathway in mammalian neural development. Cell Res. 1999. 9:179–188.
Article22. Yang X, Handler M, Shen J. Role of presenilin-1 in murine neural development. Ann N Y Acad Sci. 2000. 920:165–170.
Article23. Goodell MA, Brose K, Paradis G, Conner AS, Mulligan RC. Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J Exp Med. 1996. 183:1797–1806.
Article24. Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999. 284:143–147.
Article25. Martinez-Serrano A, Lundberg C, Horellou P, Fischer W, Bentlage C, Campbell K, McKay RD, Mallet J, Bjorklund A. CNS-derived neural progenitor cells for gene transfer of nerve growth factor to the adult rat brain: complete rescue of axotomized cholinergic neurons after transplantation into the septum. J Neurosci. 1995. 15:5668–5680.
Article26. Rietze RL, Valcanis H, Brooker GF, Thomas T, Voss AK, Bartlett PF. Purification of a pluripotent neural stem cell from the adult mouse brain. Nature. 2001. 412:736–739.
Article27. Ohkawara B, Okuno M, Ishii T, Horiuchi M, Tomooka Y. Characterization of a multipotent neural progenitor cell line cloned from an adult p53-/- mouse cerebellum. Brain Res. 2003. 959:11–19.
Article28. Coyle JT, Puttfarcken P. Oxidative stress, glutamate, and neurodegenerative disorders. Science. 1993. 262:689–695.
Article29. Shivakumar BR, Kolluri SV, Ravindranath V. Glutathione and protein thiol homeostasis in brain during reperfusion after cerebral ischemia. J Pharmacol Exp Ther. 1995. 274:1167–1173.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Establishment of a Multipotent Neural Stem Cell Line from the Adult Mouse Cerebrum
- Establishment and Characterization of Multipotent Germ Line Stem Cells (MGSCs) from Neonatal Mouse Testis
- Establishment of a Method for Cryopreservation of Neural Stem Cells
- The Differential Developmental Neurotoxicity of Valproic Acid on Anterior and Posterior Neural Induction of Human Pluripotent Stem Cells
- Expression of mRNAs for Neurotrophic Factors in Human Neural Stem Cells Derived from Fetal Telencephalon