Korean J Physiol Pharmacol.
2013 Jun;17(3):181-187. 10.4196/kjpp.2013.17.3.181.
Contradictory Effects of Superoxide and Hydrogen Peroxide on KCa3.1 in Human Endothelial Cells
- Affiliations
-
- 1Department of Physiology and Medical Research Institute, School of Medicine, Ewha Womans University, Seoul 158-710, Korea. shush@ewha.ac.kr
- KMID: 1429296
- DOI: http://doi.org/10.4196/kjpp.2013.17.3.181
Abstract
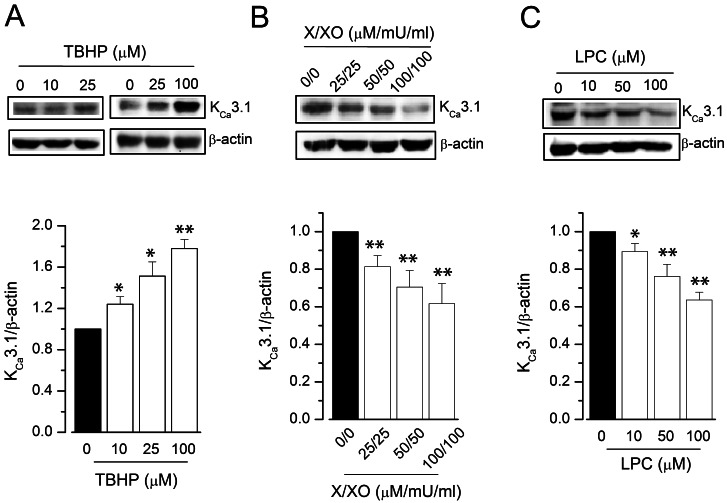
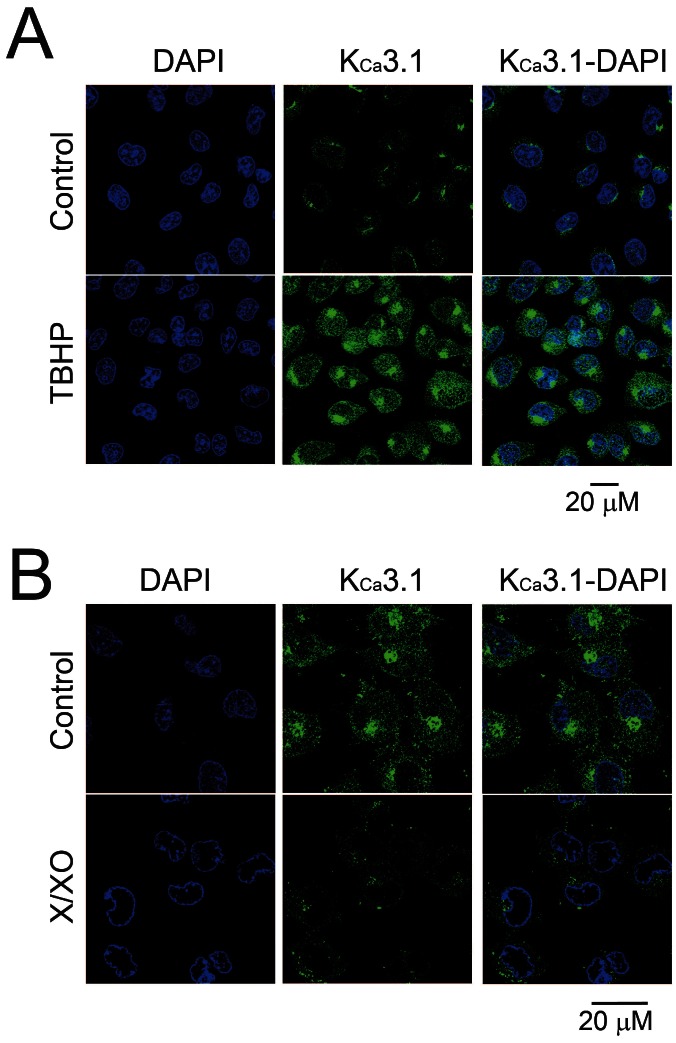
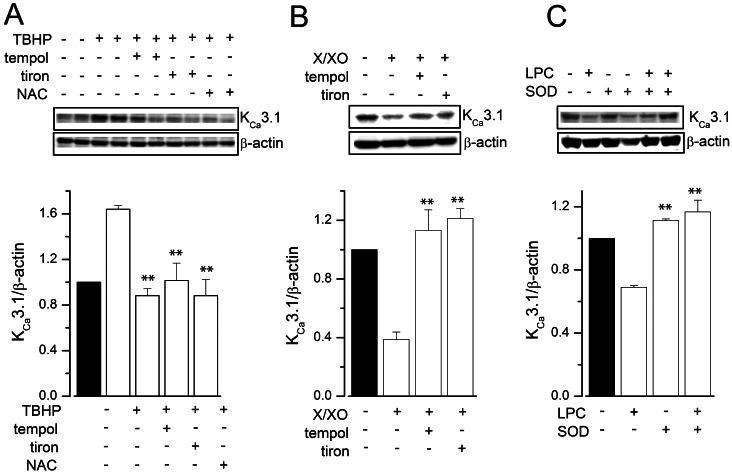
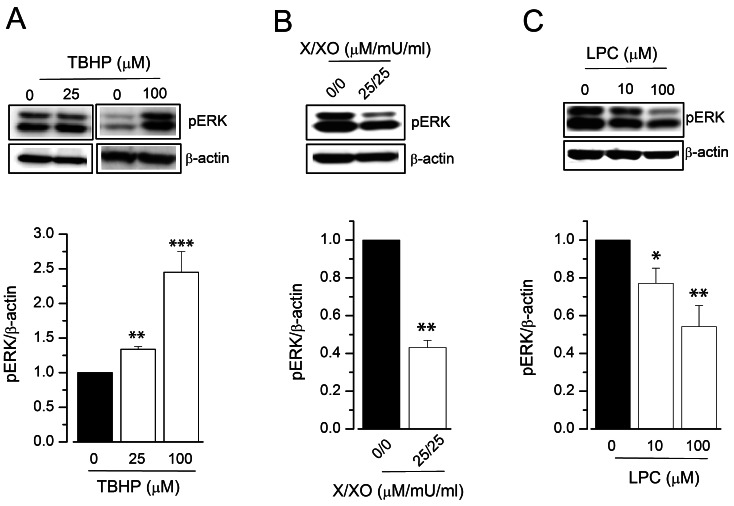
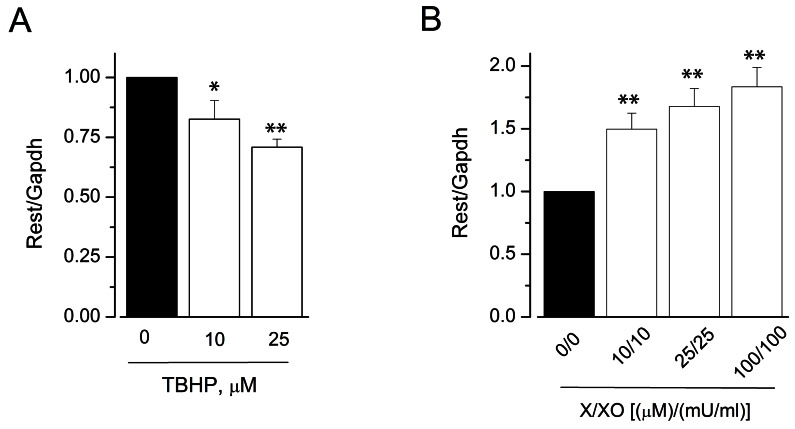
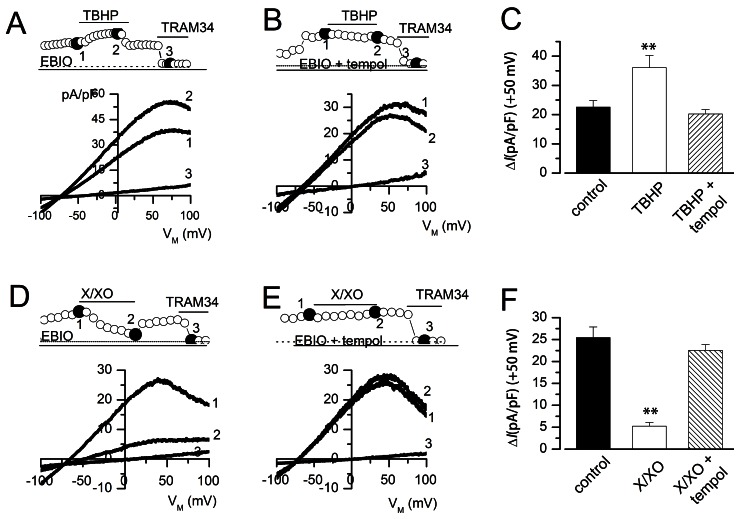
- Reactive oxygen species (ROS) are generated in various cells, including vascular smooth muscle and endothelial cells, and regulate ion channel functions. KCa3.1 plays an important role in endothelial functions. However, the effects of superoxide and hydrogen peroxide radicals on the expression of this ion channel in the endothelium remain unclear. In this study, we examined the effects of ROS donors on KCa3.1 expression and the K+ current in primary cultured human umbilical vein endothelial cells (HUVECs). The hydrogen peroxide donor, tert-butyl hydroperoxide (TBHP), upregulated KCa3.1 expression, while the superoxide donors, xanthine/xanthine oxidase mixture (X/XO) and lysopho-sphatidylcholine (LPC), downregulated its expression, in a concentration-dependent manner. These ROS donor effects were prevented by antioxidants or superoxide dismustase. Phosphorylated extracellular signal-regulated kinase (pERK) was upregulated by TBHP and downregulated by X/XO. In addition, repressor element-1-silencing transcription factor (REST) was downregulated by TBHP, and upregulated by X/XO. Furthermore, KCa3.1 current, which was activated by clamping cells with 1 microM Ca2+ and applying the KCa3.1 activator 1-ethyl-2-benzimidazolinone, was further augmented by TBHP, and inhibited by X/XO. These effects were prevented by antioxidants. The results suggest that hydrogen peroxide increases KCa3.1 expression by upregulating pERK and downregulating REST, and augments the K+ current. On the other hand, superoxide reduces KCa3.1 expression by downregulating pERK and upregulating REST, and inhibits the K+ current. ROS thereby play a key role in both physiological and pathological processes in endothelial cells by regulating KCa3.1 and endothelial function.
MeSH Terms
-
Antioxidants
Benzimidazoles
Constriction
Endothelial Cells
Endothelium
Hand
Human Umbilical Vein Endothelial Cells
Humans
Hydrogen
Hydrogen Peroxide
Ion Channels
Muscle, Smooth, Vascular
Oxidoreductases
Pathologic Processes
Phosphotransferases
Reactive Oxygen Species
Superoxides
tert-Butylhydroperoxide
Tissue Donors
Transcription Factors
Antioxidants
Benzimidazoles
Hydrogen
Hydrogen Peroxide
Ion Channels
Oxidoreductases
Phosphotransferases
Reactive Oxygen Species
Superoxides
Transcription Factors
tert-Butylhydroperoxide
Figure
Cited by 1 articles
-
Ameliorative Effect of a Selective Endothelin ETA Receptor Antagonist in Rat Model of L-Methionine-induced Vascular Dementia
Gautamjeet S Mangat, Amteshwar S Jaggi, Nirmal Singh
Korean J Physiol Pharmacol. 2014;18(3):201-209. doi: 10.4196/kjpp.2014.18.3.201.
Reference
-
1. Félétou M, Vanhoutte PM. Endothelial dysfunction: a multifaceted disorder (The Wiggers Award Lecture). Am J Physiol Heart Circ Physiol. 2006; 291:H985–H1002. PMID: 16632549.
Article2. Li JM, Shah AM. Endothelial cell superoxide generation: regulation and relevance for cardiovascular pathophysiology. Am J Physiol Regul Integr Comp Physiol. 2004; 287:R1014–R1030. PMID: 15475499.
Article3. Choi S, Park S, Liang GH, Kim JA, Suh SH. Superoxide generated by lysophosphatidylcholine induces endothelial nitric oxide synthase downregulation in human endothelial cells. Cell Physiol Biochem. 2010; 25:233–240. PMID: 20110684.
Article4. Matoba T, Shimokawa H, Kubota H, Morikawa K, Fujiki T, Kunihiro I, Mukai Y, Hirakawa Y, Takeshita A. Hydrogen peroxide is an endothelium-derived hyperpolarizing factor in human mesenteric arteries. Biochem Biophys Res Commun. 2002; 290:909–913. PMID: 11798159.
Article5. Matoba T, Shimokawa H, Nakashima M, Hirakawa Y, Mukai Y, Hirano K, Kanaide H, Takeshita A. Hydrogen peroxide is an endothelium-derived hyperpolarizing factor in mice. J Clin Invest. 2000; 106:1521–1530. PMID: 11120759.
Article6. Chang KC, Chung SY, Chong WS, Suh JS, Kim SH, Noh HK, Seong BW, Ko HJ, Chun KW. Possible superoxide radical-induced alteration of vascular reactivity in aortas from streptozotocin-treated rats. J Pharmacol Exp Ther. 1993; 266:992–1000. PMID: 8394927.7. Kanie N, Kamata K. Contractile responses in spontaneously diabetic mice. I. Involvement of superoxide anion in enhanced contractile response of aorta to norepinephrine in C57BL/KsJ (db/db) mice. Gen Pharmacol. 2000; 35:311–318. PMID: 11922961.8. Krippeit-Drews P, Haberland C, Fingerle J, Drews G, Lang F. Effects of H2O2 on membrane potential and [Ca2+]i of cultured rat arterial smooth muscle cells. Biochem Biophys Res Commun. 1995; 209:139–145. PMID: 7537038.9. Wei EP, Kontos HA, Beckman JS. Mechanisms of cerebral vasodilation by superoxide, hydrogen peroxide, and peroxynitrite. Am J Physiol. 1996; 271:H1262–H1266. PMID: 8853367.
Article10. Ichinari K, Kakei M, Matsuoka T, Nakashima H, Tanaka H. Direct activation of the ATP-sensitive potassium channel by oxygen free radicals in guinea-pig ventricular cells: its potentiation by MgADP. J Mol Cell Cardiol. 1996; 28:1867–1877. PMID: 8899545.11. Taglialatela M, Castaldo P, Iossa S, Pannaccione A, Fresi A, Ficker E, Annunziato L. Regulation of the human ether-a-gogo related gene (HERG) K+ channels by reactive oxygen species. Proc Natl Acad Sci U S A. 1997; 94:11698–11703. PMID: 9326673.12. Thomas GP, Sims SM, Cook MA, Karmazyn M. Hydrogen peroxide-induced stimulation of L-type calcium current in guinea pig ventricular myocytes and its inhibition by adenosine A1 receptor activation. J Pharmacol Exp Ther. 1998; 286:1208–1214. PMID: 9732380.13. Pei ZM, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI. Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature. 2000; 406:731–734. PMID: 10963598.14. Grupe M, Myers G, Penner R, Fleig A. Activation of store-operated I(CRAC) by hydrogen peroxide. Cell Calcium. 2010; 48:1–9. PMID: 20646759.
Article15. Duprat F, Guillemare E, Romey G, Fink M, Lesage F, Lazdunski M, Honore E. Susceptibility of cloned K+ channels to reactive oxygen species. Proc Natl Acad Sci U S A. 1995; 92:11796–11800. PMID: 8524851.16. Caouette D, Dongmo C, Bérubé J, Fournier D, Daleau P. Hydrogen peroxide modulates the Kv1.5 channel expressed in a mammalian cell line. Naunyn Schmiedebergs Arch Pharmacol. 2003; 368:479–486. PMID: 14614593.
Article17. Liu Y, Terata K, Rusch NJ, Gutterman DD. High glucose impairs voltage-gated K+ channel current in rat small coronary arteries. Circ Res. 2001; 89:146–152. PMID: 11463721.18. Liu Y, Gutterman DD. Oxidative stress and potassium channel function. Clin Exp Pharmacol Physiol. 2002; 29:305–311. PMID: 11985541.
Article19. Ahn SC, Seol GH, Kim JA, Suh SH. Characteristics and a functional implication of Ca2+-activated K+ current in mouse aortic endothelial cells. Pflugers Arch. 2004; 447:426–435. PMID: 14648123.20. Choi S, Kim JA, Na HY, Kim JE, Park S, Han KH, Kim YJ, Suh SH. NADPH oxidase 2-derived superoxide downregulates endothelial KCa3.1 in preeclampsia. Free Radic Biol Med. 2013; 57:10–21. PMID: 23261940.21. Park S, Kim JA, Joo KY, Choi S, Choi EN, Shin JA, Han KH, Jung SC, Suh SH. Globotriaosylceramide leads to KCa3.1 channel dysfunction: a new insight into endothelial dysfunction in Fabry disease. Cardiovasc Res. 2011; 89:290–299. PMID: 20971723.22. Si H, Heyken WT, Wölfle SE, Tysiac M, Schubert R, Grgic I, Vilianovich L, Giebing G, Maier T, Gross V, Bader M, de Wit C, Hoyer J, Köhler R. Impaired endothelium-derived hyperpolarizing factor-mediated dilations and increased blood pressure in mice deficient of the intermediate-conductance Ca2+-activated K+ channel. Circ Res. 2006; 99:537–544. PMID: 16873714.23. Jaffe EA, Nachman RL, Becker CG, Minick CR. Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J Clin Invest. 1973; 52:2745–2756. PMID: 4355998.
Article24. Lauf PK, Misri S, Chimote AA, Adragna NC. Apparent intermediate K conductance channel hyposmotic activation in human lens epithelial cells. Am J Physiol Cell Physiol. 2008; 294:C820–C832. PMID: 18184876.
Article25. Tharp DL, Bowles DK. The intermediate-conductance Ca2+-activated K+ channel (KCa3.1) in vascular disease. Cardiovasc Hematol Agents Med Chem. 2009; 7:1–11. PMID: 19149539.26. Cheong A, Bingham AJ, Li J, Kumar B, Sukumar P, Munsch C, Buckley NJ, Neylon CB, Porter KE, Beech DJ, Wood IC. Downregulated REST transcription factor is a switch enabling critical potassium channel expression and cell proliferation. Mol Cell. 2005; 20:45–52. PMID: 16209944.
Article27. Katusic ZS. Superoxide anion and endothelial regulation of arterial tone. Free Radic Biol Med. 1996; 20:443–448. PMID: 8720916.
Article28. Cosentino F, Patton S, d'Uscio LV, Werner ER, Werner-Felmayer G, Moreau P, Malinski T, Lüscher TF. Tetrahydrobiopterin alters superoxide and nitric oxide release in prehypertensive rats. J Clin Invest. 1998; 101:1530–1537. PMID: 9525996.
Article29. Stroes E, Hijmering M, van Zandvoort M, Wever R, Rabelink TJ, van Faassen EE. Origin of superoxide production by endothelial nitric oxide synthase. FEBS Lett. 1998; 438:161–164. PMID: 9827538.
Article30. Fulton D, McGiff JC, Wolin MS, Kaminski P, Quilley J. Evidence against a cytochrome P450-derived reactive oxygen species as the mediator of the nitric oxide-independent vasodilator effect of bradykinin in the perfused heart of the rat. J Pharmacol Exp Ther. 1997; 280:702–709. PMID: 9023282.31. Pomposiello S, Rhaleb NE, Alva M, Carretero OA. Reactive oxygen species: role in the relaxation induced by bradykinin or arachidonic acid via EDHF in isolated porcine coronary arteries. J Cardiovasc Pharmacol. 1999; 34:567–574. PMID: 10511133.
Article32. Lee SK, Lee JY, Joo HK, Cho EJ, Kim CS, Lee SD, Park JB, Jeon BH. Tat-mediated p66shc transduction decreased phosphorylation of endothelial nitric oxide synthase in endothelial Cells. Korean J Physiol Pharmacol. 2012; 16:199–204. PMID: 22802702.
Article33. Liu Y, Bubolz AH, Mendoza S, Zhang DX, Gutterman DD. H2O2 is the transferrable factor mediating flow-induced dilation in human coronary arterioles. Circ Res. 2011; 108:566–573. PMID: 21233456.34. Brakemeier S, Kersten A, Eichler I, Grgic I, Zakrzewicz A, Hopp H, Köhler R, Hoyer J. Shear stress-induced up-regulation of the intermediate-conductance Ca2+-activated K+ channel in human endothelium. Cardiovasc Res. 2003; 60:488–496. PMID: 14659794.35. Winterbourn CC, Hampton MB. Thiol chemistry and specificity in redox signaling. Free Radic Biol Med. 2008; 45:549–561. PMID: 18544350.
Article36. Gopalakrishnan V. REST and the RESTless: in stem cells and beyond. Future Neurol. 2009; 4:317–329. PMID: 19885378.
Article37. Westbrook TF, Martin ES, Schlabach MR, Leng Y, Liang AC, Feng B, Zhao JJ, Roberts TM, Mandel G, Hannon GJ, Depinho RA, Chin L, Elledge SJ. A genetic screen for candidate tumor suppressors identifies REST. Cell. 2005; 121:837–848. PMID: 15960972.
Article38. Liu Y, Terata K, Chai Q, Li H, Kleinman LH, Gutterman DD. Peroxynitrite inhibits Ca2+-activated K+ channel activity in smooth muscle of human coronary arterioles. Circ Res. 2002; 91:1070–1076. PMID: 12456494.39. Bychkov R, Pieper K, Ried C, Milosheva M, Bychkov E, Luft FC, Haller H. Hydrogen peroxide, potassium currents, and membrane potential in human endothelial cells. Circulation. 1999; 99:1719–1725. PMID: 10190882.
Article40. Barlow RS, White RE. Hydrogen peroxide relaxes porcine coronary arteries by stimulating BKCa channel activity. Am J Physiol. 1998; 275:H1283–H1289. PMID: 9746477.41. Dreher D, Junod AF. Differential effects of superoxide, hydrogen peroxide, and hydroxyl radical on intracellular calcium in human endothelial cells. J Cell Physiol. 1995; 162:147–153. PMID: 7814447.
Article42. Doan TN, Gentry DL, Taylor AA, Elliott SJ. Hydrogen peroxide activates agonist-sensitive Ca2+-flux pathways in canine venous endothelial cells. Biochem J. 1994; 297:209–215. PMID: 8280101.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Effects of Hydrogen Peroxide on the Migration and Proliferation of the Human Keratinocytes during Wound Healing
- Effects of Reactive Oxygen Species on Hyperactivation of Human Sperms
- Tat-Mediated p66shc Transduction Decreased Phosphorylation of Endothelial Nitric Oxide Synthase in Endothelial Cells
- Effects of Reactive Oxygen Species on Hyperactivation and Penetration Assay of Human Spermatozoa
- Effect of Hydrogen Peroxide-induced Oxidative Stress on the Senescence of Trabecular Meshwork Cells