Tat-Mediated p66shc Transduction Decreased Phosphorylation of Endothelial Nitric Oxide Synthase in Endothelial Cells
- Affiliations
-
- 1Infection Signaling Network Research Center, Research Institute of Medical Sciences, Department of Physiology, School of Medicine, Chungnam National University, Daejeon 301-131, Korea. bhjeon@cnu.ac.kr
- 2Department of Sports Science, Chungnam National University, Daejeon 305-765, Korea.
- KMID: 1768014
- DOI: http://doi.org/10.4196/kjpp.2012.16.3.199
Abstract
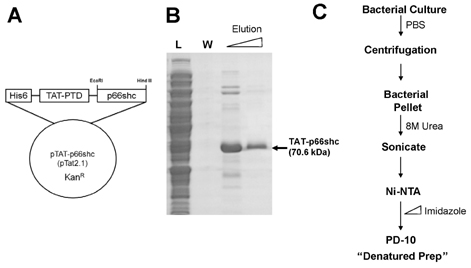
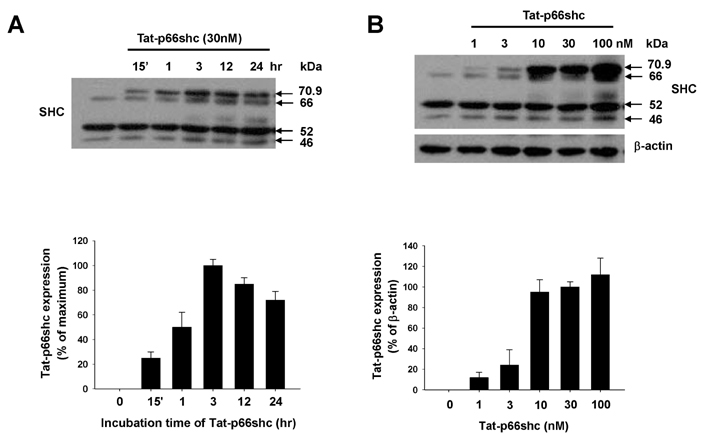
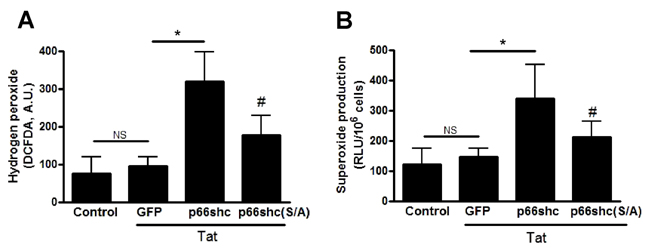
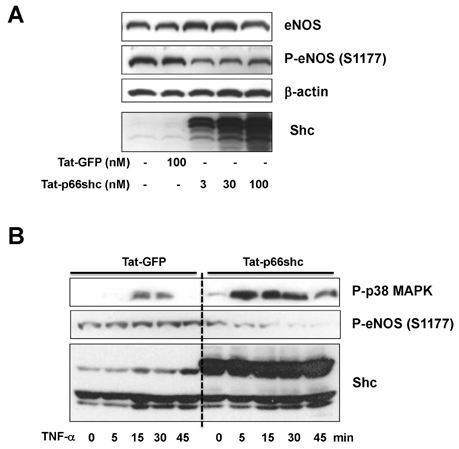
- We evaluated the role of Tat-mediated p66shc transduction on the activation of endothelial nitric oxide synthase in cultured mouse endothelial cells. To construct the Tat-p66shc fusion protein, human full length p66shc cDNA was fused with the Tat-protein transduction domain. Transduction of TAT-p66shc showed a concentration- and time-dependent manner in endothelial cells. Tat-mediated p66shc transduction showed increased hydrogen peroxide and superoxide production, compared with Tat-p66shc (S/A), serine 36 residue mutant of p66shc. Tat-mediated p66shc transduction decreased endothelial nitric oxide synthase phosphorylation in endothelial cells. Furthermore, Tat-mediated p66shc transduction augmented TNF-alpha-induced p38 MAPK phosphorylation in endothelial cells. These results suggest that Tat-mediated p66shc transduction efficiently inhibited endothelial nitric oxide synthase phosphorylation in endothelial cells.
Keyword
MeSH Terms
Figure
Cited by 3 articles
-
Contradictory Effects of Superoxide and Hydrogen Peroxide on KCa3.1 in Human Endothelial Cells
Shinkyu Choi, Hye-Young Na, Ji Aee Kim, Sung-Eun Cho, Suk Hyo Suh
Korean J Physiol Pharmacol. 2013;17(3):181-187. doi: 10.4196/kjpp.2013.17.3.181.Expression Profile of Neuro-Endocrine-Immune Network in Rats with Vascular Endothelial Dysfunction
Lujin Li, Zhenghua Jia, Ling Xu, Yiling Wu, Qingshan Zheng
Korean J Physiol Pharmacol. 2014;18(2):177-182. doi: 10.4196/kjpp.2014.18.2.177.Trichostatin A Modulates Angiotensin II-induced Vasoconstriction and Blood Pressure Via Inhibition of p66shc Activation
Gun Kang, Yu Ran Lee, Hee Kyoung Joo, Myoung Soo Park, Cuk-Seong Kim, Sunga Choi, Byeong Hwa Jeon
Korean J Physiol Pharmacol. 2015;19(5):467-472. doi: 10.4196/kjpp.2015.19.5.467.
Reference
-
1. Migliaccio E, Mele S, Salcini AE, Pelicci G, Lai KM, Superti-Furga G, Pawson T, Di Fiore PP, Lanfrancone L, Pelicci PG. Opposite effects of the p52shc/p46shc and p66shc splicing isoforms on the EGF receptor-MAP kinase-fos signalling pathway. EMBO J. 1997. 16:706–716.2. Stocker R, Keaney JF Jr. Role of oxidative modifications in atherosclerosis. Physiol Rev. 2004. 84:1381–1478.3. Molavi B, Mehta JL. Oxidative stress in cardiovascular disease: molecular basis of its deleterious effects, its detection, and therapeutic considerations. Curr Opin Cardiol. 2004. 19:488–493.4. Madamanchi NR, Vendrov A, Runge MS. Oxidative stress and vascular disease. Arterioscler Thromb Vasc Biol. 2005. 25:29–38.5. Napoli C, Martin-Padura I, de Nigris F, Giorgio M, Mansueto G, Somma P, Condorelli M, Sica G, De Rosa G, Pelicci P. Deletion of the p66Shc longevity gene reduces systemic and tissue oxidative stress, vascular cell apoptosis, and early atherogenesis in mice fed a high-fat diet. Proc Natl Acad Sci USA. 2003. 100:2112–2116.6. Kim CS, Son SJ, Kim EK, Kim SN, Yoo DG, Kim HS, Ryoo SW, Lee SD, Irani K, Jeon BH. Apurinic/apyrimidinic endonuclease1/redox factor-1 inhibits monocyte adhesion in endothelial cells. Cardiovasc Res. 2006. 69:520–526.7. Kawashima S. Malfunction of vascular control in lifestyle-related diseases: endothelial nitric oxide (NO) synthase/NO system in atherosclerosis. J Pharmacol Sci. 2004. 96:411–419.8. Martin-Padura I, de Nigris F, Migliaccio E, Mansueto G, Minardi S, Rienzo M, Lerman LO, Stendardo M, Giorgio M, De Rosa G, Pelicci PG, Napoli C. p66Shc deletion confers vascular protection in advanced atherosclerosis in hypercholesterolemic apolipoprotein E knockout mice. Endothelium. 2008. 15:276–287.9. Camici GG, Schiavoni M, Francia P, Bachschmid M, Martin-Padura I, Hersberger M, Tanner FC, Pelicci P, Volpe M, Anversa P, Lüscher TF, Cosentino F. Genetic deletion of p66(Shc) adaptor protein prevents hyperglycemia-induced endothelial dysfunction and oxidative stress. Proc Natl Acad Sci USA. 2007. 104:5217–5222.10. Yamamori T, White AR, Mattagajasingh I, Khanday FA, Haile A, Qi B, Jeon BH, Bugayenko A, Kasuno K, Berkowitz DE, Irani K. P66shc regulates endothelial NO production and endothelium-dependent vasorelaxation: implications for age-associated vascular dysfunction. J Mol Cell Cardiol. 2005. 39:992–995.11. Lee SK, Chung JI, Park MS, Joo HK, Lee EJ, Cho EJ, Park JB, Ryoo S, Irani K, Jeon BH. Apurinic/apyrimidinic endonuclease 1 inhibits protein kinase C-mediated p66shc phosphorylation and vasoconstriction. Cardiovasc Res. 2011. 91:502–509.12. Lee SK, Kim HS, Song YJ, Joo HK, Lee JY, Lee KH, Cho EJ, Cho CH, Park JB, Jeon BH. Alteration of p66shc is associated with endothelial dysfunction in the abdominal aortic coarctation of rats. FEBS Lett. 2008. 582:2561–2566.13. Becker-Hapak M, McAllister SS, Dowdy SF. TAT-mediated protein transduction into mammalian cells. Methods. 2001. 24:247–256.14. Song YJ, Lee JY, Joo HK, Kim HS, Lee SK, Lee KH, Cho CH, Park JB, Jeon BH. Tat-APE1/ref-1 protein inhibits TNF-alpha-induced endothelial cell activation. Biochem Biophys Res Commun. 2008. 368:68–73.15. Lee JY, Park KS, Cho EJ, Joo HK, Lee SK, Lee SD, Park JB, Chang SJ, Jeon BH. Human HOXA5 homeodomain enhances protein transduction and its application to vascular inflammation. Biochem Biophys Res Commun. 2011. 410:312–316.16. Angkeow P, Deshpande SS, Qi B, Liu YX, Park YC, Jeon BH, Ozaki M, Irani K. Redox factor-1: an extra-nuclear role in the regulation of endothelial oxidative stress and apoptosis. Cell Death Differ. 2002. 9:717–725.17. Wender PA, Mitchell DJ, Pattabiraman K, Pelkey ET, Steinman L, Rothbard JB. The design, synthesis, and evaluation of molecules that enable or enhance cellular uptake: peptoid molecular transporters. Proc Natl Acad Sci USA. 2000. 97:13003–13008.18. Francia P, delli Gatti C, Bachschmid M, Martin-Padura I, Savoia C, Migliaccio E, Pelicci PG, Schiavoni M, Lüscher TF, Volpe M, Cosentino F. Deletion of p66shc gene protects against age-related endothelial dysfunction. Circulation. 2004. 110:2889–2895.19. Wu Z, Rogers B, Kachi S, Hackett SF, Sick A, Campochiaro PA. Reduction of p66Shc suppresses oxidative damage in retinal pigmented epithelial cells and retina. J Cell Physiol. 2006. 209:996–1005.20. Anderson TJ. Nitric oxide, atherosclerosis and the clinical relevance of endothelial dysfunction. Heart Fail Rev. 2003. 8:71–86.21. Kim DD, Sánchez FA, Durán RG, Kanetaka T, Durán WN. Endothelial nitric oxide synthase is a molecular vascular target for the Chinese herb Danshen in hypertension. Am J Physiol Heart Circ Physiol. 2007. 292:H2131–H2137.22. Cho EJ, Park MS, Kim SS, Kang G, Choi S, Lee YR, Chang SJ, Lee KH, Lee SD, Park JB, Jeon BH. Vasorelaxing activity of ulmus davidiana ethanol extracts in rats: activation of endothelial nitric oxide synthase. Korean J Physiol Pharmacol. 2011. 15:339–344.23. Shin W, Cuong TD, Lee JH, Min B, Jeon BH, Lim HK, Ryoo S. Arginase inhibition by ethylacetate extract of caesalpinia sappan lignum contributes to activation of endothelial nitric oxide synthase. Korean J Physiol Pharmacol. 2011. 15:123–128.24. Lee SK, Kim YS, Kim CS, Son SJ, Yoo DG, Lee KH, Lee SD, Park JB, Jeon BH. p66shc adaptor protein suppresses the activation of endothelial nitric oxide synthase in mouse embryonic fibroblasts. Korean J Physiol Pharmacol. 2006. 10:155–159.25. Grethe S, Ares MP, Andersson T, Pörn-Ares MI. p38 MAPK mediates TNF-induced apoptosis in endothelial cells via phosphorylation and downregulation of Bcl-x(L). Exp Cell Res. 2004. 298:632–642.26. Yu JH, Kim CS, Yoo DG, Song YJ, Joo HK, Kang G, Jo JY, Park JB, Jeon BH. NADPH oxidase and mitochondrial ROS are involved in the TNF-alpha-induced vascular cell adhesion molecule-1 and monocyte adhesion in cultured endothelial cells. Korean J Physiol Pharmacol. 2006. 10:217–222.27. Li G, Barrett EJ, Barrett MO, Cao W, Liu Z. Tumor necrosis factor-alpha induces insulin resistance in endothelial cells via a p38 mitogen-activated protein kinase-dependent pathway. Endocrinology. 2007. 148:3356–3363.28. Zhang XH, Yokoo H, Nishioka H, Fujii H, Matsuda N, Hayashi T, Hattori Y. Beneficial effect of the oligomerized polyphenol oligonol on high glucose-induced changes in eNOS phosphorylation and dephosphorylation in endothelial cells. Br J Pharmacol. 2010. 159:928–938.29. Xing F, Jiang Y, Liu J, Zhao K, Mo Y, Liu Z, Zeng Y. Downregulation of human endothelial nitric oxide synthase promoter activity by p38 mitogen-activated protein kinase activation. Biochem Cell Biol. 2006. 84:780–788.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- p66shc Adaptor Protein Suppresses the Activation of Endothelial Nitric Oxide Synthase in Mouse Embryonic Fibroblasts
- Immunohistochemical Expression of Placental Nitric Oxide Synthase in Preeclampsia and Normal Pregnancy
- Genetic Polymorphism of Endothelial Nitric Oxide Synthase in Korean Schizophrenic Patients
- Nitric oxide in liver fibrosis: The role of inducible nitric oxide synthase
- The Peroxisome Proliferator-Activated Receptor delta Agonist, GW501516, Inhibits Angiogenesis through Dephosphorylation of Endothelial Nitric Oxide Synthase