Int J Stem Cells.
2020 Nov;13(3):394-403. 10.15283/ijsc20072.
Bone Marrow-Derived Mesenchymal Stem Cells Isolated from Patients with Cirrhosis and Healthy Volunteers Show Comparable Characteristics
- Affiliations
-
- 1Department of Internal Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- 2Cell Therapy and Tissue Engineering Center, Yonsei University Wonju College of Medicine, Wonju, Korea
- 3Regeneration Medicine Research Center, Yonsei University Wonju College of Medicine, Wonju, Korea
- 4Mitohormesis Research Center, Yonsei University Wonju College of Medicine, Wonju, Korea
- 5Department of Physiology, Yonsei University Wonju College of Medicine, Wonju, Korea
- KMID: 2508913
- DOI: http://doi.org/10.15283/ijsc20072
Abstract
- Background and Objectives
Autologous or allogeneic bone marrow-derived mesenchymal stem cells (BMSCs) have been applied in clinical trials to treat liver disease. However, only a few studies are comparing the characteristics of autologous MSCs from patients and allogeneic MSCs from normal subjects.
Methods and Results
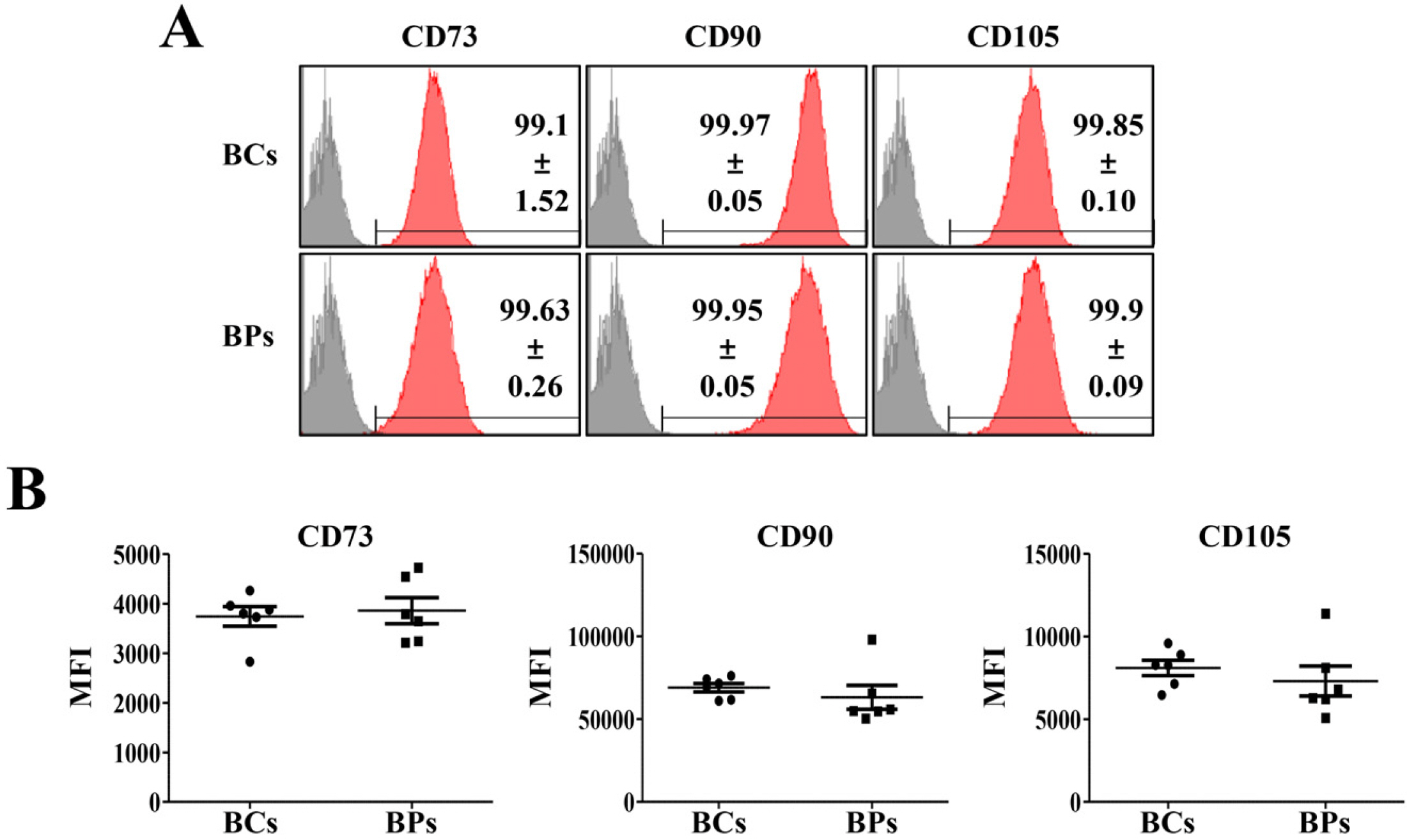
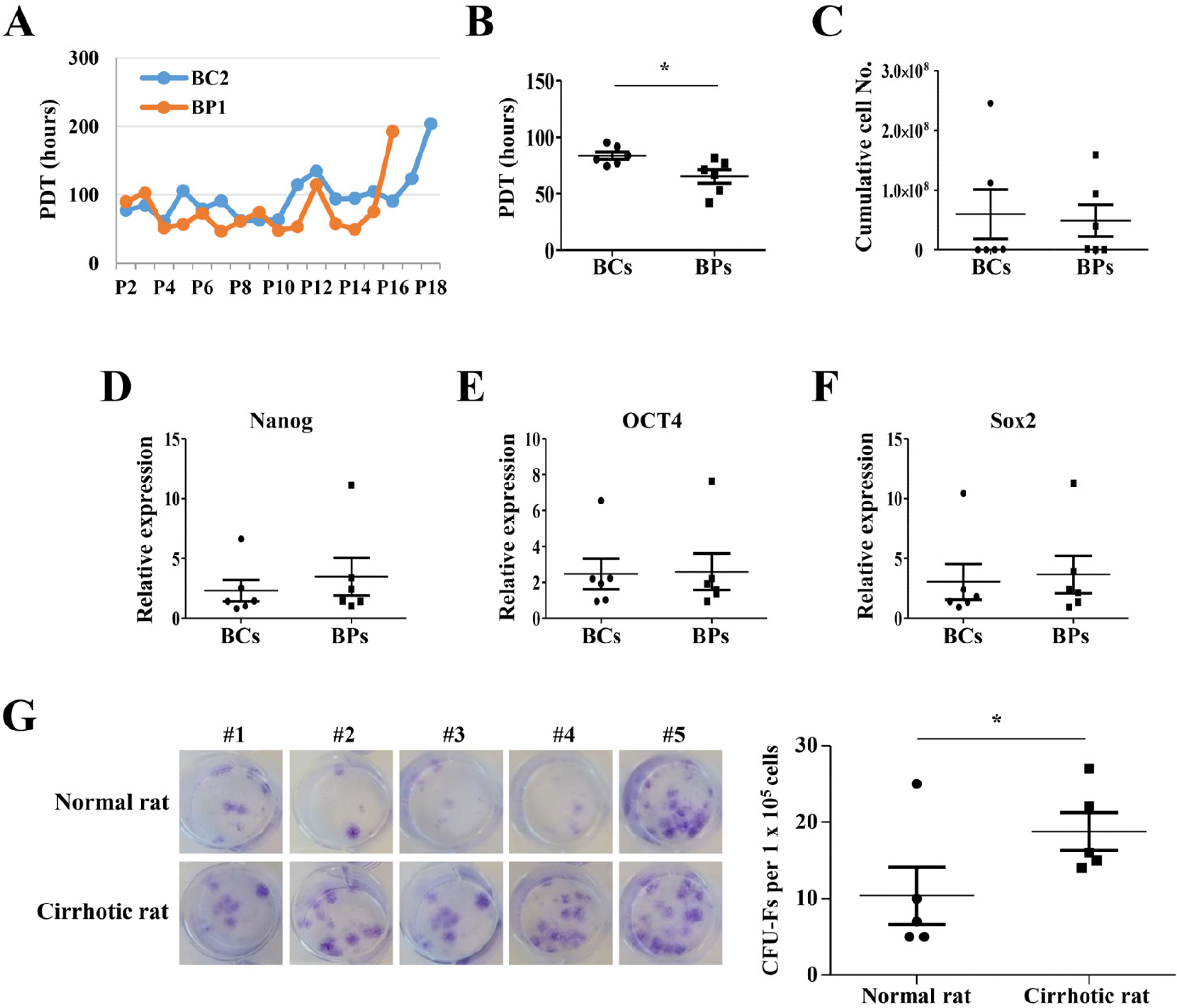
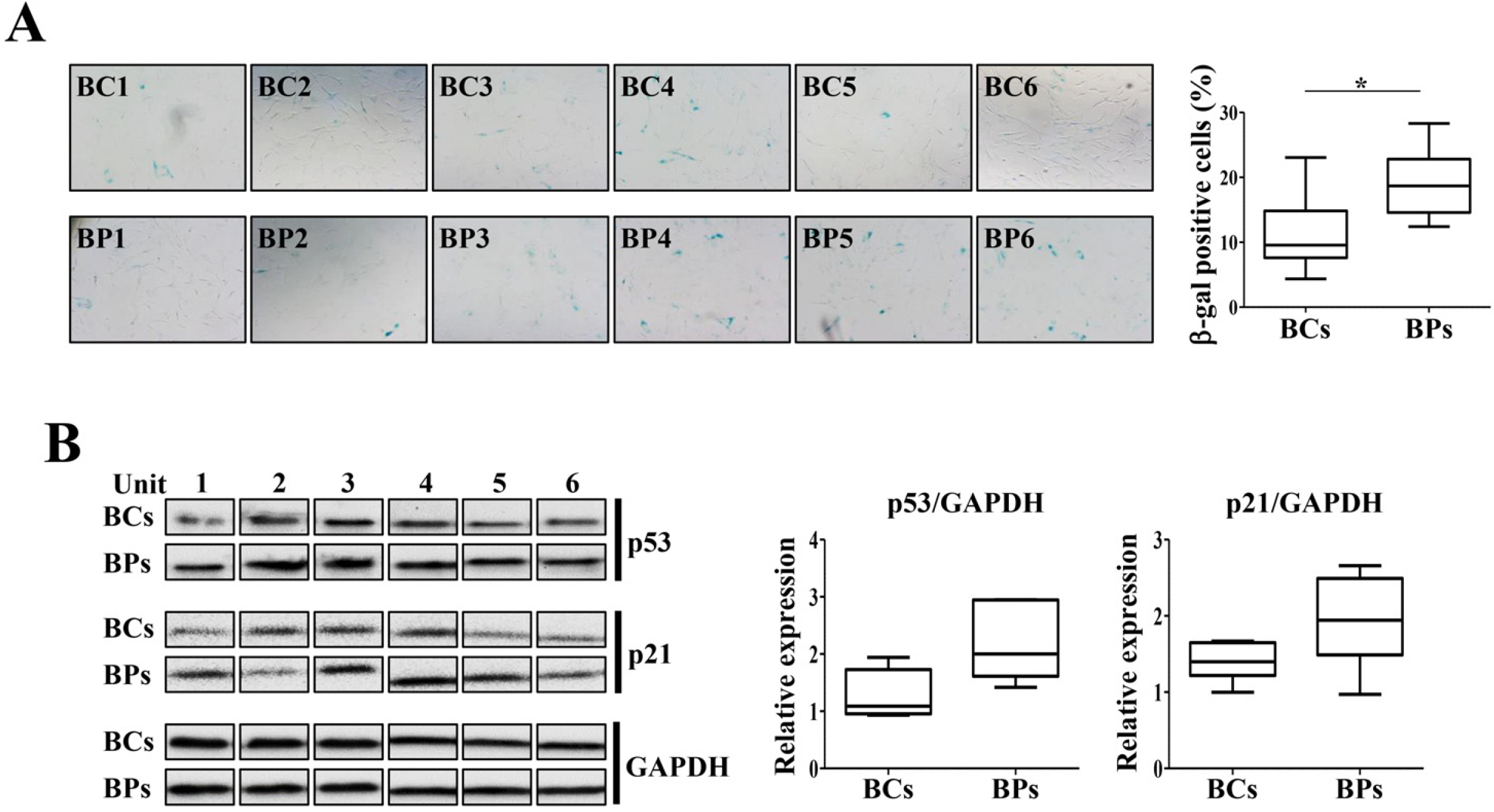
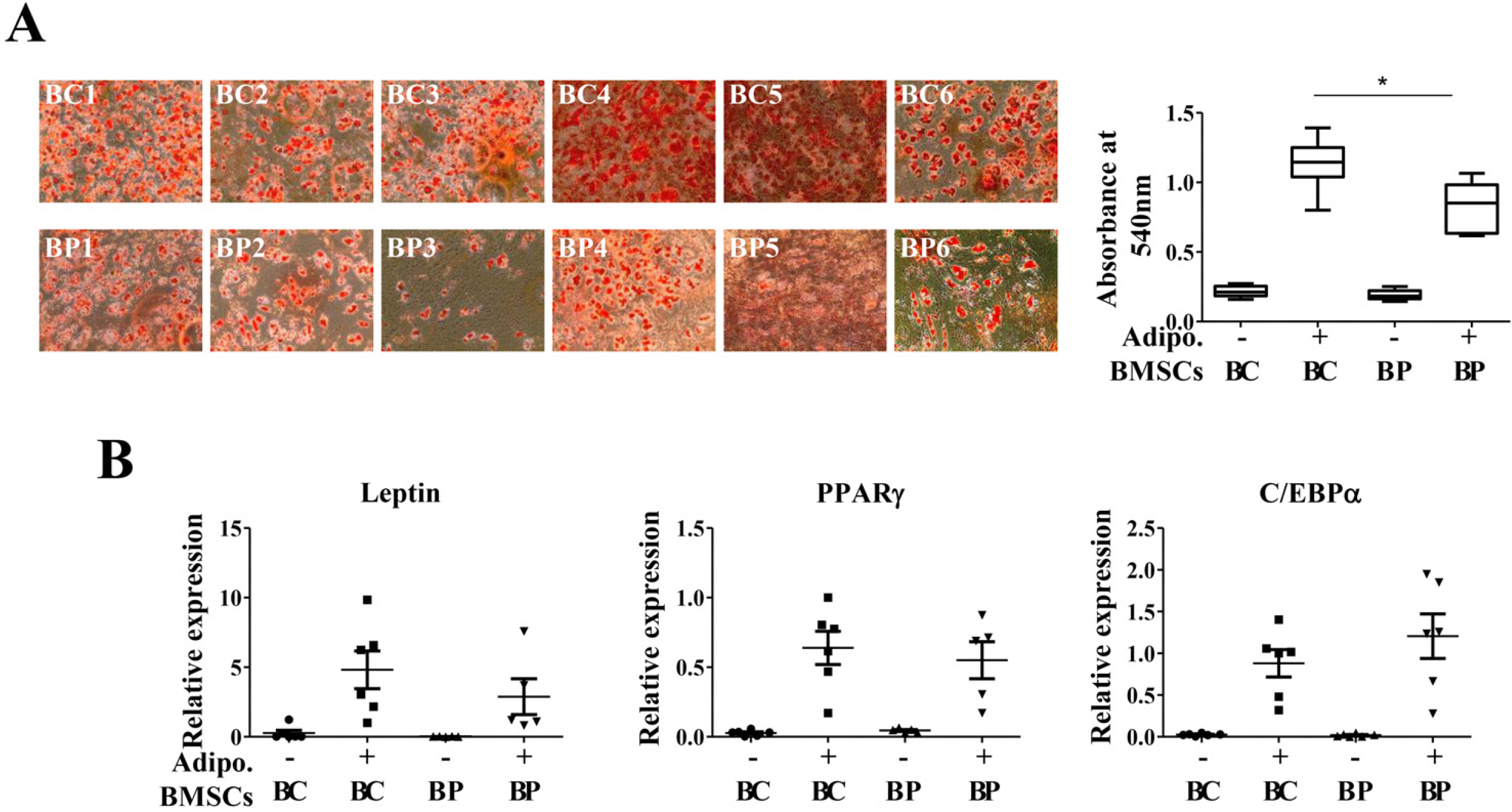
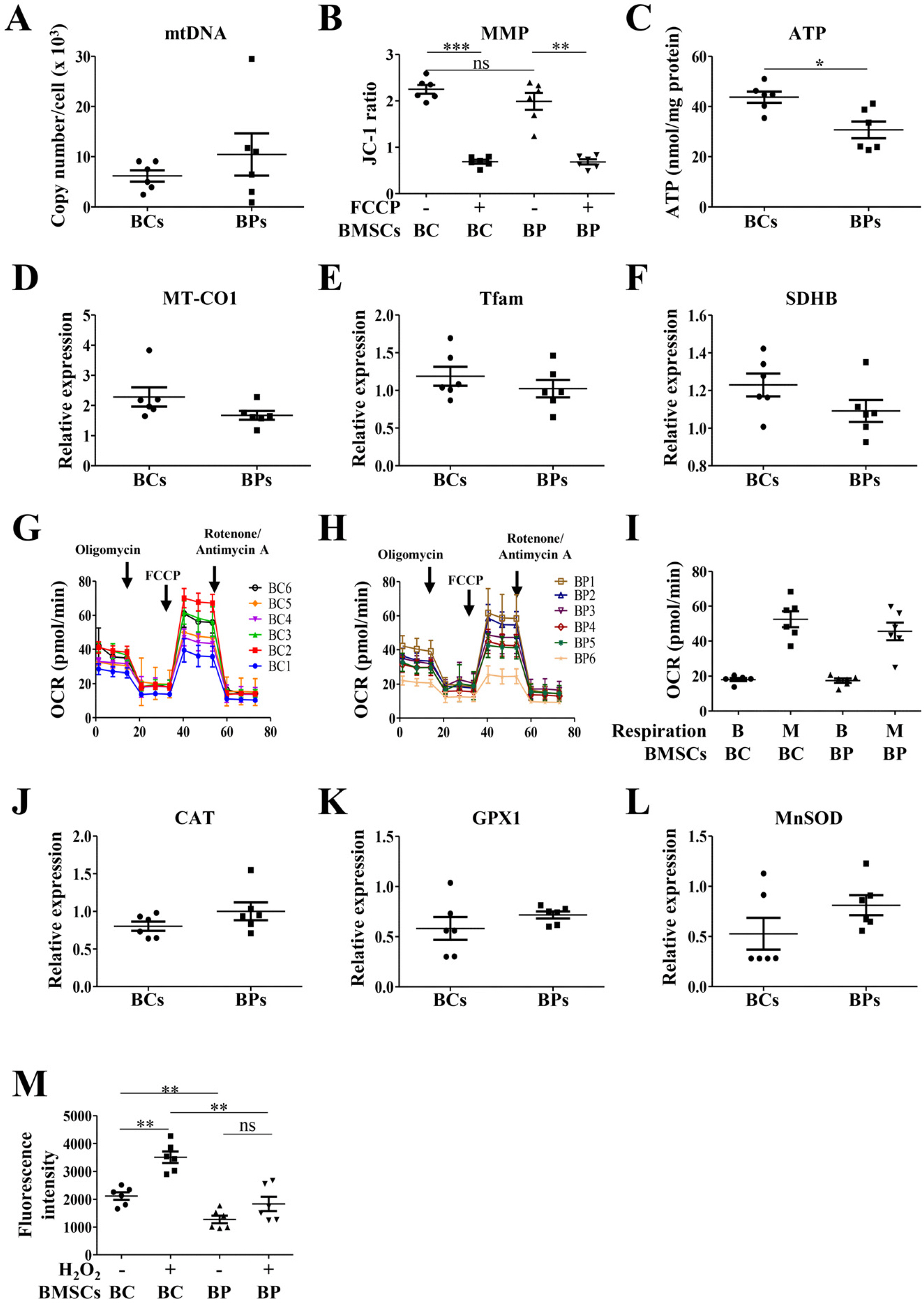
We compared the characteristics of BMSCs (BCs and BPs, respectively) isolated from six healthy volunteers and six patients with cirrhosis. In passage 3 (P3), senescent population and expression of p53 and p21 were slightly higher in BPs, but the average population doubling time for P3–P5 in BPs was approximately 65.3±11.1 h, which is 18.4 h shorter than that in BCs (83.7±9.2 h). No difference was observed in the expression of CD73, CD90, or CD105 between BCs and BPs. Adipogenic differentiation slightly increased in BCs, but the expression levels of leptin, peroxisome proliferator-activated receptor γ, and CCAAT-enhancer-binding protein α did not vary between differentiated BCs and BPs. While ATP and reactive oxygen species levels were slightly lower in BPs, mitochondrial membrane potential, oxygen consumption rate, and expression of mitochondria-related genes such as cytochrome c oxidase 1 were not significantly different between BCs and BPs.
Conclusions
Taken together, there are marginal differences in the proliferation, differentiation, and mitochondrial activities of BCs and BPs, but both BMSCs from patients with cirrhosis and healthy volunteers show comparable characteristics.
Figure
Reference
-
References
1. Singal AK, Anand BS. 2013; Recent trends in the epidemiology of alcoholic liver disease. Clin Liver Dis (Hoboken). 2:53–56. DOI: 10.1002/cld.168. PMID: 30992823. PMCID: PMC6448620.
Article2. Ichai P, Samuel D. 2011; Epidemiology of liver failure. Clin Res Hepatol Gastroenterol. 35:610–617. DOI: 10.1016/j.clinre.2011.03.010. PMID: 21550329.
Article3. Fallowfield JA, Iredale JP. 2004; Targeted treatments for cirrhosis. Expert Opin Ther Targets. 8:423–435. DOI: 10.1517/14728222.8.5.423. PMID: 15469393.
Article4. Houlihan DD, Newsome PN. 2008; Critical review of clinical trials of bone marrow stem cells in liver disease. Gastroentero-logy. 135:438–450. DOI: 10.1053/j.gastro.2008.05.040. PMID: 18585384.
Article5. Souza BS, Nogueira RC, de Oliveira SA, de Freitas LA, Lyra LG, Ribeiro dos Santos R, Lyra AC, Soares MB. 2009; Current status of stem cell therapy for liver diseases. Cell Transplant. 18:1261–1279. DOI: 10.3727/096368909X470522. PMID: 19660179.
Article6. Bird TG, Lorenzini S, Forbes SJ. 2008; Activation of stem cells in hepatic diseases. Cell Tissue Res. 331:283–300. DOI: 10.1007/s00441-007-0542-z. PMID: 18046579. PMCID: PMC3034134.
Article7. Peggins JO, McMahon TF, Beierschmitt WP, Weiner M. 1987; Comparison of hepatic and renal metabolism of acetami-nophen in male and female miniature swine. Drug Metab Dispos. 15:270–273. PMID: 2882990.8. Muraca M. 2011; Evolving concepts in cell therapy of liver disease and current clinical perspectives. Dig Liver Dis. 43:180–187. DOI: 10.1016/j.dld.2010.08.007. PMID: 20869923.
Article9. Stutchfield BM, Forbes SJ, Wigmore SJ. 2010; Prospects for stem cell transplantation in the treatment of hepatic disease. Liver Transpl. 16:827–836. DOI: 10.1002/lt.22083. PMID: 20583084.
Article10. Campagnoli C, Roberts IA, Kumar S, Bennett PR, Bellantuono I, Fisk NM. 2001; Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood. 98:2396–2402. DOI: 10.1182/blood.V98.8.2396. PMID: 11588036.
Article11. Erices A, Conget P, Minguell JJ. 2000; Mesenchymal progenitor cells in human umbilical cord blood. Br J Haematol. 109:235–242. DOI: 10.1046/j.1365-2141.2000.01986.x. PMID: 10848804.
Article12. Young HE, Steele TA, Bray RA, Hudson J, Floyd JA, Hawkins K, Thomas K, Austin T, Edwards C, Cuzzourt J, Duenzl M, Lucas PA, Black AC Jr. 2001; Human reserve pluripotent mesenchymal stem cells are present in the connective tissues of skeletal muscle and dermis derived from fetal, adult, and geriatric donors. Anat Rec. 264:51–62. DOI: 10.1002/ar.1128. PMID: 11505371.
Article13. Xiang GA, Zhang GQ, Fang CH, Gao P, Chen KY. 2005; A preliminary study of the homing capacity of allograft mesenchymal stem cells to rat liver. Di Yi Jun Yi Da Xue Xue Bao. 25:994–997. Chinese.14. Brooke G, Tong H, Levesque JP, Atkinson K. 2008; Molecular trafficking mechanisms of multipotent mesenchymal stem cells derived from human bone marrow and placenta. Stem Cells Dev. 17:929–940. DOI: 10.1089/scd.2007.0156. PMID: 18564033.
Article15. Schwartz RE, Reyes M, Koodie L, Jiang Y, Blackstad M, Lund T, Lenvik T, Johnson S, Hu WS, Verfaillie CM. 2002; Multipotent adult progenitor cells from bone marrow differentiate into functional hepatocyte-like cells. J Clin Invest. 109:1291–1302. DOI: 10.1172/JCI0215182. PMID: 12021244. PMCID: PMC150983.
Article16. De Miguel MP, Fuentes-Julián S, Blázquez-Martínez A, Pascual CY, Aller MA, Arias J, Arnalich-Montiel F. 2012; Immunosuppressive properties of mesenchymal stem cells: advances and applications. Curr Mol Med. 12:574–591. DOI: 10.2174/156652412800619950. PMID: 22515979.
Article17. Higashiyama R, Inagaki Y, Hong YY, Kushida M, Nakao S, Niioka M, Watanabe T, Okano H, Matsuzaki Y, Shiota G, Okazaki I. 2007; Bone marrow-derived cells express matrix metalloproteinases and contribute to regression of liver fibrosis in mice. Hepatology. 45:213–222. DOI: 10.1002/hep.21477. PMID: 17187438.
Article18. Cho KA, Woo SY, Seoh JY, Han HS, Ryu KH. 2012; Mesenchy-mal stem cells restore CCl4-induced liver injury by an antioxidative process. Cell Biol Int. 36:1267–1274. DOI: 10.1042/CBI20110634. PMID: 23035905.
Article19. Kim G, Eom YW, Baik SK, Shin Y, Lim YL, Kim MY, Kwon SO, Chang SJ. 2015; Therapeutic effects of mesenchymal stem cells for patients with chronic liver diseases: systematic review and meta-analysis. J Korean Med Sci. 30:1405–1415. DOI: 10.3346/jkms.2015.30.10.1405. PMID: 26425036. PMCID: PMC4575928.
Article20. Alfaifi M, Eom YW, Newsome PN, Baik SK. 2018; Mesenchymal stromal cell therapy for liver diseases. J Hepatol. 68:1272–1285. DOI: 10.1016/j.jhep.2018.01.030. PMID: 29425678.
Article21. Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, Medrano EE, Linskens M, Rubelj I, Pereira-Smith O, Peacocke M, Campisi J. 1995; A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci U S A. 92:9363–9367. DOI: 10.1073/pnas.92.20.9363. PMID: 7568133. PMCID: PMC40985.
Article22. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. 2006; Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 8:315–317. DOI: 10.1080/14653240600855905. PMID: 16923606.
Article23. Eom YW, Oh JE, Lee JI, Baik SK, Rhee KJ, Shin HC, Kim YM, Ahn CM, Kong JH, Kim HS, Shim KY. 2014; The role of growth factors in maintenance of stemness in bone marrow-derived mesenchymal stem cells. Biochem Biophys Res Commun. 445:16–22. DOI: 10.1016/j.bbrc.2014.01.084. PMID: 24491556.
Article24. DiMauro S, Schon EA. 2003; Mitochondrial respiratory-chain diseases. N Engl J Med. 348:2656–2668. DOI: 10.1056/NEJMra022567. PMID: 12826641.
Article25. Zamzami N, Marchetti P, Castedo M, Decaudin D, Macho A, Hirsch T, Susin SA, Petit PX, Mignotte B, Kroemer G. 1995; Sequential reduction of mitochondrial transmembrane potential and generation of reactive oxygen species in early programmed cell death. J Exp Med. 182:367–377. DOI: 10.1084/jem.182.2.367. PMID: 7629499. PMCID: PMC2192111.
Article26. Li X, Fang P, Mai J, Choi ET, Wang H, Yang XF. 2013; Targeting mitochondrial reactive oxygen species as novel therapy for inflammatory diseases and cancers. J Hematol Oncol. 6:19. DOI: 10.1186/1756-8722-6-19. PMID: 23442817. PMCID: PMC3599349.
Article27. Zhang M, Du Y, Lu R, Shu Y, Zhao W, Li Z, Zhang Y, Liu R, Yang T, Luo S, Gao M, Zhang Y, Zhang G, Liu J, Lu Y. 2016; Cholesterol retards senescence in bone marrow mesenchymal stem cells by modulating autophagy and ROS/p53/p21Cip1/Waf1 pathway. Oxid Med Cell Longev. 2016:7524308. DOI: 10.1155/2016/7524308. PMID: 27703600. PMCID: PMC5040816.28. Denu RA, Hematti P. 2016; Effects of oxidative stress on mesenchymal stem cell biology. Oxid Med Cell Longev. 2016:2989076. DOI: 10.1155/2016/2989076. PMID: 27413419. PMCID: PMC4928004.
Article29. Tofiño-Vian M, Guillén MI, Pérez Del Caz MD, Castejón MA, Alcaraz MJ. 2017; Extracellular vesicles from adipose-derived mesenchymal stem cells downregulate senescence features in osteoarthritic osteoblasts. Oxid Med Cell Longev. 2017:7197598. DOI: 10.1155/2017/7197598. PMID: 29230269. PMCID: PMC5694590.
Article30. Xu M, Bradley EW, Weivoda MM, Hwang SM, Pirtskhalava T, Decklever T, Curran GL, Ogrodnik M, Jurk D, Johnson KO, Lowe V, Tchkonia T, Westendorf JJ, Kirkland JL. 2017; Transplanted senescent cells induce an osteoarthritis-like condition in mice. J Gerontol A Biol Sci Med Sci. 72:780–785. DOI: 10.1093/gerona/glw154. PMID: 27516624. PMCID: PMC5861939.
Article31. McCulloch K, Litherland GJ, Rai TS. 2017; Cellular senescence in osteoarthritis pathology. Aging Cell. 16:210–218. DOI: 10.1111/acel.12562. PMID: 28124466. PMCID: PMC5334539.
Article32. Platas J, Guillén MI, Pérez Del Caz MD, Gomar F, Castejón MA, Mirabet V, Alcaraz MJ. 2016; Paracrine effects of human adipose-derived mesenchymal stem cells in inflammatory stress-induced senescence features of osteoarthritic chondrocytes. Aging (Albany NY). 8:1703–1717. DOI: 10.18632/aging.101007. PMID: 27490266. PMCID: PMC5032691.
Article33. Yu B, Sondag GR, Malcuit C, Kim MH, Safadi FF. 2016; Macrophage-associated Osteoactivin/GPNMB mediates me-senchymal stem cell survival, proliferation, and migration via a CD44-dependent mechanism. J Cell Biochem. 117:1511–1521. DOI: 10.1002/jcb.25394. PMID: 26442636.
Article34. Buravkova LB, Rylova YV, Andreeva ER, Kulikov AV, Pogodina MV, Zhivotovsky B, Gogvadze V. 2013; Low ATP level is sufficient to maintain the uncommitted state of multipotent mesenchymal stem cells. Biochim Biophys Acta. 1830:4418–4425. DOI: 10.1016/j.bbagen.2013.05.029. PMID: 23742825.
Article35. Coppi E, Pugliese AM, Urbani S, Melani A, Cerbai E, Mazzanti B, Bosi A, Saccardi R, Pedata F. 2007; ATP modulates cell proliferation and elicits two different electrophysiolo-gical responses in human mesenchymal stem cells. Stem Cells. 25:1840–1849. DOI: 10.1634/stemcells.2006-0669. PMID: 17446563.
Article36. Zhang H, Menzies KJ, Auwerx J. 2018; The role of mitochondria in stem cell fate and aging. Development. 145:dev143420. DOI: 10.1242/dev.143420. PMID: 29654217. PMCID: PMC5964648.
Article37. Chen CT, Shih YR, Kuo TK, Lee OK, Wei YH. 2008; Coordi-nated changes of mitochondrial biogenesis and antioxidant enzymes during osteogenic differentiation of human mesenchymal stem cells. Stem Cells. 26:960–968. DOI: 10.1634/stemcells.2007-0509. PMID: 18218821.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Concise Review: Differentiation of Human Adult Stem Cells Into Hepatocyte-like Cells In vitro
- Isolation of Mesenchymal Stem Cells from the Mononuclear Cells Remaining in the Bone Marrow Processing Kit
- Clinical Safety and Efficacy of Autologous Bone Marrow-Derived Mesenchymal Stem Cell Transplantation in Sensorineural Hearing Loss Patients
- Characterization of multipotent mesenchymal stem cells isolated from adipose tissue and bone marrow in pigs
- Differential Potential of Stem Cells Following Their Origin: Subacromial Bursa, Bone Marrow, Umbilical Cord Blood