Cancer Res Treat.
2019 Apr;51(2):696-705. 10.4143/crt.2018.249.
Disulfiram, a Re-positioned Aldehyde Dehydrogenase Inhibitor, Enhances Radiosensitivity of Human Glioblastoma Cells In Vitro
- Affiliations
-
- 1Department of Radiation Oncology, Konkuk University Medical Center, Seoul, Korea.
- 2Department of Radiation Oncology, Seoul National University College of Medicine, Seoul, Korea. ihkim@snu.ac.kr
- 3Cancer Research Institute, Seoul National University, Seoul, Korea.
- 4Division of Pediatric Neurosurgery, Pediatric Clinical Neuroscience Center, Seoul National University Children's Hospital, Seoul, Korea.
- KMID: 2464415
- DOI: http://doi.org/10.4143/crt.2018.249
Abstract
- PURPOSE
Glioblastoma, the most common brain tumor in adults, has poor prognosis. The purpose of this study was to determine the effect of disulfiram (DSF), an aldehyde dehydrogenase inhibitor, on in vitro radiosensitivity of glioblastoma cells with different methylation status of Oâ¶-methylguanine-DNA methyltransferase (MGMT) promoter and the underlying mechanism of such effect.
MATERIALS AND METHODS
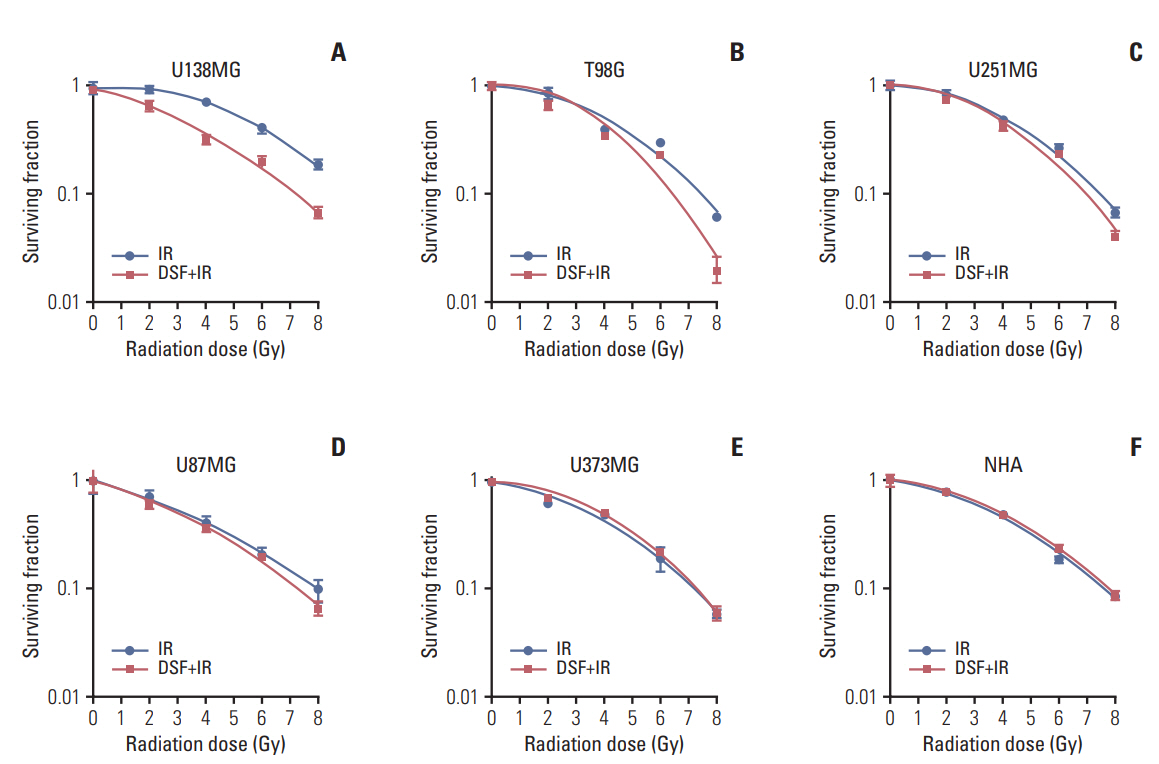
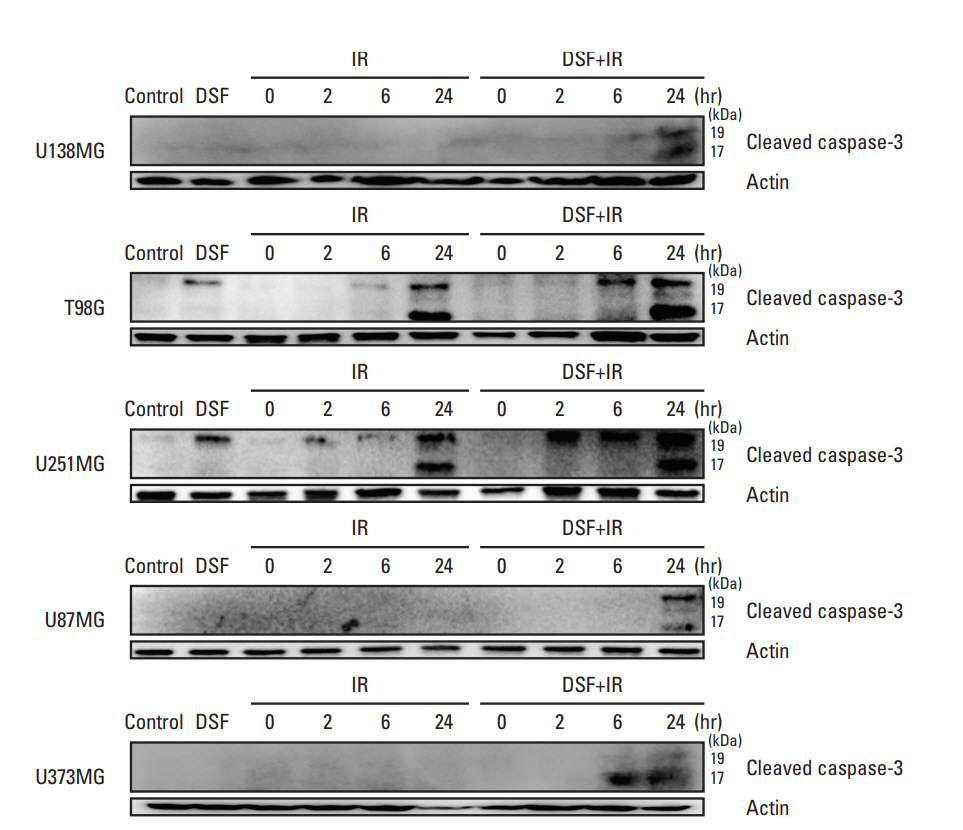
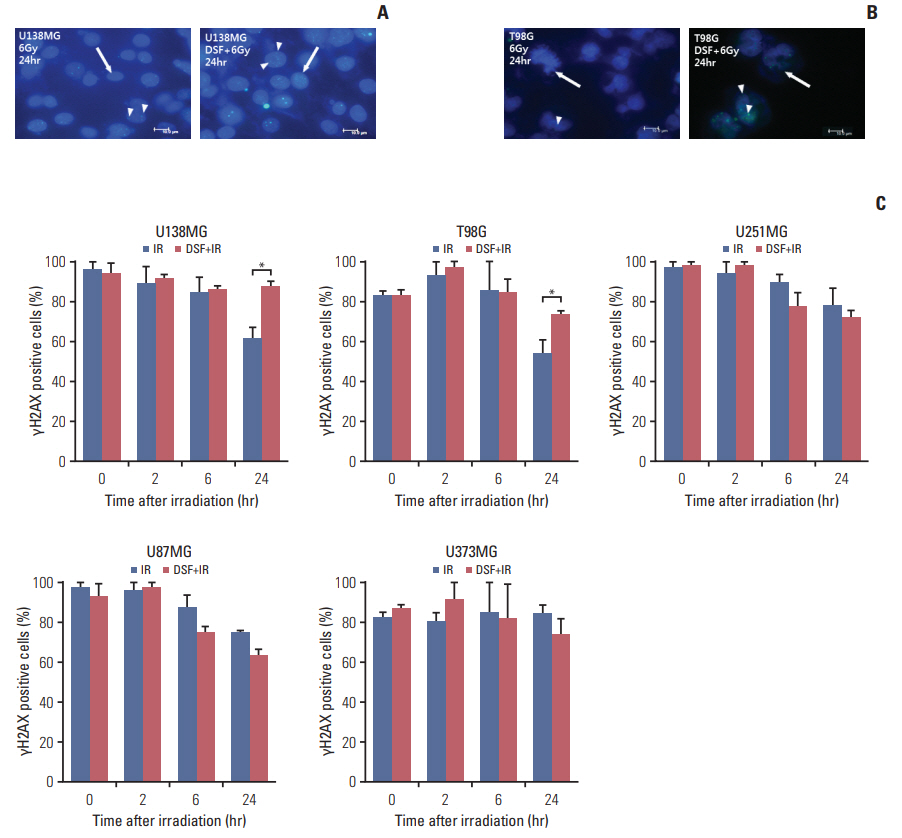
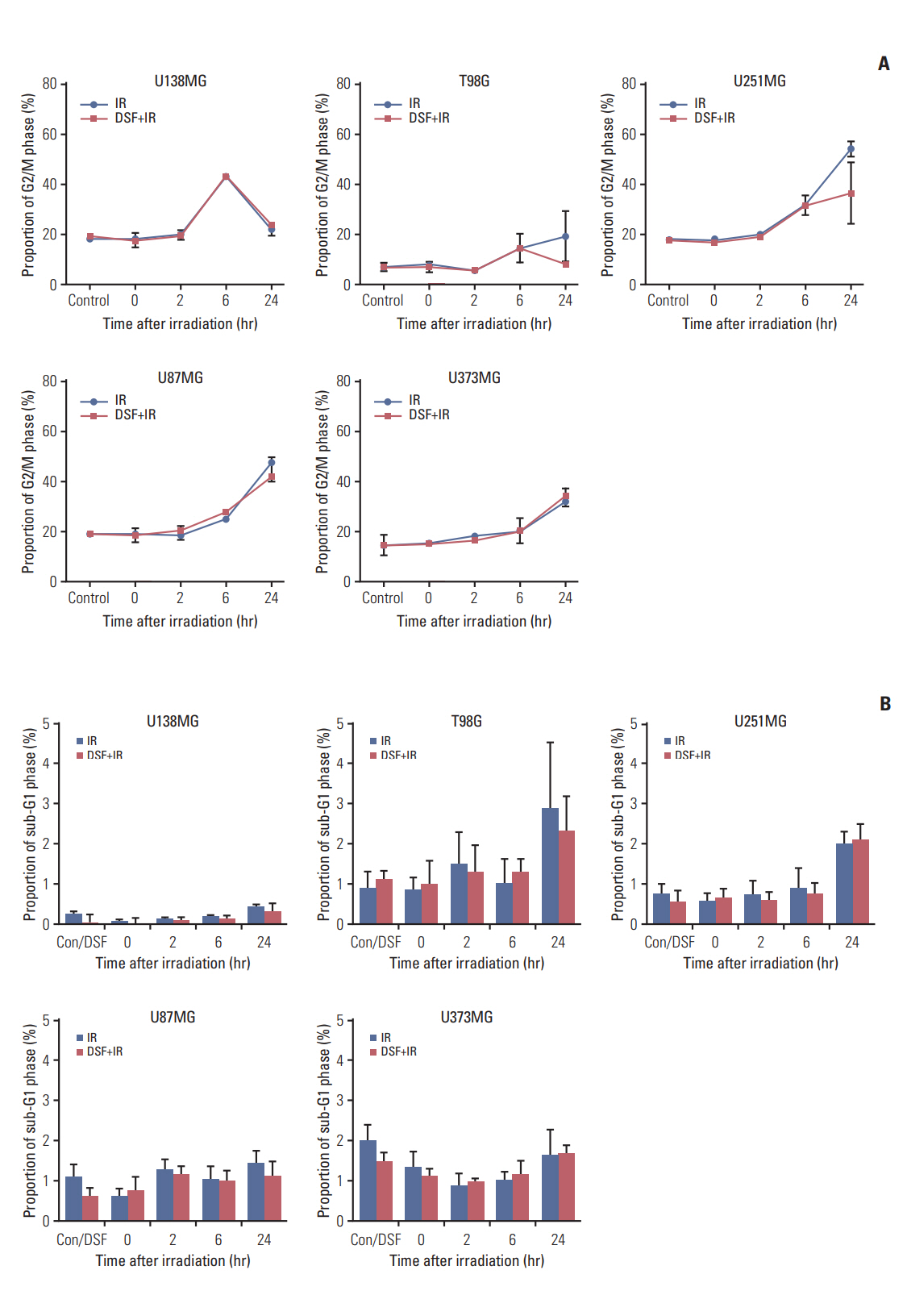
Five human glioblastoma cells (U138MG, T98G, U251MG, U87MG, and U373MG) and one normal human astrocyte (NHA) cell were cultured and treated with DSF or 6MV X-rays (0, 2, 4, 6, and 8 Gy). For combined treatment, cells were treated with DSF before irradiation. Surviving fractions fit from cell survival based on colony forming ability. Apoptosis, DNA damage repair, and cell cycle distributionwere assayed bywestern blot for cleaved caspase-3, γH2AX staining, and flow cytometry, respectively.
RESULTS
DSF induced radiosensitization in most of the glioblastoma cells, especially, in the cells with radioresistance as wildtype unmethylated promoter (MGMT-wt), but did not in normal NHA cell. DSF augmented or induced cleavage of caspase-3 in all cells after irradiation. DSF inhibited repair of radiation-induced DNA damage in MGMT-wt cells, but not in cells with methylated MGMT promoter. DSF abrogated radiation-induced G2/M arrest in T98G and U251MG cells.
CONCLUSION
Radiosensitivity of glioblastoma cells were preferentially enhanced by pre-irradiation DSF treatment compared to normal cell, especially radioresistant cells such as MGMT-wt cells. Induction of apoptosis or inhibition of DNA damage repair may underlie DSF-induced radiosensitization. Clinical benefit of combining DSF with radiotherapy should be investigated in the future.
Keyword
MeSH Terms
Figure
Reference
-
References
1. Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009; 10:459–66.
Article2. Mirimanoff RO, Gorlia T, Mason W, Van den Bent MJ, Kortmann RD, Fisher B, et al. Radiotherapy and temozolomide for newly diagnosed glioblastoma: recursive partitioning analysis of the EORTC 26981/22981-NCIC CE3 phase III randomized trial. J Clin Oncol. 2006; 24:2563–9.
Article3. Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med. 2005; 352:997–1003.4. Costello JF, Futscher BW, Tano K, Graunke DM, Pieper RO. Graded methylation in the promoter and body of the O6-methylguanine DNA methyltransferase (MGMT) gene correlates with MGMT expression in human glioma cells. J Biol Chem. 1994; 269:17228–37.
Article5. Wee CW, Kim E, Kim N, Kim IA, Kim TM, Kim YJ, et al. Novel recursive partitioning analysis classification for newly diagnosed glioblastoma: a multi-institutional study highlighting the MGMT promoter methylation and IDH1 gene mutation status. Radiother Oncol. 2017; 123:106–11.
Article6. Chinot OL, Wick W, Mason W, Henriksson R, Saran F, Nishikawa R, et al. Bevacizumab plus radiotherapy-temozolomide for newly diagnosed glioblastoma. N Engl J Med. 2014; 370:709–22.
Article7. Gilbert MR, Dignam JJ, Armstrong TS, Wefel JS, Blumenthal DT, Vogelbaum MA, et al. A randomized trial of bevacizumab for newly diagnosed glioblastoma. N Engl J Med. 2014; 370:699–708.
Article8. Stupp R, Taillibert S, Kanner AA, Kesari S, Steinberg DM, Toms SA, et al. Maintenance therapy with tumor-treating fields plus temozolomide vs temozolomide alone for glioblastoma: a randomized clinical trial. JAMA. 2015; 314:2535–43.9. Wright C, Moore RD. Disulfiram treatment of alcoholism. Am J Med. 1990; 88:647–55.
Article10. Yang EJ, Wu C, Liu Y, Lv J, Shim JS. Revisiting non-cancer drugs for cancer therapy. Curr Top Med Chem. 2016; 16:2144–55.11. Choi SA, Choi JW, Wang KC, Phi JH, Lee JY, Park KD, et al. Disulfiram modulates stemness and metabolism of brain tumor initiating cells in atypical teratoid/rhabdoid tumors. Neuro Oncol. 2015; 17:810–21.
Article12. Kast RE, Karpel-Massler G, Halatsch ME. CUSP9* treatment protocol for recurrent glioblastoma: aprepitant, artesunate, auranofin, captopril, celecoxib, disulfiram, itraconazole, ritonavir, sertraline augmenting continuous low dose temozolomide. Oncotarget. 2014; 5:8052–82.
Article13. Paranjpe A, Zhang R, Ali-Osman F, Bobustuc GC, Srivenugopal KS. Disulfiram is a direct and potent inhibitor of human O6-methylguanine-DNA methyltransferase (MGMT) in brain tumor cells and mouse brain and markedly increases the alkylating DNA damage. Carcinogenesis. 2014; 35:692–702.
Article14. Srivenugopal KS, Rawat A, Niture SK, Paranjpe A, Velu C, Venugopal SN, et al. Posttranslational regulation of O(6)-methylguanine-DNA methyltransferase (MGMT) and new opportunities for treatment of brain cancers. Mini Rev Med Chem. 2016; 16:455–64.15. Triscott J, Lee C, Hu K, Fotovati A, Berns R, Pambid M, et al. Disulfiram, a drug widely used to control alcoholism, suppresses the self-renewal of glioblastoma and over-rides resistance to temozolomide. Oncotarget. 2012; 3:1112–23.16. Lee YE, Choi SA, Kwack PA, Kim HJ, Kim IH, Wang KC, et al. Repositioning disulfiram as a radiosensitizer against atypical teratoid/rhabdoid tumor. Neuro Oncol. 2017; 19:1079–87.
Article17. Rae C, Tesson M, Babich JW, Boyd M, Sorensen A, Mairs RJ. The role of copper in disulfiram-induced toxicity and radiosensitization of cancer cells. J Nucl Med. 2013; 54:953–60.
Article18. Cong J, Wang Y, Zhang X, Zhang N, Liu L, Soukup K, et al. A novel chemoradiation targeting stem and nonstem pancreatic cancer cells by repurposing disulfiram. Cancer Lett. 2017; 409:9–19.
Article19. Kim JH, Moon SH, No M, Kim JJ, Choi EJ, Cho BJ, et al. Isotype-specific inhibition of histone deacetylases: identification of optimal targets for radiosensitization. Cancer Res Treat. 2016; 48:1130–40.
Article20. Fowler JF. The linear-quadratic formula and progress in fractionated radiotherapy. Br J Radiol. 1989; 62:679–94.
Article21. Kim JH, Kim IH, Shin JH, Kim HJ, Kim IA. Sequence-dependent radiosensitization of histone deacetylase inhibitors trichostatin A and SK-7041. Cancer Res Treat. 2013; 45:334–42.
Article22. Kim HJ, Kim JH, Chie EK, Young PD, Kim IA, Kim IH. DNMT (DNA methyltransferase) inhibitors radiosensitize human cancer cells by suppressing DNA repair activity. Radiat Oncol. 2012; 7:39.
Article23. Wick W, Steinbach JP, Platten M, Hartmann C, Wenz F, von Deimling A, et al. Enzastaurin before and concomitant with radiation therapy, followed by enzastaurin maintenance therapy, in patients with newly diagnosed glioblastoma without MGMT promoter hypermethylation. Neuro Oncol. 2013; 15:1405–12.
Article24. Nabors LB, Fink KL, Mikkelsen T, Grujicic D, Tarnawski R, Nam DH, et al. Two cilengitide regimens in combination with standard treatment for patients with newly diagnosed glioblastoma and unmethylated MGMT gene promoter: results of the open-label, controlled, randomized phase II CORE study. Neuro Oncol. 2015; 17:708–17.
Article25. Kaina B, Margison GP, Christmann M. Targeting O(6)-methylguanine-DNA methyltransferase with specific inhibitors as a strategy in cancer therapy. Cell Mol Life Sci. 2010; 67:3663–81.26. Blumenthal DT, Rankin C, Stelzer KJ, Spence AM, Sloan AE, Moore DF Jr, et al. A Phase III study of radiation therapy (RT) and O(6)-benzylguanine+BCNU versus RT and BCNU alone and methylation status in newly diagnosed glioblastoma and gliosarcoma: Southwest Oncology Group (SWOG) study S0001. Int J Clin Oncol. 2015; 20:650–8.27. Cvek B. Nonprofit drugs as the salvation of the world's healthcare systems: the case of Antabuse (disulfiram). Drug Discov Today. 2012; 17:409–12.
Article28. Kovic B, Xie F. Economic evaluation of bevacizumab for the first-line treatment of newly diagnosed glioblastoma multiforme. J Clin Oncol. 2015; 33:2296–302.
Article29. Bernard-Arnoux F, Lamure M, Ducray F, Aulagner G, Honnorat J, Armoiry X. The cost-effectiveness of tumor-treating fields therapy in patients with newly diagnosed glioblastoma. Neuro Oncol. 2016; 18:1129–36.
Article30. Huang J, Campian JL, Gujar AD, Tran DD, Lockhart AC, DeWees TA, et al. A phase I study to repurpose disulfiram in combination with temozolomide to treat newly diagnosed glioblastoma after chemoradiotherapy. J Neurooncol. 2016; 128:259–66.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Epileptic Seizure Due to Disulfiram Treatment
- Hypotension Caused by a Disulfiram-Alcohol Reaction
- The role of mitochondrial aldehyde dehydrogenase (ALDH) deficiency in Koreans with alcoholic liver disease
- Evaluation of the effects of disulfiram, an alcohol-aversive agent with anti-cancer activity, on mouse bone marrow cells
- Inhibitor of DNA-binding 4 contributes to the maintenance and expansion of cancer stem cells in 4T1 mouse mammary cancer cell line