Cancer Res Treat.
2019 Apr;51(2):632-648. 10.4143/crt.2018.060.
Effect of Estradiol in an Azoxymethane/Dextran Sulfate Sodium-Treated Mouse Model of Colorectal Cancer: Implication for Sex Difference in Colorectal Cancer Development
- Affiliations
-
- 1Seoul National University College of Medicine, Seoul, Korea.
- 2Department of Internal Medicine, Seoul National University Bundang Hospital, Seongnam, Korea. nayoungkim49@empas.com
- 3Department of Internal Medicine and Liver Research Institute, Seoul National University College of Medicine, Seoul, Korea.
- 4Tumor Microenvironment Global Core Research Center, Seoul National University College of Pharmacy, Seoul, Korea.
- 5Department of Pathology, Seoul National University Bundang Hospital, Seongnam, Korea.
- KMID: 2464410
- DOI: http://doi.org/10.4143/crt.2018.060
Abstract
- PURPOSE
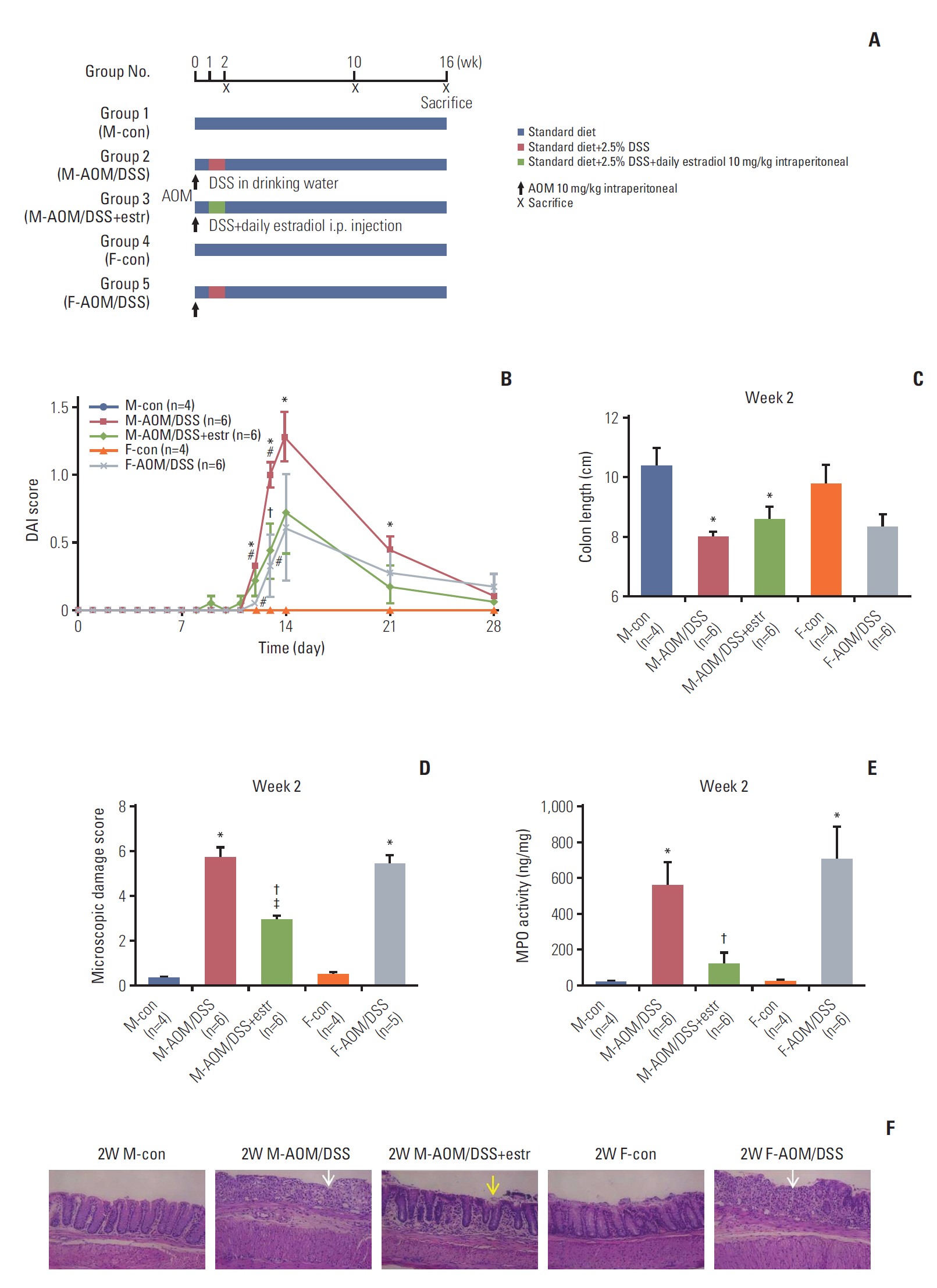
This study demonstrates that estradiol downregulates inflammation and inhibits colorectal cancer (CRC) development in azoxymethane/dextran sulfate sodium (AOM/DSS) mouse model.
MATERIALS AND METHODS
AOM/DSS-treated male and female mice were sacrificed at weeks 2, 10, and 16, to assess estrogen effects on colitis and carcinogenesis. Macroscopic and histologic severity of colitis and Western blot and quantitative real-time polymerase chain reaction were evaluated, to measure inflammatory mediators and cytokines.
RESULTS
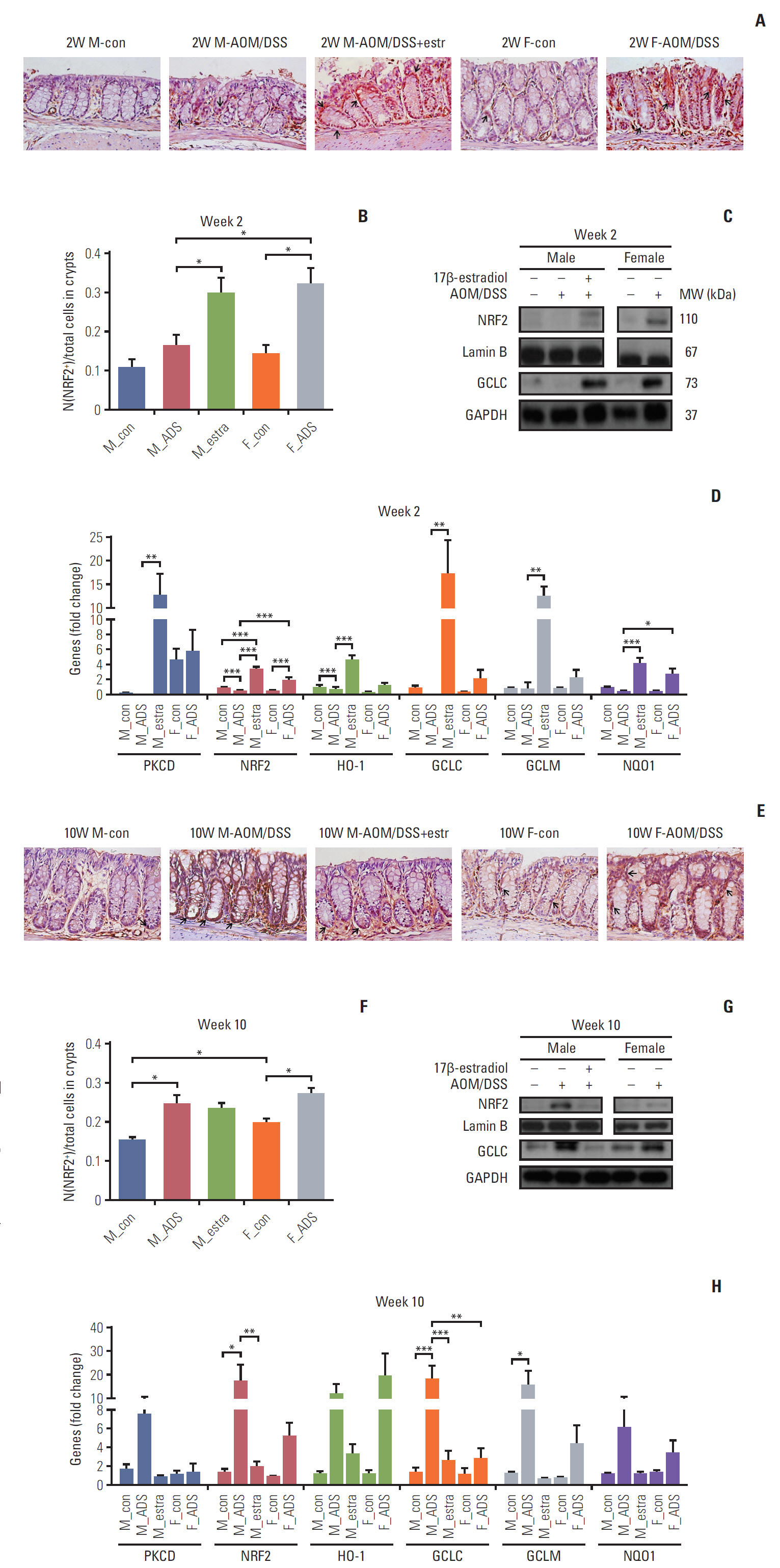
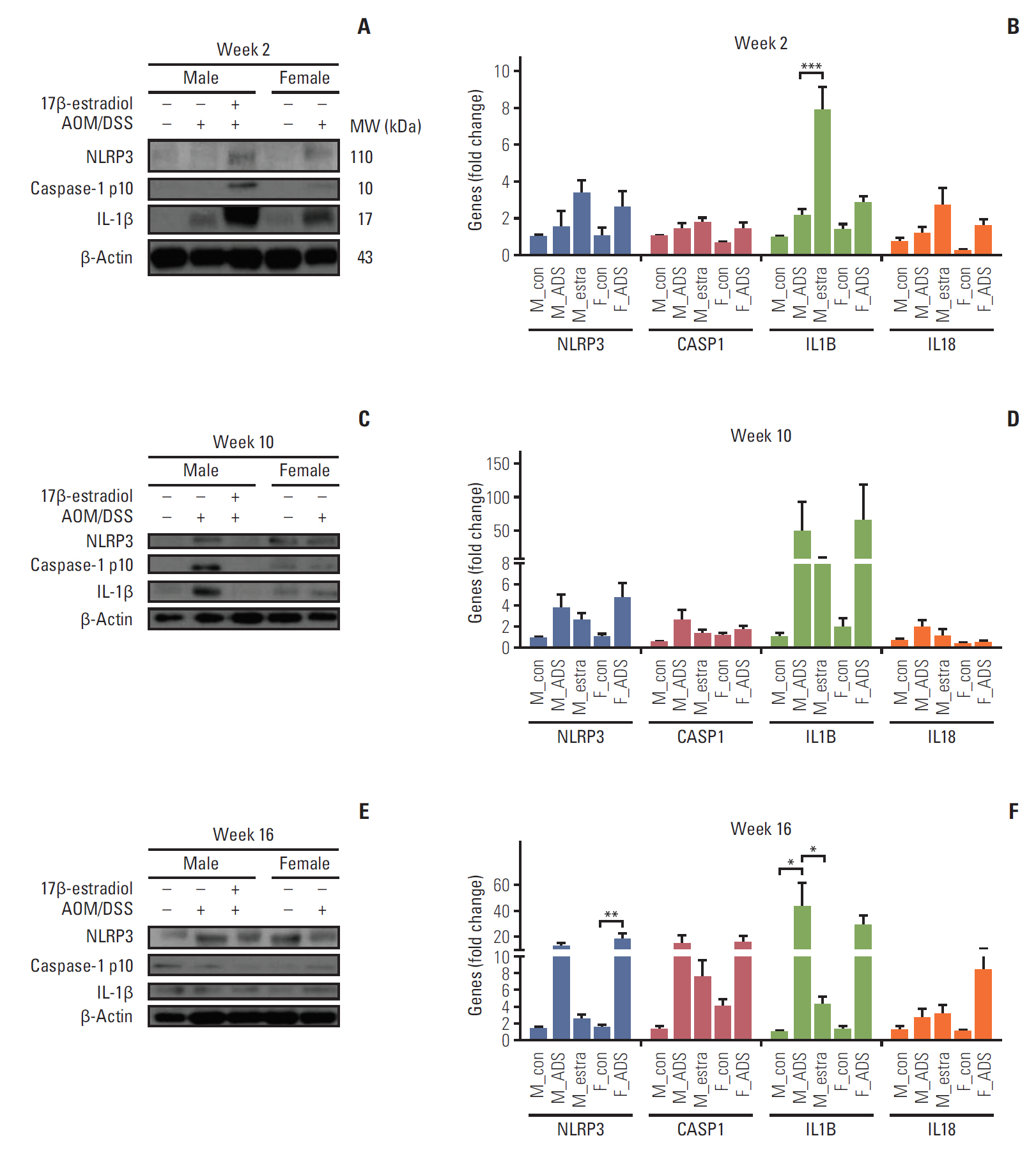
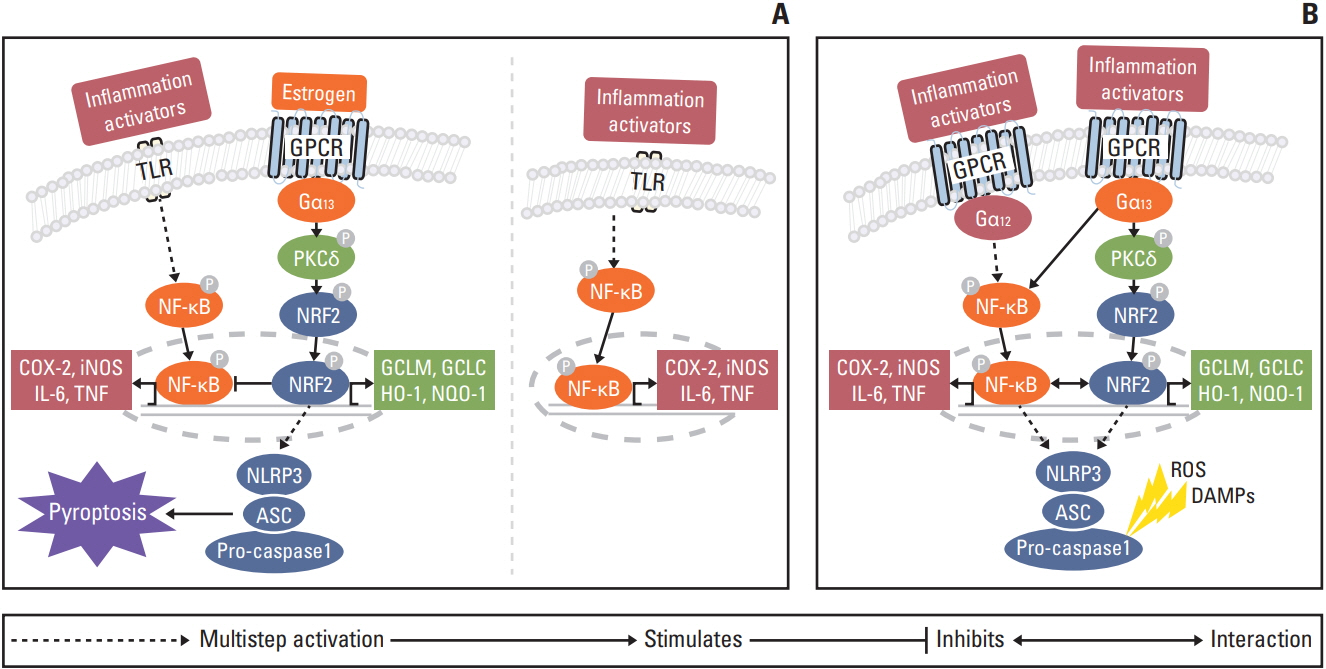
Compared with AOM/DSS-treated male mice (M-AOM/DSS group), AOM/DSS-treated male mice with estradiol administration (M-AOM/DSS+estr group) displayed at week 2 significantly decreased severity of colitis. At weeks 10 and 16, AOM/DSS-treated female mice (F-AOM/DSS group) and the M-AOM/DSS+estr group showed significantly lower tumor multiplicity compared with the M-AOM/DSS group. At week 2, F-AOM/DSS group had a lower level of nuclear factor-κB (NF-κB) expression and higher level of nuclear factor erythroid 2-related factor 2 (Nrf2) expression, compared to the M-AOM/DSS group. At week 2, expression levels of NF-κB and its related mediators decreased in the M-AOM/DSS+estr group, while levels of Nrf2 and Nrf2-related anti-oxidant enzymes increased. In addition, estradiol significantly increased Nod-like receptor protein 3 (NLRP3) inflammasome expressions in AOM/DSS-treated male mice. In contrast, at weeks 10 and 16, Nrf2 and its-related anti-oxidant enzymes and NLRP3 inflammasome were highly expressed in M-AOM/DSS group and in F-AOM/DSS group, who developed cancer.
CONCLUSION
The data suggest that estradiol inhibits the initiation of CRC by regulating Nrf2-related pathways. Moreover, these imply the dual role of Nrf2 and NLRP3 inflammasome, including promotion of tumor progression upon tumor initiation.
Keyword
MeSH Terms
-
Animals
Blotting, Western
Carcinogenesis
Colitis
Colorectal Neoplasms*
Cytokines
Estradiol*
Estrogens
Female
Humans
Inflammasomes
Inflammation
Male
Mice*
NF-E2-Related Factor 2
NF-kappa B
Real-Time Polymerase Chain Reaction
Sex Characteristics*
Sodium
Cytokines
Estradiol
Estrogens
Inflammasomes
NF-E2-Related Factor 2
NF-kappa B
Sodium
Figure
Cited by 2 articles
-
Sex- and Gender-related Issues of Gut Microbiota in Gastrointestinal Tract Diseases
Nayoung Kim
Korean J Gastroenterol. 2021;78(1):9-23. doi: 10.4166/kjg.2021.409.Anti–PD-L1 Antibody and/or 17β-Estradiol Treatment Induces Changes in the Gut Microbiome in MC38 Colon Tumor Model
Chin-Hee Song, Nayoung Kim, Ryoung Hee Nam, Soo In Choi, Jae Young Jang, Jina Choi, Ha-Na Lee
Cancer Res Treat. 2023;55(3):894-909. doi: 10.4143/crt.2022.1427.
Reference
-
References
1. Alteri R, Kramer J, Simpson S. Colorectal cancer facts and figures 2014-2016. Atlanta, GA: American Cancer Society;2014.2. Gierisch JM, Coeytaux RR, Urrutia RP, Havrilesky LJ, Moorman PG, Lowery WJ, et al. Oral contraceptive use and risk of breast, cervical, colorectal, and endometrial cancers: a systematic review. Cancer Epidemiol Biomarkers Prev. 2013; 22:1931–43.
Article3. Chlebowski RT, Wactawski-Wende J, Ritenbaugh C, Hubbell FA, Ascensao J, Rodabough RJ, et al. Estrogen plus progestin and colorectal cancer in postmenopausal women. N Engl J Med. 2004; 350:991–1004.
Article4. Weyant MJ, Carothers AM, Mahmoud NN, Bradlow HL, Remotti H, Bilinski RT, et al. Reciprocal expression of ERalpha and ERbeta is associated with estrogen-mediated modulation of intestinal tumorigenesis. Cancer Res. 2001; 61:2547–51.5. Amos-Landgraf JM, Heijmans J, Wielenga MC, Dunkin E, Krentz KJ, Clipson L, et al. Sex disparity in colonic adenomagenesis involves promotion by male hormones, not protection by female hormones. Proc Natl Acad Sci U S A. 2014; 111:16514–9.
Article6. Lee SM, Kim N, Son HJ, Park JH, Nam RH, Ham MH, et al. The effect of sex on the azoxymethane/dextran sulfate sodium-treated mice model of colon cancer. J Cancer Prev. 2016; 21:271–8.
Article7. Wu J, Williams D, Walter GA, Thompson WE, Sidell N. Estrogen increases Nrf2 activity through activation of the PI3K pathway in MCF-7 breast cancer cells. Exp Cell Res. 2014; 328:351–60.
Article8. Cho MK, Kim WD, Ki SH, Hwang JI, Choi S, Lee CH, et al. Role of Galpha12 and Galpha13 as novel switches for the activity of Nrf2, a key antioxidative transcription factor. Mol Cell Biol. 2007; 27:6195–208.9. Li T, Xiao X, Zhang J, Zhu Y, Hu Y, Zang J, et al. Age and sex differences in vascular responsiveness in healthy and trauma patients: contribution of estrogen receptor-mediated Rho kinase and PKC pathways. Am J Physiol Heart Circ Physiol. 2014; 306:H1105–15.
Article10. Wang Y, Zhu L, Kuokkanen S, Pollard JW. Activation of protein synthesis in mouse uterine epithelial cells by estradiol-17beta is mediated by a PKC-ERK1/2-mTOR signaling pathway. Proc Natl Acad Sci U S A. 2015; 112:E1382–91.11. O'Mahony F, Alzamora R, Chung HL, Thomas W, Harvey BJ. Genomic priming of the antisecretory response to estrogen in rat distal colon throughout the estrous cycle. Mol Endocrinol. 2009; 23:1885–99.12. Khor TO, Huang MT, Prawan A, Liu Y, Hao X, Yu S, et al. Increased susceptibility of Nrf2 knockout mice to colitis-associated colorectal cancer. Cancer Prev Res (Phila). 2008; 1:187–91.
Article13. Li W, Khor TO, Xu C, Shen G, Jeong WS, Yu S, et al. Activation of Nrf2-antioxidant signaling attenuates NFkappaB-inflammatory response and elicits apoptosis. Biochem Pharmacol. 2008; 76:1485–9.14. Niture SK, Khatri R, Jaiswal AK. Regulation of Nrf2-an update. Free Radic Biol Med. 2014; 66:36–44.
Article15. Zhao C, Gillette DD, Li X, Zhang Z, Wen H. Nuclear factor E2-related factor-2 (Nrf2) is required for NLRP3 and AIM2 inflammasome activation. J Biol Chem. 2014; 289:17020–9.
Article16. Miao EA, Rajan JV, Aderem A. Caspase-1-induced pyroptotic cell death. Immunol Rev. 2011; 243:206–14.
Article17. Kepp O, Galluzzi L, Zitvogel L, Kroemer G. Pyroptosis: a cell death modality of its kind? Eur J Immunol. 2010; 40:627–30.18. Saleiro D, Murillo G, Benya RV, Bissonnette M, Hart J, Mehta RG. Estrogen receptor-beta protects against colitis-associated neoplasia in mice. Int J Cancer. 2012; 131:2553–61.19. Choi YJ, Choi YJ, Kim N, Nam RH, Lee S, Lee HS, et al. Acai berries inhibit colon tumorigenesis in azoxymethane/dextran sulfate sodium-treated mice. Gut Liver. 2017; 11:243–52.20. Suzuki R, Kohno H, Sugie S, Tanaka T. Sequential observations on the occurrence of preneoplastic and neoplastic lesions in mouse colon treated with azoxymethane and dextran sodium sulfate. Cancer Sci. 2004; 95:721–7.
Article21. Lee SE, Yang H, Jeong SI, Jin YH, Park CS, Park YS. Induction of heme oxygenase-1 inhibits cell death in crotonaldehyde-stimulated HepG2 cells via the PKC-delta-p38-Nrf2 pathway. PLoS One. 2012; 7:e41676.22. Bratton MR, Antoon JW, Duong BN, Frigo DE, Tilghman S, Collins-Burow BM, et al. Galphao potentiates estrogen receptor alpha activity via the ERK signaling pathway. J Endocrinol. 2012; 214:45–54.23. Ki SH, Choi MJ, Lee CH, Kim SG. Galpha12 specifically regulates COX-2 induction by sphingosine 1-phosphate. Role for JNK-dependent ubiquitination and degradation of IkappaBalpha. J Biol Chem. 2007; 282:1938–47.24. DiDonato JA, Mercurio F, Karin M. NF-kappaB and the link between inflammation and cancer. Immunol Rev. 2012; 246:379–400.25. Kalaitzidis D, Gilmore TD. Transcription factor cross-talk: the estrogen receptor and NF-kappaB. Trends Endocrinol Metab. 2005; 16:46–52.26. Kim W, Lee HN, Jang JH, Kim SH, Lee YH, Hahn YI, et al. 15-Deoxy-delta(12,14)-prostaglandin J2 exerts proresolving effects through nuclear factor E2-related factor 2-induced expression of CD36 and heme oxygenase-1. Antioxid Redox Signal. 2017; 27:1412–31.27. Haisenleder DJ, Schoenfelder AH, Marcinko ES, Geddis LM, Marshall JC. Estimation of estradiol in mouse serum samples: evaluation of commercial estradiol immunoassays. Endocrinology. 2011; 152:4443–7.
Article28. Lee JY, Kim N, Kim YS, Nam RH, Ham MH, Lee HS, et al. Repeated water avoidance stress alters mucosal mast cell counts, interleukin-1beta levels with sex differences in the distal colon of Wistar rats. J Neurogastroenterol Motil. 2016; 22:694–704.29. Satoh H, Moriguchi T, Saigusa D, Baird L, Yu L, Rokutan H, et al. NRF2 intensifies host defense systems to prevent lung carcinogenesis, but after tumor initiation accelerates malignant cell growth. Cancer Res. 2016; 76:3088–96.
Article30. Fabbi M, Carbotti G, Ferrini S. Context-dependent role of IL-18 in cancer biology and counter-regulation by IL-18BP. J Leukoc Biol. 2015; 97:665–75.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Epithelial Cell-specific Deletion of Microsomal Prostaglandin E Synthase-1 Does Not Influence Colon Tumor Development in Mice
- Colorectal Cancer and Prostaglandin
- Effects of Selenium on Colon Carcinogenesis Induced by Azoxymethane and Dextran Sodium Sulfate in Mouse Model with High-Iron Diet
- Induction of colorectal carcinogenesis in the C57BL/6J and A/J mouse strains with a reduced DSS dose in the AOM/DSS model
- Chemopreventive effect of alpha-viniferin in azoxymethane-induced mouse colorectal tumor and Caco-2 cells