Nutr Res Pract.
2018 Feb;12(1):20-28. 10.4162/nrp.2018.12.1.20.
Anti-hyperglycemic effects and signaling mechanism of Perilla frutescens sprout extract
- Affiliations
-
- 1Jeonju AgroBio-Materials Institute, 111-27, Wonjangdong-gil, Deokjin-gu, Jeonju, Jeonbuk 54810, Korea. Seon02@jami.re.kr
- KMID: 2429015
- DOI: http://doi.org/10.4162/nrp.2018.12.1.20
Abstract
- BACKGROUND/OBJECTIVES
Perilla frutescens (L.) Britton var. (PF) sprout is a plant of the labiate family. We have previously reported the protective effects of PF sprout extract on cytokine-induced β-cell damage. However, the mechanism of action of the PF sprout extract in type 2 diabetes (T2DM) has not been investigated. The present study was designed to study the effects of PF sprout extract and signaling mechanisms in the T2DM mice model using C57BL/KsJ-db/db (db/db) mice.
MATERIALS/METHODS
Male db/db mice were orally administered PF sprout extract (100, 300, and 1,000 mg/kg of body weight) or rosiglitazone (RGZ, positive drug, 1 mg/kg of body weight) for 4 weeks. Signaling mechanisms were analyzed using liver tissues and HepG2 cells.
RESULTS
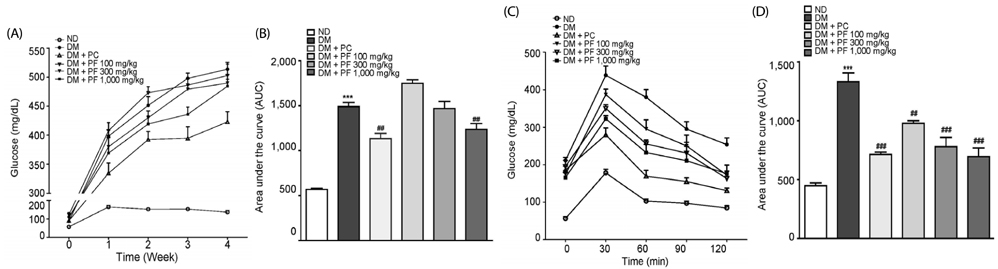
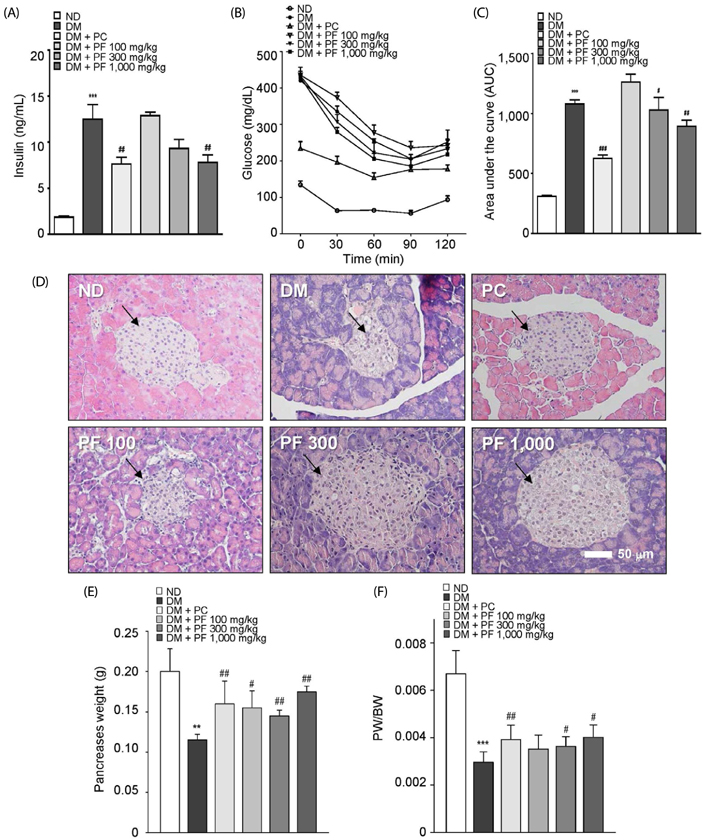
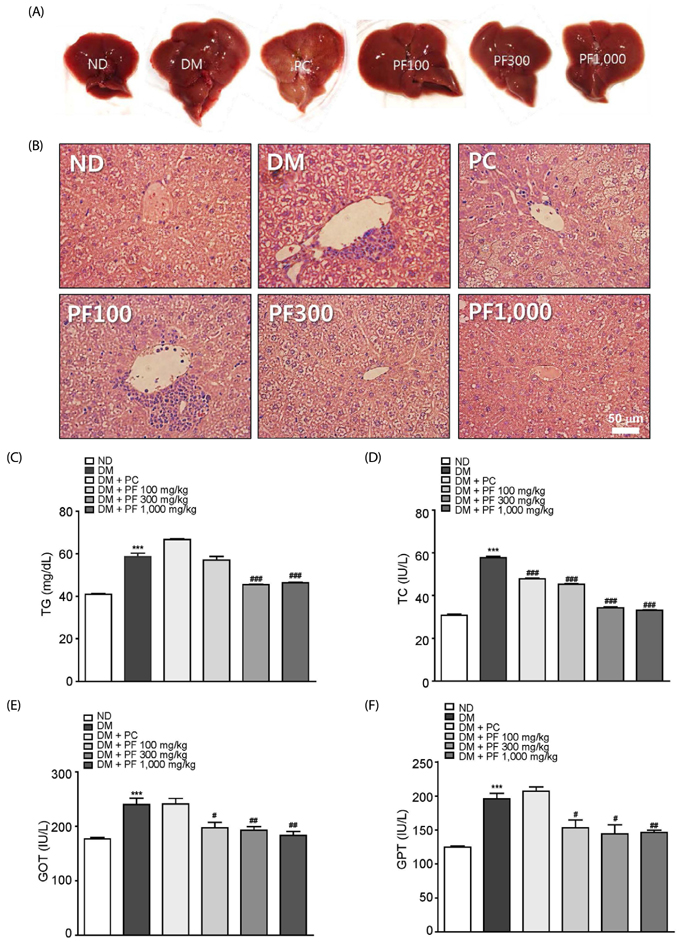
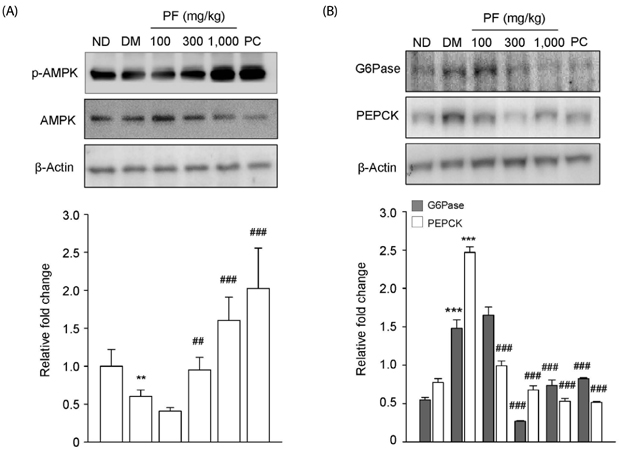
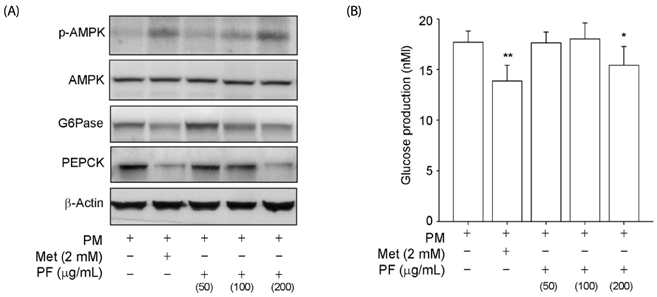
The PF sprout extract (300 and 1,000 mg/kg) significantly reduced the fasting blood glucose, serum insulin, triglyceride and total cholesterol levels in db/db mice. PF sprout extract also significantly improved glucose intolerance and insulin sensitivity, decreased hepatic gluconeogenic protein expression, and ameliorated histological alterations of the pancreas and liver. Levels of phosphorylated AMP-activated protein kinase (AMPK) protein expression also increased in the liver after treatment with the extract. In addition, an increase in the phosphorylation of AMPK and decrease in the phosphoenolpyruvate carboxykinase and glucose 6-phosphatase proteins in HepG2 cells were also observed.
CONCLUSIONS
Our results sugges that PF sprout displays beneficial effects in the prevention and treatment of type 2 diabetes via modulation of the AMPK pathway and inhibition of gluconeogenesis in the liver.
MeSH Terms
-
AMP-Activated Protein Kinases
Animals
Blood Glucose
Cholesterol
Diabetes Mellitus
Fasting
Gluconeogenesis
Glucose Intolerance
Glucose-6-Phosphatase
Hep G2 Cells
Humans
Insulin
Insulin Resistance
Liver
Male
Mice
Pancreas
Perilla frutescens*
Perilla*
Phosphoenolpyruvate
Phosphorylation
Plants
Triglycerides
AMP-Activated Protein Kinases
Blood Glucose
Cholesterol
Glucose-6-Phosphatase
Insulin
Phosphoenolpyruvate
Figure
Reference
-
1. Rahier J, Guiot Y, Goebbels RM, Sempoux C, Henquin JC. Pancreatic beta-cell mass in European subjects with type 2 diabetes. Diabetes Obes Metab. 2008; 10:Suppl 4. 32–42.2. Yki Järvinen H. Glucose toxicity. Endocr Rev. 1992; 13:415–431.
Article3. Bae UJ, Park SH, Jung SY, Park BH, Chae SW. Hypoglycemic effects of aqueous persimmon leaf extract in a murine model of diabetes. Mol Med Rep. 2015; 12:2547–2554.
Article4. Sen S, Querques MA, Chakrabarti S. North American Ginseng (Panax quinquefolius) prevents hyperglycemia and associated pancreatic abnormalities in diabetes. J Med Food. 2013; 16:587–592.
Article5. Hsu YJ, Lee TH, Chang CL, Huang YT, Yang WC. Anti-hyperglycemic effects and mechanism of Bidens pilosa water extract. J Ethnopharmacol. 2009; 122:379–383.
Article6. Nyarko AK, Asare-Anane H, Ofosuhene M, Addy ME. Extract of Ocimum canum lowers blood glucose and facilitates insulin release by isolated pancreatic beta-islet cells. Phytomedicine. 2002; 9:346–351.
Article7. Zheng J, Woo SL, Hu X, Botchlett R, Chen L, Huo Y, Wu C. Metformin and metabolic diseases: a focus on hepatic aspects. Front Med. 2015; 9:173–186.
Article8. Derosa G, Maffioli P. α-Glucosidase inhibitors and their use in clinical practice. Arch Med Sci. 2012; 8:899–906.9. Molavi B, Rassouli N, Bagwe S, Rasouli N. A review of thiazolidinediones and metformin in the treatment of type 2 diabetes with focus on cardiovascular complications. Vasc Health Risk Manag. 2007; 3:967–973.10. Inzucchi SE. Oral antihyperglycemic therapy for type 2 diabetes: scientific review. JAMA. 2002; 287:360–372.11. Vidal-Puig A, O'Rahilly S. Metabolism. Controlling the glucose factory. Nature. 2001; 413:125–126.12. Mihaylova MM, Shaw RJ. The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol. 2011; 13:1016–1023.
Article13. Kurita N, Koike S. Synergistic antimicrobial effect of acetic acid, sodium chloride and essential oil components. Agric Biol Chem. 1982; 46:1655–1660.
Article14. Ueda H, Yamazaki C, Yamazaki M. Inhibitory effect of Perilla leaf extract and luteolin on mouse skin tumor promotion. Biol Pharm Bull. 2003; 26:560–563.
Article15. Makino T, Furuta Y, Wakushima H, Fujii H, Saito K, Kano Y. Anti-allergic effect of Perilla frutescens and its active constituents. Phytother Res. 2003; 17:240–243.16. Banno N, Akihisa T, Tokuda H, Yasukawa K, Higashihara H, Ukiya M, Watanabe K, Kimura Y, Hasegawa J, Nishino H. Triterpene acids from the leaves of Perilla frutescens and their anti-inflammatory and antitumor-promoting effects. Biosci Biotechnol Biochem. 2004; 68:85–90.
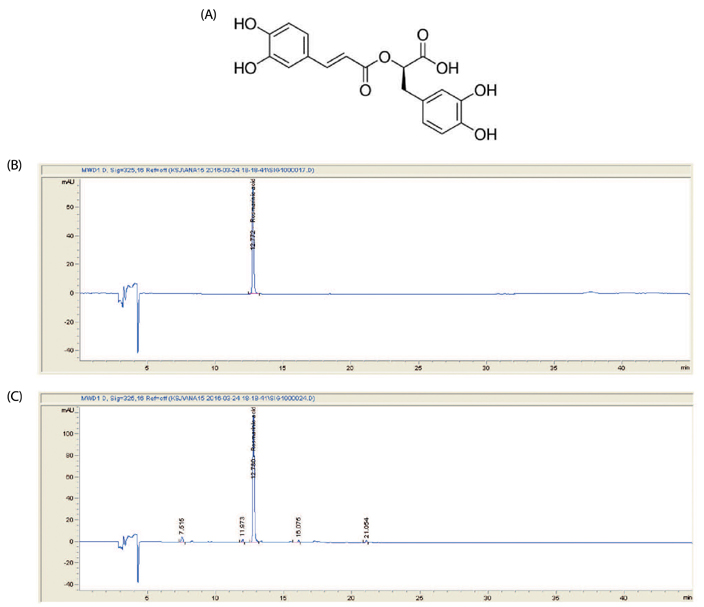
Article17. Paśko P, Sajewicz M, Gorinstein S, Zachwieja Z. Analysis of selected phenolic acids and flavonoids in Amaranthus cruentus and Chenopodium quinoa seeds and sprouts by HPLC. Acta Chromatogr. 2008; 20:661–672.
Article18. Lucarini R, Bernardes WA, Ferreira DS, Tozatti MG, Furtado R, Bastos JK, Pauletti PM, Januário AH, Silva ML, Cunha WR. In vivo analgesic and anti-inflammatory activities of Rosmarinus officinalis aqueous extracts, rosmarinic acid and its acetyl ester derivative. Pharm Biol. 2013; 51:1087–1090.
Article19. Karthikkumar V, Sivagami G, Vinothkumar R, Rajkumar D, Nalini N. Modulatory efficacy of rosmarinic acid on premalignant lesions and antioxidant status in 1,2-dimethylhydrazine induced rat colon carcinogenesis. Environ Toxicol Pharmacol. 2012; 34:949–958.
Article20. Pereira P, Tysca D, Oliveira P, da Silva Brum LF, Picada JN, Ardenghi P. Neurobehavioral and genotoxic aspects of rosmarinic acid. Pharmacol Res. 2005; 52:199–203.
Article21. Runtuwene J, Cheng KC, Asakawa A, Amitani H, Amitani M, Morinaga A, Takimoto Y, Kairupan BH, Inui A. Rosmarinic acid ameliorates hyperglycemia and insulin sensitivity in diabetic rats, potentially by modulating the expression of PEPCK and GLUT4. Drug Des Devel Ther. 2016; 10:2193–2202.22. Collins QF, Liu HY, Pi J, Liu Z, Quon MJ, Cao W. Epigallocatechin-3-gallate (EGCG), a green tea polyphenol, suppresses hepatic gluconeogenesis through 5'-AMP-activated protein kinase. J Biol Chem. 2007; 282:30143–30149.
Article23. Govindaraj J, Sorimuthu Pillai S. Rosmarinic acid modulates the antioxidant status and protects pancreatic tissues from glucolipotoxicity mediated oxidative stress in high-fat diet: streptozotocininduced diabetic rats. Mol Cell Biochem. 2015; 404:143–159.
Article24. Kwon YI, Vattem DA, Shetty K. Evaluation of clonal herbs of Lamiaceae species for management of diabetes and hypertension. Asia Pac J Clin Nutr. 2006; 15:107–118.25. Valenti L, Bugianesi E, Pajvani U, Targher G. Nonalcoholic fatty liver disease: cause or consequence of type 2 diabetes? Liver Int. 2016; 36:1563–1579.
Article26. Giandalia A, Romeo EL, Ruffo MC, Muscianisi M, Giorgianni L, Forte F, Cucinotta D, Russo GT. Clinical correlates of persistently elevated liver enzymes in type 2 diabetic outpatients. Prim Care Diabetes. 2017; 11:226–232.
Article27. Huang M, Zhao P, Xiong M, Zhou Q, Zheng S, Ma X, Xu C, Yang J, Yang X, Zhang TC. Antidiabetic activity of perylenequinonoid-rich extract from Shiraia bambusicola in KK-Ay mice with spontaneous type 2 diabetes mellitus. J Ethnopharmacol. 2016; 191:71–81.
Article28. Sanaei M, Ebrahimi M, Banazadeh Z, Shafiee G, Khatami F, Ahadi Z, Heshmat R. Consequences of AphanizomenonFlos-aquae(AFA) extract (Stemtech (TM) ) on metabolic profile of patients with type 2 diabetes. J Diabetes Metab Disord. 2015; 14:50.
Article29. Koo , Flechner L, Qi L, Zhang X, Screaton RA, Jeffries S, Hedrick S, Xu W, Boussouar F, Brindle P, Takemori H, Montminy M. The CREB coactivator TORC2 is a key regulator of fasting glucose metabolism. Nature. 2005; 437:1109–1111.
Article30. Lochhead PA, Salt IP, Walker KS, Hardie DG, Sutherland C. 5-aminoimidazole-4-carboxamide riboside mimics the effects of insulin on the expression of the 2 key gluconeogenic genes PEPCK and glucose-6-phosphatase. Diabetes. 2000; 49:896–903.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Anti-Helicobacter pylori activity of acomplex mixture of Lactobacillus paracasei HP7 including the extract of Perilla frutescens var. acuta and Glycyrrhiza glabra
- The Neuro-Protective Effect of the Methanolic Extract of Perilla frutescens var. japonica and Rosmarinic Acid against H2O2-Induced Oxidative Stress in C6 Glial Cells
- Inhibition of Proinflammatory Cytokine Generation in Lung Inflammation by the Leaves of Perilla frutescens and Its Constituents
- Inhibitory Effects on Oral Microbial Activity and Production of Lipopolysaccharides-Induced Pro-Inflammatory Mediators in Raw264.7 Macrophages of Ethanol Extract of Perilla flutescens (L.) Britton
- Inhibitory activities of Perilla frutescens britton leaf extract against the growth, migration, and adhesion of human cancer cells