J Periodontal Implant Sci.
2018 Oct;48(5):284-294. 10.5051/jpis.2018.48.5.284.
Estrogen reinforces barrier formation and protects against tumor necrosis factor alpha-induced barrier dysfunction in oral epithelial cells
- Affiliations
-
- 1Department of Oral Microbiology and Immunology, Dental Research Institute, Seoul National University School of Dentistry, Seoul, Korea. youngnim@snu.ac.kr
- KMID: 2424862
- DOI: http://doi.org/10.5051/jpis.2018.48.5.284
Abstract
- PURPOSE
Epithelial barrier dysfunction is involved in the pathophysiology of periodontitis and oral lichen planus. Estrogens have been shown to enhance the physical barrier function of intestinal and esophageal epithelia, and we aimed to investigate the effect of estradiol (E2) on the regulation of physical barrier and tight junction (TJ) proteins in human oral epithelial cell monolayers.
METHODS
HOK-16B cell monolayers cultured on transwells were treated with E2, an estrogen receptor (ER) antagonist (ICI 182,780), tumor necrosis factor alpha (TNFα), or dexamethasone (Dexa), and the transepithelial electrical resistance (TER) was then measured. Cell proliferation was measured by the cell counting kit (CCK)-8 assay. The levels of TJ proteins and nuclear translocation of nuclear factor (NF)-κB were examined by confocal microscopy.
RESULTS
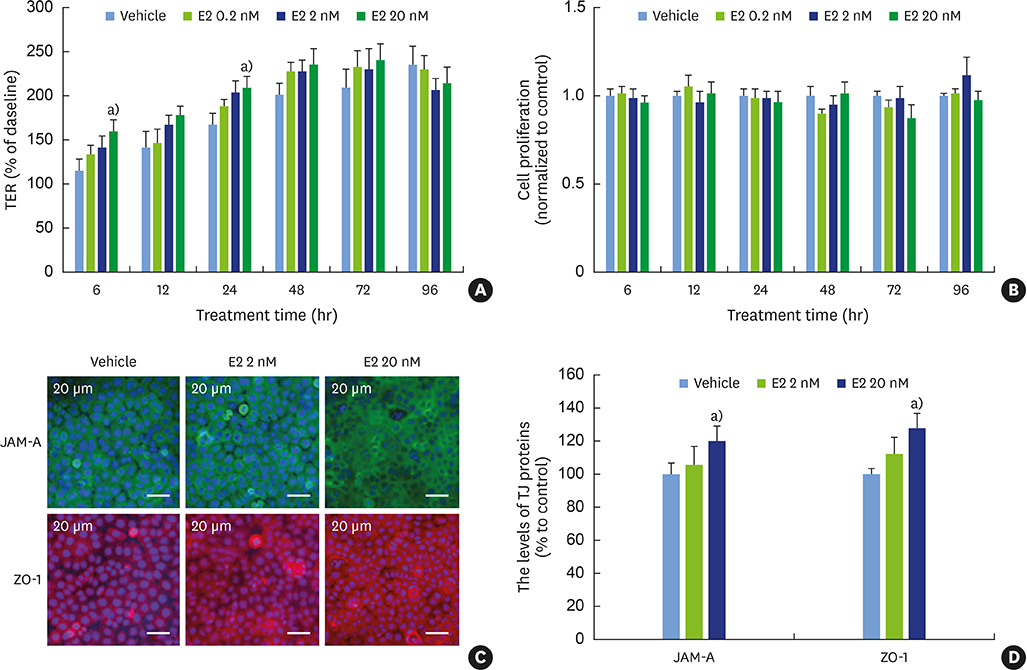
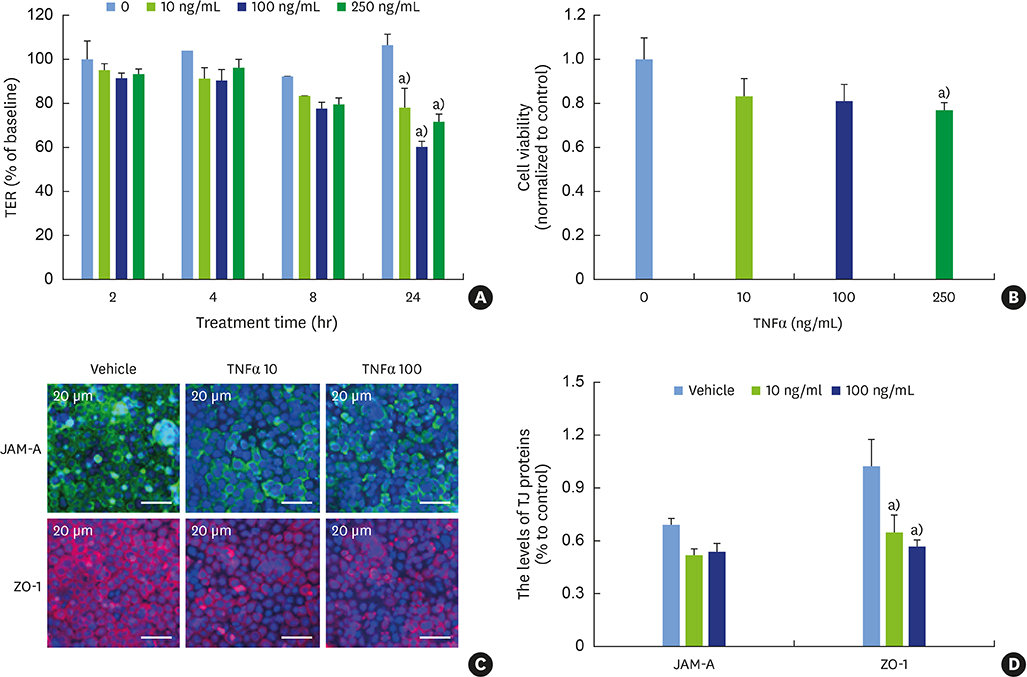
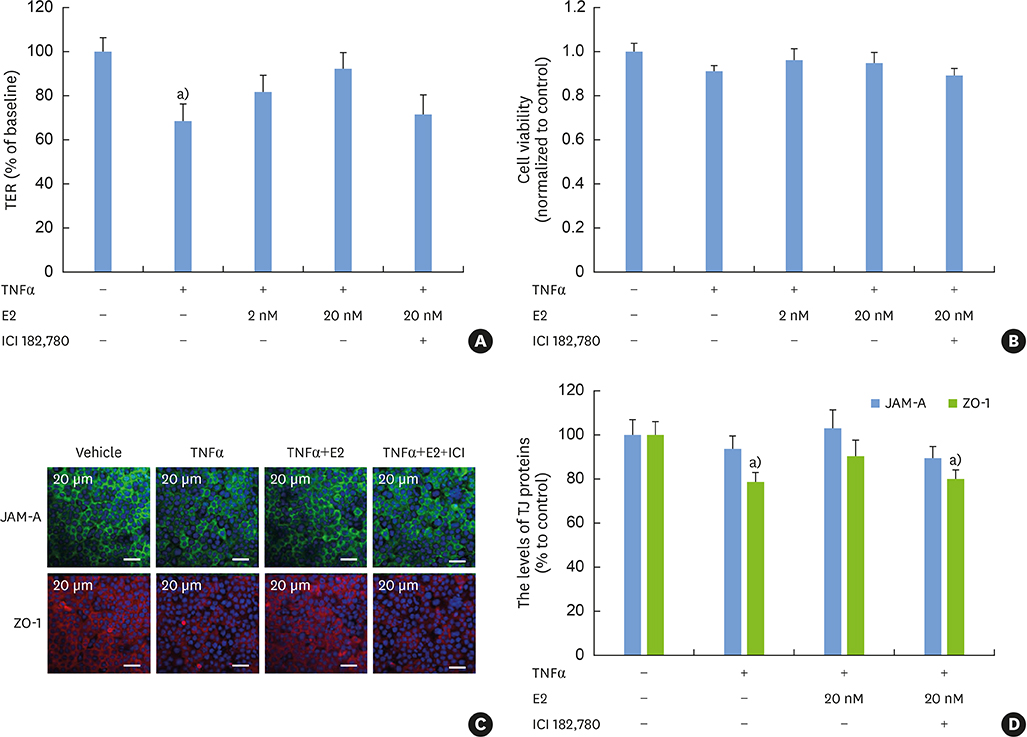
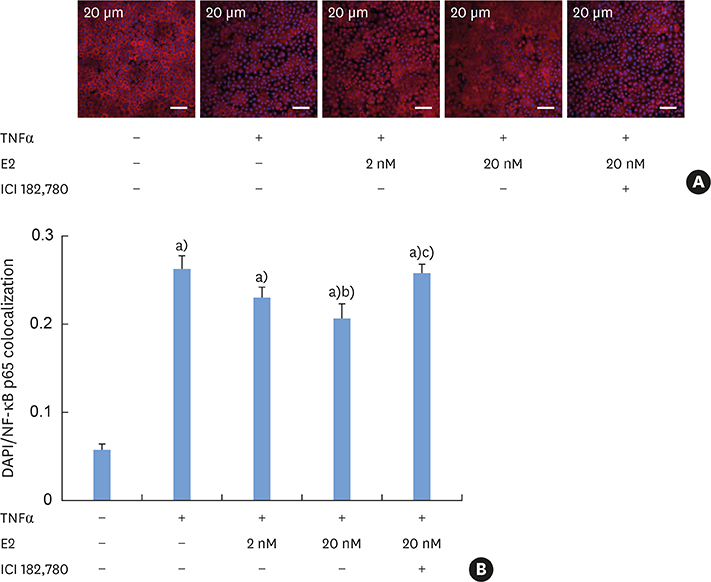
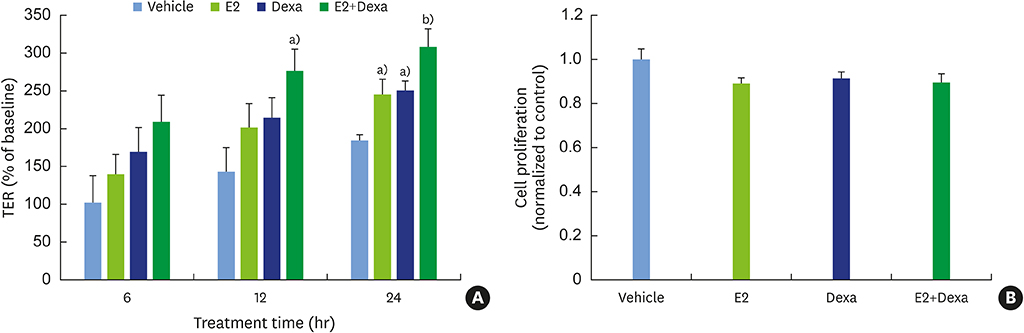
E2 treatment increased the TER and the levels of junctional adhesion molecule (JAM)-A and zonula occludens (ZO)-1 in a dose-dependent manner, without affecting cell proliferation during barrier formation. Treatment of the tight-junctioned cell monolayers with TNFα induced decreases in the TER and the levels of ZO-1 and nuclear translocation of NF-κB. These TNFα-induced changes were inhibited by E2, and this effect was completely reversed by co-treatment with ICI 182,780. Furthermore, E2 and Dexa presented an additive effect on the epithelial barrier function.
CONCLUSIONS
E2 reinforces the physical barrier of oral epithelial cells through the nuclear ER-dependent upregulation of TJ proteins. The protective effect of E2 on the TNFα-induced impairment of the epithelial barrier and its additive effect with Dexa suggest its potential use to treat oral inflammatory diseases involving epithelial barrier dysfunction.
MeSH Terms
-
Architectural Accessibility
Cell Count
Cell Proliferation
Dexamethasone
Electric Impedance
Epithelial Cells*
Estradiol
Estrogens*
Humans
Junctional Adhesion Molecule A
Junctional Adhesion Molecules
Lichen Planus, Oral
Microscopy, Confocal
NF-kappa B
Periodontitis
Tight Junctions
Tumor Necrosis Factor-alpha*
Up-Regulation
Dexamethasone
Estradiol
Estrogens
Junctional Adhesion Molecule A
Junctional Adhesion Molecules
NF-kappa B
Tumor Necrosis Factor-alpha
Figure
Cited by 1 articles
-
Vitamin D maintains E-cadherin intercellular junctions by downregulating MMP-9 production in human gingival keratinocytes treated by TNF-α
Changseok Oh, Hyun Jung Kim, Hyun-Man Kim
J Periodontal Implant Sci. 2019;49(5):270-286. doi: 10.5051/jpis.2019.49.5.270.
Reference
-
1. Ivanov AI, Parkos CA, Nusrat A. Cytoskeletal regulation of epithelial barrier function during inflammation. Am J Pathol. 2010; 177:512–524.
Article2. Belibasakis GN, Kast JI, Thurnheer T, Akdis CA, Bostanci N. The expression of gingival epithelial junctions in response to subgingival biofilms. Virulence. 2015; 6:704–709.
Article3. Brunner PM, Leung DY, Guttman-Yassky E. Immunologic, microbial, and epithelial interactions in atopic dermatitis. Ann Allergy Asthma Immunol. 2018; 120:34–41.
Article4. Turner JR. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol. 2009; 9:799–809.
Article5. Khalmuratova R, Park JW, Shin HW. Immune cell responses and mucosal barrier disruptions in chronic rhinosinusitis. Immune Netw. 2017; 17:60–67.
Article6. Swindle EJ, Collins JE, Davies DE. Breakdown in epithelial barrier function in patients with asthma: identification of novel therapeutic approaches. J Allergy Clin Immunol. 2009; 124:23–34.
Article7. Choi YS, Kim YC, Jo AR, Ji S, Koo KT, Ko Y, et al. P. gingivalis and dextran sulfate sodium induce periodontitis through the disruption of physical barriers in mice. Eur J Inflamm. 2013; 11:419–431.
Article8. Choi YS, Kim Y, Yoon HJ, Baek KJ, Alam J, Park HK, et al. The presence of bacteria within tissue provides insights into the pathogenesis of oral lichen planus. Sci Rep. 2016; 6:29186.
Article9. Katz J, Yang QB, Zhang P, Potempa J, Travis J, Michalek SM, et al. Hydrolysis of epithelial junctional proteins by P. gingivalis gingipains. Infect Immun. 2002; 70:2512–2518.
Article10. Ellen RP, Ko KS, Lo CM, Grove DA, Ishihara K. Insertional inactivation of the prtP gene of Treponema denticola confirms dentilisin's disruption of epithelial junctions. J Mol Microbiol Biotechnol. 2000; 2:518–516.11. Damek-Poprawa M, Korostoff J, Gill R, DiRienzo JM. Cell junction remodeling in gingival tissue exposed to a microbial toxin. J Dent Res. 2013; 92:518–523.
Article12. Capaldo CT, Nusrat A. Cytokine regulation of tight junctions. Biochim Biophys Acta. 2009; 1788:864–871.
Article13. Takano K, Kojima T, Sawada N, Himi T. Role of tight junctions in signal transduction: an update. EXCLI J. 2014; 13:1145–1162.14. Cui J, Shen Y, Li R. Estrogen synthesis and signaling pathways during aging: from periphery to brain. Trends Mol Med. 2013; 19:197–209.
Article15. Looijer-van Langen M, Hotte N, Dieleman LA, Albert E, Mulder C, Madsen KL. Estrogen receptor-β signaling modulates epithelial barrier function. Am J Physiol Gastrointest Liver Physiol. 2011; 300:G621–G626.
Article16. Braniste V, Leveque M, Buisson-Brenac C, Bueno L, Fioramonti J, Houdeau E. Oestradiol decreases colonic permeability through oestrogen receptor beta-mediated up-regulation of occludin and junctional adhesion molecule-A in epithelial cells. J Physiol. 2009; 587:3317–3328.
Article17. Honda J, Iijima K, Asanuma K, Ara N, Shiroki T, Kondo Y, et al. Estrogen enhances esophageal barrier function by potentiating occludin expression. Dig Dis Sci. 2016; 61:1028–1038.
Article18. Välimaa H, Savolainen S, Soukka T, Silvoniemi P, Mäkelä S, Kujari H, et al. Estrogen receptor-beta is the predominant estrogen receptor subtype in human oral epithelium and salivary glands. J Endocrinol. 2004; 180:55–62.
Article19. Park NH, Min BM, Li SL, Huang MZ, Cherick HM, Doniger J. Immortalization of normal human oral keratinocytes with type 16 human papillomavirus. Carcinogenesis. 1991; 12:1627–1631.
Article20. Fujita T, Yumoto H, Shiba H, Ouhara K, Miyagawa T, Nagahara T, et al. Irsogladine maleate regulates epithelial barrier function in tumor necrosis factor-α-stimulated human gingival epithelial cells. J Periodontal Res. 2012; 47:55–61.
Article21. Kielgast F, Schmidt H, Braubach P, Winkelmann VE, Thompson KE, Frick M, et al. Glucocorticoids regulate tight junction permeability of lung epithelia by modulating claudin 8. Am J Respir Cell Mol Biol. 2016; 54:707–717.
Article22. Ye D, Ma I, Ma TY. Molecular mechanism of tumor necrosis factor-alpha modulation of intestinal epithelial tight junction barrier. Am J Physiol Gastrointest Liver Physiol. 2006; 290:G496–G504.23. Ma TY, Iwamoto GK, Hoa NT, Akotia V, Pedram A, Boivin MA, et al. TNF-alpha-induced increase in intestinal epithelial tight junction permeability requires NF-kappa B activation. Am J Physiol Gastrointest Liver Physiol. 2004; 286:G367–G376.24. Ma TY, Boivin MA, Ye D, Pedram A, Said HM. Mechanism of TNF-α modulation of Caco-2 intestinal epithelial tight junction barrier: role of myosin light-chain kinase protein expression. Am J Physiol Gastrointest Liver Physiol. 2005; 288:G422–G430.
Article25. Sun WH, Keller ET, Stebler BS, Ershler WB. Estrogen inhibits phorbol ester-induced I kappa B alpha transcription and protein degradation. Biochem Biophys Res Commun. 1998; 244:691–695.
Article26. Wen Y, Yang S, Liu R, Perez E, Yi KD, Koulen P, et al. Estrogen attenuates nuclear factor-kappa B activation induced by transient cerebral ischemia. Brain Res. 2004; 1008:147–154.
Article27. Hsu SM, Chen YC, Jiang MC. 17 beta-estradiol inhibits tumor necrosis factor-alpha-induced nuclear factor-kappa B activation by increasing nuclear factor-kappa B p105 level in MCF-7 breast cancer cells. Biochem Biophys Res Commun. 2000; 279:47–52.
Article28. Quaedackers ME, van den Brink CE, van der Saag PT, Tertoolen LG. Direct interaction between estrogen receptor alpha and NF-kappaB in the nucleus of living cells. Mol Cell Endocrinol. 2007; 273:42–50.
Article29. Pelzer T, Neumann M, de Jager T, Jazbutyte V, Neyses L. Estrogen effects in the myocardium: inhibition of NF-kappaB DNA binding by estrogen receptor-alpha and -beta. Biochem Biophys Res Commun. 2001; 286:1153–1157.
Article30. Kalaitzidis D, Gilmore TD. Transcription factor cross-talk: the estrogen receptor and NF-kappaB. Trends Endocrinol Metab. 2005; 16:46–52.31. Ghisletti S, Meda C, Maggi A, Vegeto E. 17beta-estradiol inhibits inflammatory gene expression by controlling NF-kappaB intracellular localization. Mol Cell Biol. 2005; 25:2957–2968.
Article32. Schaefer TM, Wright JA, Pioli PA, Wira CR. IL-1beta-mediated proinflammatory responses are inhibited by estradiol via down-regulation of IL-1 receptor type I in uterine epithelial cells. J Immunol. 2005; 175:6509–6516.
Article33. Vegeto E, Belcredito S, Etteri S, Ghisletti S, Brusadelli A, Meda C, et al. Estrogen receptor-alpha mediates the brain antiinflammatory activity of estradiol. Proc Natl Acad Sci U S A. 2003; 100:9614–9619.
Article34. Miyagawa T, Fujita T, Yumoto H, Yoshimoto T, Kajiya M, Ouhara K, et al. Azithromycin recovers reductions in barrier function in human gingival epithelial cells stimulated with tumor necrosis factor-α. Arch Oral Biol. 2016; 62:64–69.
Article35. Tsai PW, Cheng YL, Hsieh WP, Lan CY. Responses of Candida albicans to the human antimicrobial peptide LL-37. J Microbiol. 2014; 52:581–589.36. Kulkarni NN, Gunnarsson HI, Yi Z, Gudmundsdottir S, Sigurjonsson OE, Agerberth B, et al. Glucocorticoid dexamethasone down-regulates basal and vitamin D3 induced cathelicidin expression in human monocytes and bronchial epithelial cell line. Immunobiology. 2016; 221:245–252.
Article37. Park K, Kim YI, Shin KO, Seo HS, Kim JY, Mann T, et al. The dietary ingredient, genistein, stimulates cathelicidin antimicrobial peptide expression through a novel S1P-dependent mechanism. J Nutr Biochem. 2014; 25:734–740.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Reversibility of Ultrastructural Changes of Cultured Human Nasal Epithelial Cells Induced by Tumor Necrosis Factor-alpha
- Berberine Prevents Intestinal Mucosal Barrier Damage During Early Phase of Sepsis in Rat through the Toll-Like Receptors Signaling Pathway
- EphrinA1/EphA2 Promotes Epithelial Hyperpermeability Involving in Lipopolysaccharide-induced Intestinal Barrier Dysfunction
- The Ultrastructural Development of Air-Blood Barrier in Human Fetal Lung
- Effects of Curcumin and Genistein on Phorbol Ester or Tumor Necrosis Factor-alpha-Induced Mucin Production from Human Airway Epithelial Cells