J Korean Neurosurg Soc.
2018 Sep;61(5):548-558. 10.3340/jkns.2017.0200.
S100ß, Matrix Metalloproteinase-9, D-dimer, and Heat Shock Protein 70 Are Serologic Biomarkers of Acute Cerebral Infarction in a Mouse Model of Transient MCA Occlusion
- Affiliations
-
- 1Department of Neurosurgery, Hallym University Kangnam Sacred Heart Hospital, Hallym University College of Medicine, Seoul, Korea.
- 2Department of Neurosurgery, Korea University Medical Center, Seoul, Korea. sungkha@yahoo.com
- KMID: 2420062
- DOI: http://doi.org/10.3340/jkns.2017.0200
Abstract
OBJECTIVE
Diagnosing acute cerebral infarction is crucial in determining prognosis of stroke patients. Although many serologic tests for prompt diagnosis are available, the clinical application of serologic tests is currently limited. We investigated whether S100β, matrix metalloproteinase-9 (MMP-9), D-dimer, and heat shock protein 70 (HSP70) can be used as biomarkers for acute cerebral infarction.
METHODS
Focal cerebral ischemia was induced using the modified intraluminal filament technique. Mice were randomly assigned to 30-minute occlusion (n=10), 60-minute occlusion (n=10), or sham (n=5) groups. Four hours later, neurological deficits were evaluated and blood samples were obtained. Infarction volumes were calculated and plasma S100β, MMP-9, D-dimer, and HSP70 levels were measured using enzyme-linked immunosorbent assay.
RESULTS
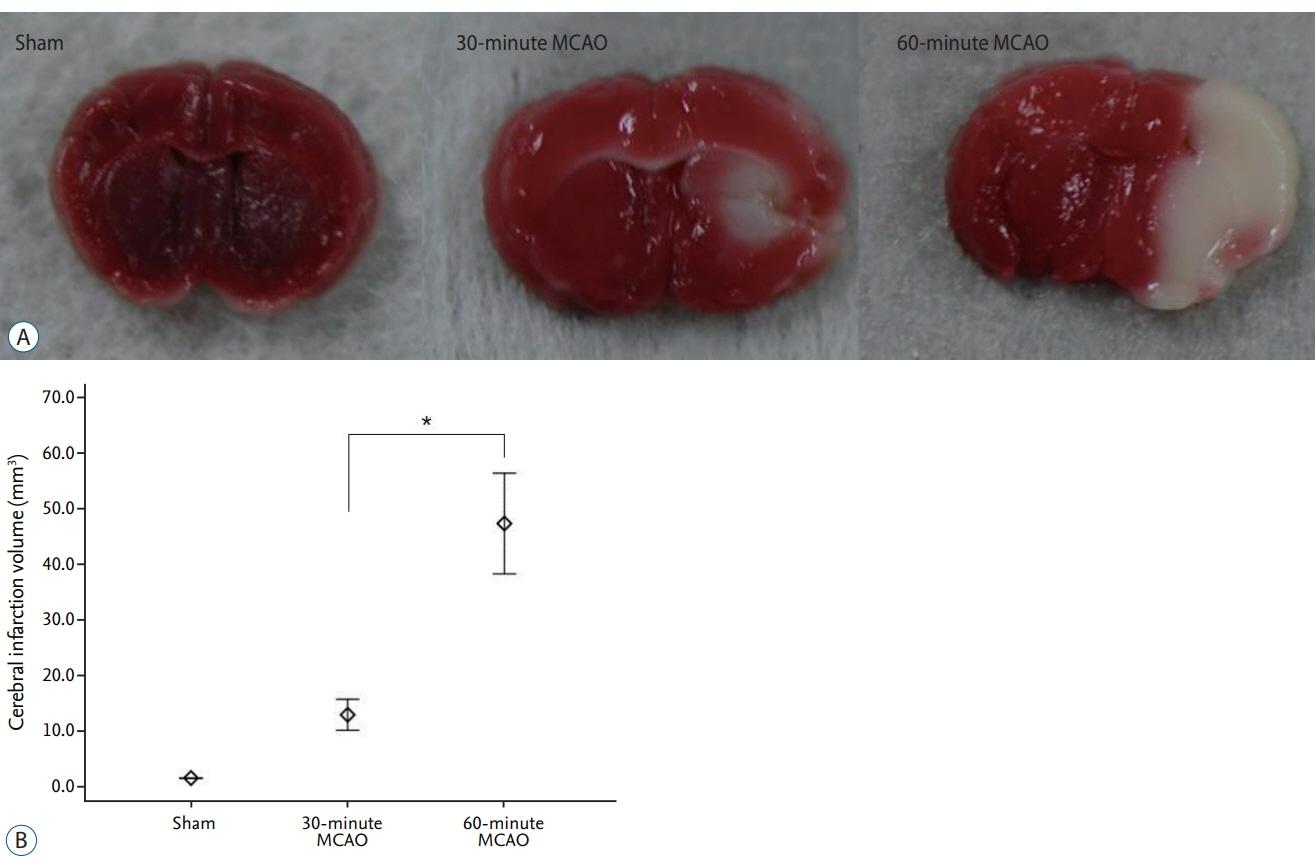
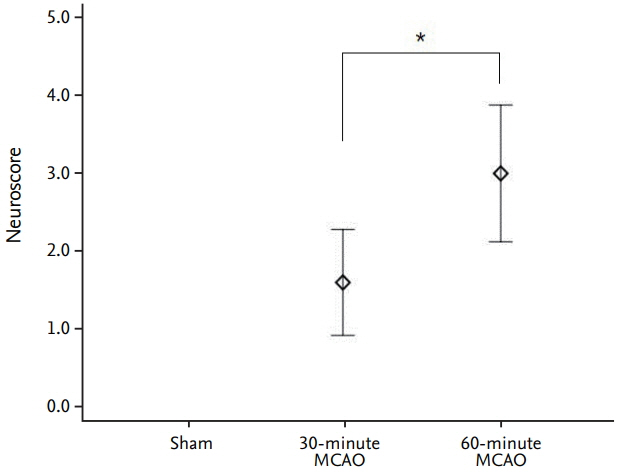
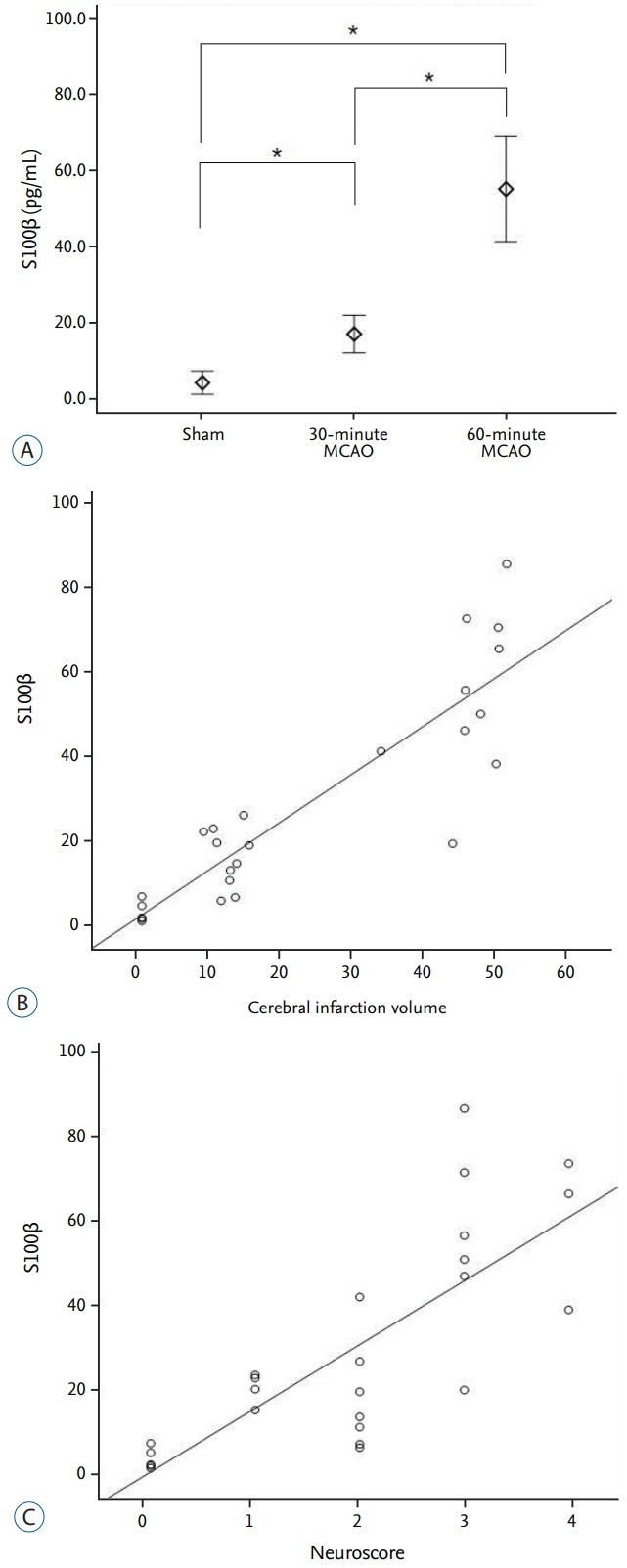
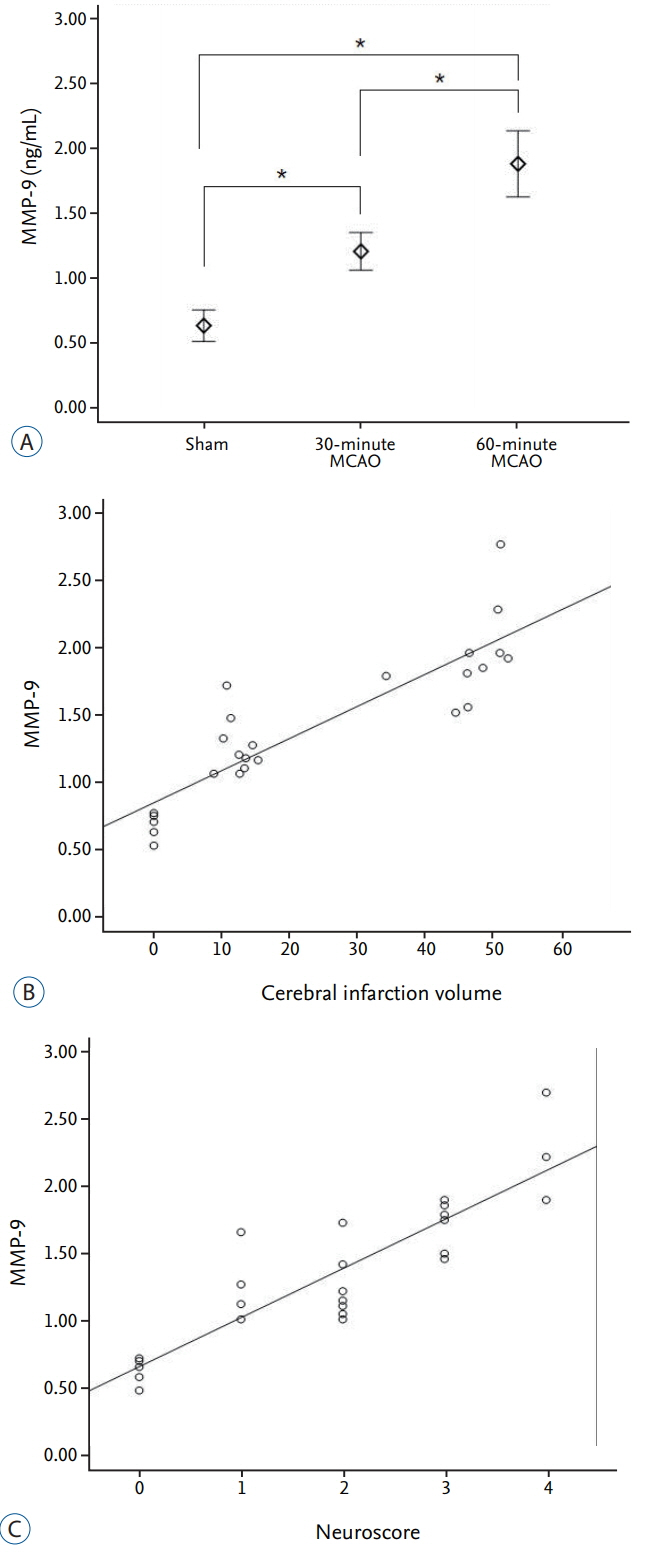
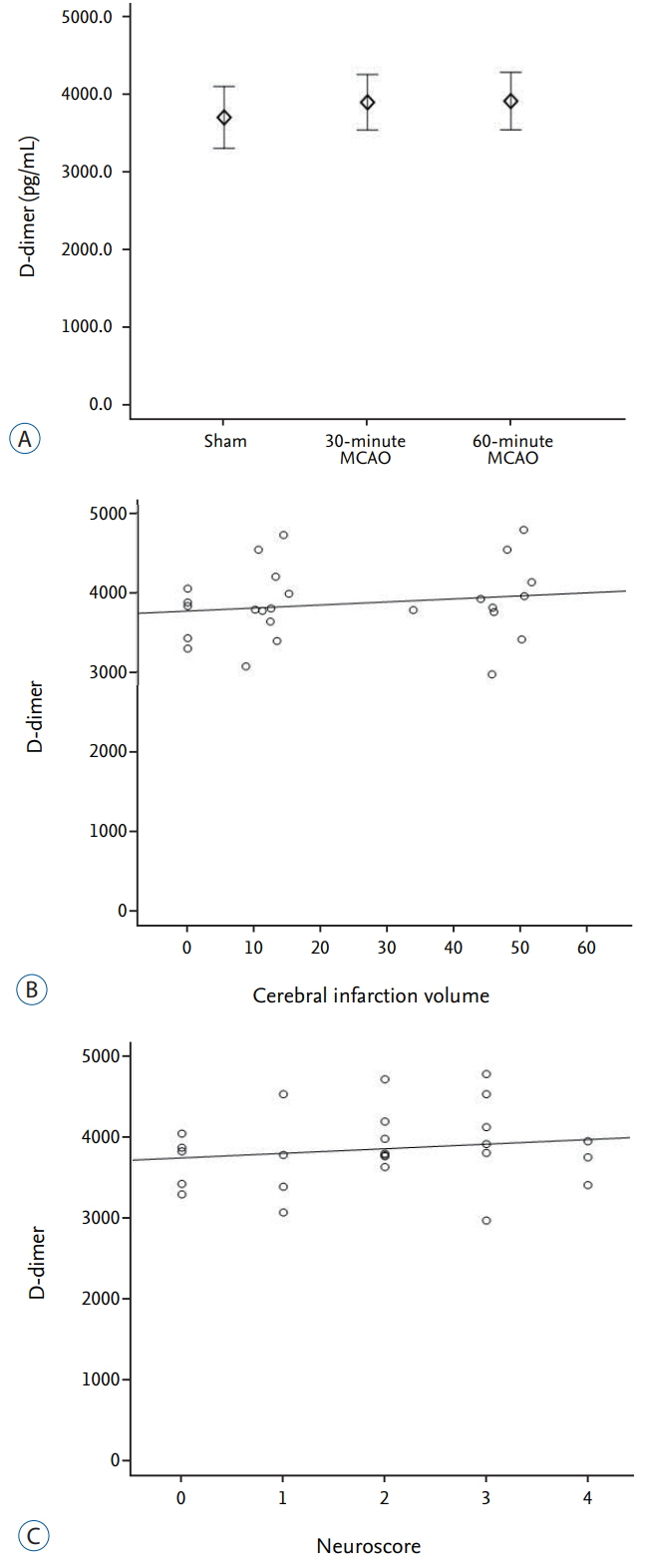
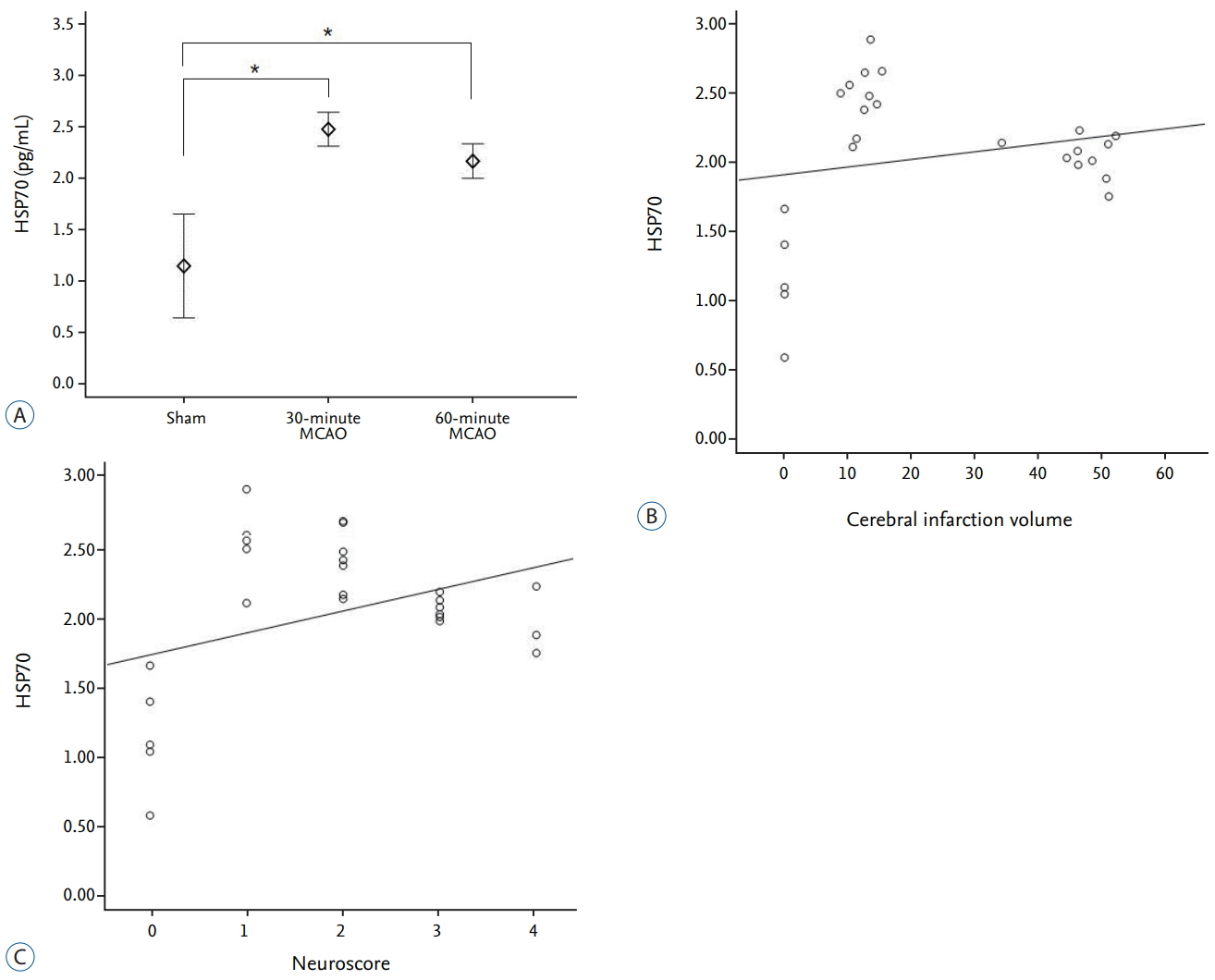
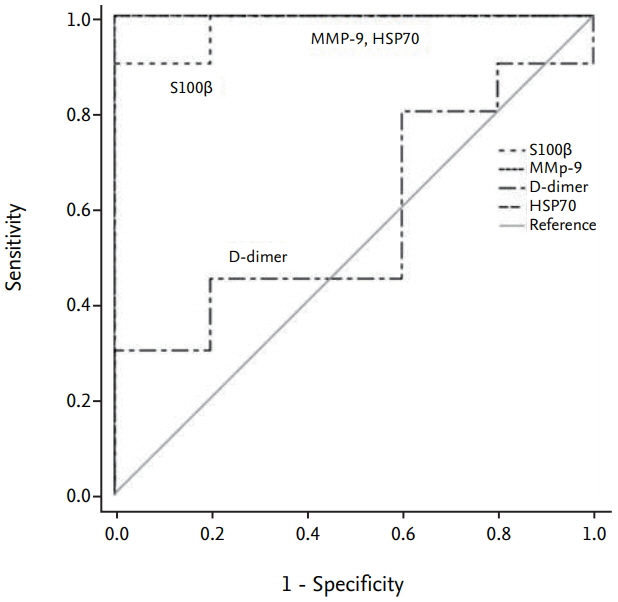
The average infarction volume was 12.32±2.31 mm³ and 46.9±7.43 mm³ in the 30- and 60-minute groups, respectively. The mean neurological score in the two ischemic groups was 1.6±0.55 and 3.2±0.70, respectively. S100β, MMP-9, and HSP70 expressions significantly increased after 4 hours of ischemia (p=0.001). Furthermore, S100β and MMP-9 expressions correlated with infarction volumes (p < 0.001) and neurological deficits (p < 0.001). There was no significant difference in D-dimer expression between groups (p=0.843). The area under the receiver operating characteristic curve (AUC) showed high sensitivity and specificity for MMP-9, HSP70 (AUC=1), and S100β (AUC=0.98).
CONCLUSION
S100β, MMP-9, and HSP70 can complement current diagnostic tools to assess cerebral infarction, suggesting their use as potential biomarkers for acute cerebral infarction.
MeSH Terms
-
Animals
Biomarkers*
Brain Ischemia
Cerebral Infarction*
Complement System Proteins
Diagnosis
Enzyme-Linked Immunosorbent Assay
Heat-Shock Proteins*
Hot Temperature*
HSP70 Heat-Shock Proteins*
Humans
Infarction
Ischemia
Matrix Metalloproteinase 9*
Mice*
Plasma
Prognosis
ROC Curve
Sensitivity and Specificity
Serologic Tests
Stroke
Biomarkers
Complement System Proteins
HSP70 Heat-Shock Proteins
Heat-Shock Proteins
Matrix Metalloproteinase 9
Figure
Reference
-
References
1. Ageno W, Finazzi S, Steidl L, Biotti MG, Mera V, Melzi D'Eril G, et al. Plasma measurement of D-dimer levels for the early diagnosis of ischemic stroke subtypes. Arch Intern Med. 162:2589–2593. 2002.
Article2. Arenillas JF, Alvarez-Sabin J, Molina CA, Chacón P, Fernández-Cadenas I, Ribó M, et al. Progression of symptomatic intracranial large artery atherosclerosis is associated with a proinflammatory state and impaired fibrinolysis. Stroke. 39:1456–1463. 2008.
Article3. Bader MK, Palmer S. What's the "hyper" in hyperacute stroke? Strategies to improve outcomes in ischemic stroke patients presenting within 6 hours. AACN Adv Crit Care. 17:194–214. 2006.4. Castellanos M, Leira R, Serena J, Blanco M, Pedraza S, Castillo J, et al. Plasma cellular-fibronectin concentration predicts hemorrhagic transformation after thrombolytic therapy in acute ischemic stroke. Stroke. 35:1671–1676. 2004.
Article5. Castillo J, Rodriguez I. Biochemical changes and inflammatory response as markers for brain ischaemia: molecular markers of diagnostic utility and prognosis in human clinical practice. Cerebrovasc Dis 17 Suppl. 1:7–18. 2004.
Article6. Foerch C, Otto B, Singer OC, Neumann-Haefelin T, Yan B, Berkefeld J, et al. Serum S100B predicts a malignant course of infarction in patients with acute middle cerebral artery occlusion. Stroke. 35:2160–2164. 2004.
Article7. Foerch C, Singer OC, Neumann-Haefelin T, du Mesnil de Rochemont R, Steinmetz H, Sitzer M. Evaluation of serum S100B as a surrogate marker for long-term outcome and infarct volume in acute middle cerebral artery infarction. Arch Neurol. 62:1130–1134. 2005.
Article8. Fujii Y, Takeuchi S, Harada A, Abe H, Sasaki O, Tanaka R. Hemostatic activation in spontaneous intracerebral hemorrhage. Stroke. 32:883–890. 2001.
Article9. Geiger S, Holdenrieder S, Stieber P, Hamann GF, Bruening R, Ma J, et al. Nucleosomes as a new prognostic marker in early cerebral stroke. J Neurol. 254:617–623. 2007.
Article10. Hacke W, Kaste M, Bluhmki E, Brozman M, Dávalos A, Guidetti D, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 359:1317–1329. 2008.
Article11. Jiang SX, Lertvorachon J, Hou ST, Konishi Y, Webster J, Mealing G, et al. Chlortetracycline and demeclocycline inhibit calpains and protect mouse neurons against glutamate toxicity and cerebral ischemia. J Biol Chem. 280:33811–33818. 2005.
Article12. Jin X, Xiao C, Tanguav RM, Yang L, Wang F, Chen M, et al. Correlation of lymphocyte heat shock protein 70 levels with neurologic deficits in elderly patients with cerebral infarction. Am J Med. 117:406–411. 2004.
Article13. Joly AL, Wettstein G, Mignot G, Ghiringhelli F, Garrido C. Dual role of heat shock proteins as regulators of apoptosis and innate immunity. J Innate Immun. 2:238–247. 2010.
Article14. Kleindienst A, Ross Bullock M. A critical analysis of the role of the neurotrophic protein S100B in acute brain injury. J Neurotrauma. 23:1185–1200. 2006.
Article15. Laskowitz DT, Kasner SE, Saver J, Remmel KS, Jauch EC, BRAIN Study Group. Clinical usefulness of a biomarker-based diagnostic test for acute stroke: the Biomarker Rapid Assessment in Ischemic Injury (BRAIN) study. Stroke. 40:77–85. 2009.
Article16. Lynch JR, Blessing R, White WD, Grocott HP, Newman MF, Laskowitz DT. Novel diagnostic test for acute stroke. Stroke. 35:57–63. 2004.
Article17. Maas MB, Furie KL. Molecular biomarkers in stroke diagnosis and prognosis. Biomark Med. 3:363–383. 2009.
Article18. Rajdev S, Hara K, Kokubo Y, Mestril R, Dillmann W, Weinstein PR, et al. Mice overexpressing rat heat shock protein 70 are protected against cerebral infarction. Ann Neurol. 47:782–791. 2000.
Article19. Reynolds MA, Kirchick HJ, Dahlen JR, Anderberg JM, McPherson PH, Nakamura KK, et al. Early biomarkers of stroke. Clin Chem. 49:1733–1739. 2003.
Article20. Rosell A, Ortega-Aznar A, Alvarez-Sabín J, Fernández-Cadenas I, Ribó M, Molina CA, et al. Increased brain expression of matrix metalloproteinase-9 after ischemic and hemorrhagic human stroke. Stroke. 37:1399–1406. 2006.
Article21. Schmidt-Kastner R, Zhang B, Belayev L, Khoutorova L, Amin R, Busto R, et al. DNA microarray analysis of cortical gene expression during early recirculation after focal brain ischemia in rat. Brain Res Mol Brain Res. 108:81–93. 2002.
Article22. Skoloudik D, Bar M, Sanák D, Bardon P, Roubec M, Langová K, et al. D-dimers increase in acute ischemic stroke patients with the large artery occlusion, but do not depend on the time of artery recanalization. J Thromb Thrombolysis. 29:477–482. 2010.
Article23. Snider BJ, Du C, Wei L, Choi DW. Cycloheximide reduces infarct volume when administered up to 6 h after mild focal ischemia in rats. Brain Res. 917:147–157. 2001.
Article24. Swanson RA, Morton MT, Tsao-Wu G, Savalos RA, Davidson C, Sharp FR. A semiautomated method for measuring brain infarct volume. J Cereb Blood Flow Metab. 10:290–293. 1990.
Article25. Whiteley W, Chong WL, Sengupta A, Sandercock P. Blood markers for the prognosis of ischemic stroke: a systematic review. Stroke. 40:e380–e389. 2009.26. Whiteley W, Wardlaw J, Dennis M, Lowe G, Rumley A, Sattar N, et al. Blood biomarkers for the diagnosis of acute cerebrovascular diseases: a prospective cohort study. Cerebrovasc Dis. 32:141–147. 2011.
Article27. Wunderlich MT, Wallesch CW, Goertler M. Release of neurobiochemical markers of brain damage is related to the neurovascular status on admission and the site of arterial occlusion in acute ischemic stroke. J Neurol Sci. 227:49–53. 2004.
Article28. Yenari MA, Giffard RG, Sapolsky RM, Steinberg GK. The neuroprotective potential of heat shock protein 70 (HSP70). Mol Med Today. 5:525–531. 1999.
Article29. Zhan X, Ander BP, Liao IH, Hansen JE, Kim C, Clements D, et al. Recombinant Fv-Hsp70 protein mediates neuroprotection after focal cerebral ischemia in rats. Stroke. 41:538–543. 2010.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Semi-Quantitative Analyses of Hippocampal Heat Shock Protein-70 Expression Based on the Duration of Ischemia and the Volume of Cerebral Infarction in Mice
- The Significance of Induction of Heat Shock Protein-70 and Glial Fibrillary Acidic Protein Messenger RNA by Delayed Postischemic Hyperthermia Following Transient Focal Ischemia
- Ischemic postconditioning may not influence early brain injury induced by focal cerebral ischemia/reperfusion in rats
- Expression of Heat Shock Protein 70 m-RNA in Rat Bladder Overdistended by Diuresis
- The Temporal Changes of Matrix Metalloproteinases in Experimental Middle Cerebral Artery Occlusion and Reperfusion