Nat Prod Sci.
2018 Mar;24(1):59-65. 10.20307/nps.2018.24.1.59.
Lignans with NADPH Oxidase 2 (NOX2)-inhibitory Activity from the Fruits of Schisandra chinensis
- Affiliations
-
- 1College of Pharmacy, Dongguk University, Goyang, Gyeonggi-do 10326, Republic of Korea. f2744@dongguk.edu, mlee@dongguk.edu
- KMID: 2409611
- DOI: http://doi.org/10.20307/nps.2018.24.1.59
Abstract
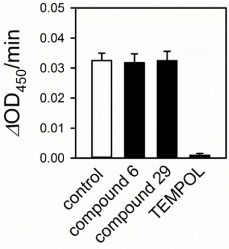
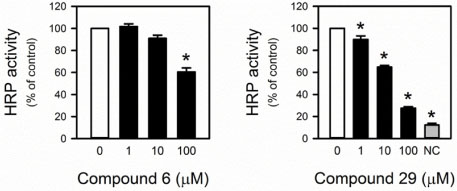
- An isoform of NADPH oxidase (NOX), NOX2 is a superoxide-generating enzyme involved in diverse pathophysiological events. Although its potential as a therapeutic target has been validated, there is no clinically available inhibitor. Herein, NOX2-inhibitory activity was screened with the constituents isolated from Schisandra chinensis, which has been reported to have antioxidant and reactive oxygen species (ROS)-scavenging effects. Among the partitions prepared from crude methanolic extract, a chloroform-soluble partition showed the highest NOX2-inhibitory activity in PLB-985 cell-based NOX2 assay. A total of twenty nine compounds (1 - 29) were identified from the chloroform fraction, including two first isolated compounds; dimethyl-malate (25) and 2-(2-hydroxyacetyl) furan (27) from this plants. Of these constituents, two compounds (gomisin T, and pregomisin) exhibited an NOX2-inhibitory effect with the ICâ‚…â‚€ of 9.4 ± 3.6, and 62.9 ± 11.3 µM, respectively. They are confirmed not to be nonspecific superoxide scavengers in a counter assay using a xanthine-xanthine oxidase system. These findings suggest the potential application of gomisin T (6) and other constituents of S. chinensis to inhibit NOX2.
MeSH Terms
Figure
Reference
-
1. Lee MY, Griendling KK. Antioxid Redox Signal. 2008; 10:1045–1059.2. Jiang F, Zhang Y, Dusting GJ. Pharmacol Rev. 2011; 63:218–242.3. Lassegue B, San Martin A, Griendling KK. Circ Res. 2012; 110:1364–1390.4. Altenhofer S, Radermacher KA, Kleikers PW, Wingler K, Schmidt HH. Antioxid Redox Signal. 2015; 23:406–427.5. Drummond GR, Selemidis S, Griendling KK, Sobey CG. Nat Rev Drug Discov. 2011; 10:453–471.6. Panossian A, Wikman G. J Ethnopharmacol. 2008; 118:183–212.7. Thandavarayan RA, Giridharan VV, Arumugam S, Suzuki K, Ko KM, Krishnamurthy P, Watanabe K, Konishi T. PLoS One. 2015; 10:e0119214.8. Wang X, Hu D, Zhang L, Lian G, Zhao S, Wang C, Yin J, Wu C, Yang J. Food Chem Toxicol. 2014; 63:119–127.9. Tucker KA, Lilly MB, Heck L Jr, Rado TA. Blood. 1987; 70:372–378.10. Debeurme F, Picciocchi A, Dagher MC, Grunwald D, Beaumel S, Fieschi F, Stasia MJ. J Biol Chem. 2010; 285:33197–33208.11. Liu H, Lai H, Jia X, Liu J, Zhang Z, Qi Y, Zhang J, Song J, Wu C, Zhang B, Xiao P. Phytomedicine. 2013; 20:1135–1143.12. Ikeya Y, Taguchi H, Yosioka I, Iitaka Y, Kobayashi H. Chem Pharm Bull. 1979; 27:1395–1401.13. Ikeya Y, Taguchi H, Yosioka I, Kobayashi H. Chem Pharm Bull. 1978; 26:3257–3260.14. Ma WH, Lu Y, Huang H, Zhou P, Chen DF. Bioorg Med Chem Lett. 2009; 19:4958–4962.15. Ikeya Y, Miki E, Okada M, Mitsuhashi H, Chai JG. Chem Pharm Bull. 1990; 38:1408–1411.16. Kanatani H. Teraba.
Article17. Wan CK, Zhu GY, Shen XL, Chattopadhyay A, Dey S, Fong WF. Biochem Pharmacol. 2006; 72:824–837.18. Cao YF, Zhang YY, Li J, Ge GB, Hu D, Liu HX, Huang T, Wang YC, Fang ZZ, Sun DX, Huo H, Yin J, Yang L. Xenobiotica. 2010; 40:38–47.19. Ikeya Y, Taguchi H, Yosioka I. Chem Pharm Bull. 1978; 26:328–331.20. Xue Y, Li X, Du X, Li X, Wang W, Yang J, Chen J, Pu J, Sun H. Phytochemistry. 2015; 116:253–261.21. Sun H, Wu F, Zhang A, Wei W, Han Y, Wang X. Biomed Chromatogr. 2013; 27:1511–1519.22. Park C, Choi YW, Hyun SK, Kwon HJ, Hwang HJ, Kim GY, Choi BT, Kim BW, Choi IW, Moon SK, Kim WJ, Choi YH. Int J Mol Med. 2009; 24:495–502.23. Seo SM, Lee HJ, Park YK, Lee MK, Park JI, Paik KH. Nat Prod Sci. 2004; 10:104–108.24. Ikeya Y, Taguchi H, Yosioka I. Chem Pharm Bull. 1982; 30:132–139.25. Chen M, Xu X, Xu B, Yang P, Liao Z, Morris-Natschke SL, Lee KH, Chen D. Molecule. 2013; 18:2297–2306.26. Ikeya Y, Taguchi H, Yosioka I. Chem Pharm Bull. 1980; 28:2422–2427.27. Lee IS, Jung KY, Oh SR, Park SH, Ahn KS, Lee HK. Biol Pharm Bull. 1999; 22:265–267.28. Denmark SE, Ahmad M. J Org Chem. 2007; 72:9630–9634.29. Utaka M, Nakatani M, Takeda A. J Org Chem. 1986; 51:1140–1141.30. Mascal M, Nikitin EB. Angew Chem Int Ed Engl. 2008; 47:7924–7926.31. Xu LJ, Huang F, Chen SB, Li LN, Chen SL, Xiao PG. J Integr Plant Biol. 2006; 48:1493–1497.32. Lee IS, Jung KY, Oh SR, Kim DS, Kim JH, Lee JJ, Lee HK, Lee SH, Kim EH, Cheong C. Arch Pharm Res. 1997; 20:633–636.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Schisandrol A and gomisin N from Schisandra chinensis extract improve hypogonadism via anti-oxidative stress in TM3 Leydig cells
- Enhancement of Allergen-induced Airway Inflammation by NOX2 Deficiency
- Roles of Reactive Oxygen Species on Insulin Resistance in Adipose Tissue
- p47phox, the phagocyte NADPH oxidase/NOX2 organizer: structure, phosphorylation and implication in diseases
- NO Production Inhibition of Lignans from Vietnamese Schisandra sphenanthera Rehd. et Wils. Fruits