Nutr Res Pract.
2023 Feb;17(1):1-12. 10.4162/nrp.2023.17.1.1.
Schisandrol A and gomisin N from Schisandra chinensis extract improve hypogonadism via anti-oxidative stress in TM3 Leydig cells
- Affiliations
-
- 1College of Pharmacy, Kyungsung University, Busan 48434, Republic of Korea
- 2College of Engineering, North Carolina A&T State University, Greensboro, NC 27411, USA
- KMID: 2539450
- DOI: http://doi.org/10.4162/nrp.2023.17.1.1
Abstract
- BACKGROUND/OBJECTIVES
Male hypogonadism is a condition where the body does not produce enough testosterone and significantly impacts health. Age, obesity, genetics, and oxidative stress are some physiological factors that may contribute to testosterone deficiency. Previous studies have shown many pharmacological benefits of Schisandra chinensis (S. chinensis) Baillon as an anti-inflammatory and antioxidant. However, the molecular mechanism of attenuating hypogonadism is yet to be well established. This research was undertaken to study the effects of S. chinensis extract (SCE) on testosterone deficiency.
MATERIALS/METHODS
S. chinensis fruit was pulverized and extracted using 60% aqueous ethanol. HPLC analysis was performed to analyze and quantify the lignans of the SCE.
RESULTS
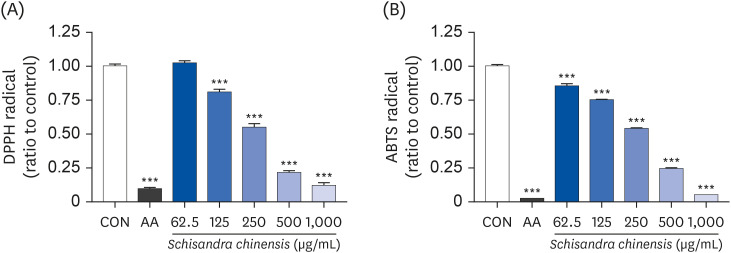
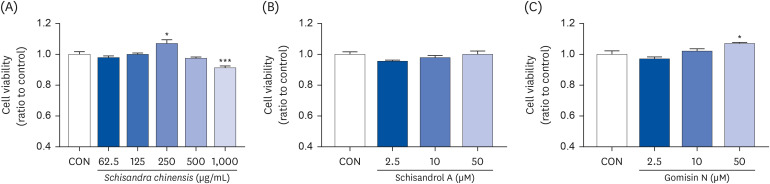
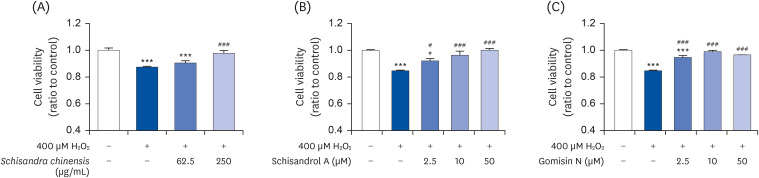
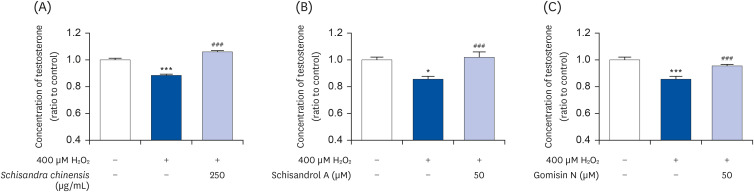
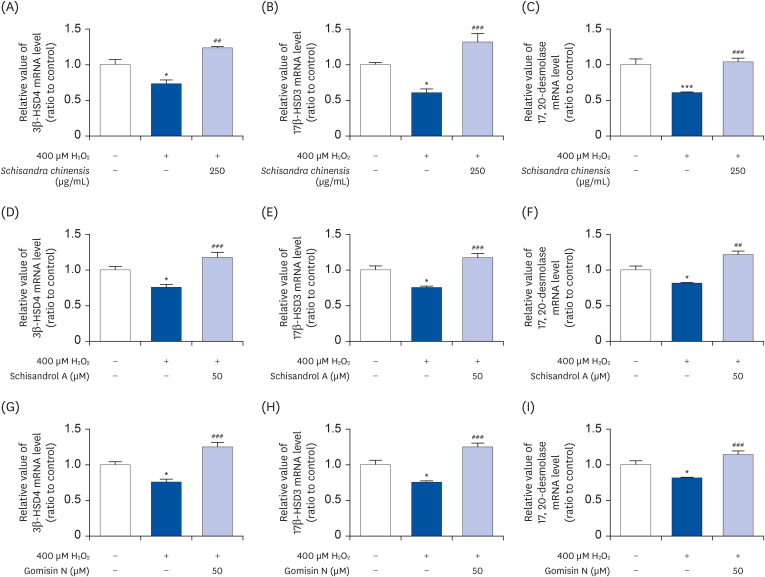
The 2,2-diphenyl-2-picrylhydrazyl and 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) scavenging assays confirmed that the SCE and its major lignans (schisandrol A and gomisin N) inhibit oxidative stress. Effects of SCE analysis on the testosterone level under oxidative stress conditions revealed that both schisandrol A and gomisin N were able to recover the lowered testosterone levels. Through mRNA expression of TM3 Leydig cell, we observed that the SCE lignans were able to induce the enzymes involved in testosterone biosynthesis-related genes such as 3β-HSD4 (P < 0.01 for SCE, and P < 0.001 for schisandrol A and gomisin N), 17β-HSD3 (P < 0.001 for SCE, schisandrol A and gomisin N), and 17, 20-desmolase (P < 0.01 for schisandrol A, and P < 0.001 for SCE and gomisin N).
CONCLUSIONS
These results support that SCE and its active components could be potential therapeutic agents for regulating and increasing testosterone production.
Figure
Reference
-
1. Diver MJ. Clinical Scince Reviews Committee of the Association for Clinical Biochemistry. Analytical and physiological factors affecting the interpretation of serum testosterone concentration in men. Ann Clin Biochem. 2006; 43:3–12. PMID: 16390603.
Article2. Hisasue S. Contemporary perspective and management of testosterone deficiency: Modifiable factors and variable management. Int J Urol. 2015; 22:1084–1095. PMID: 26303309.
Article3. Finkelstein JS, Yu EW, Burnett-Bowie SA. Gonadal steroids and body composition, strength, and sexual function in men. N Engl J Med. 2013; 369:2457.
Article4. Sattler FR, Castaneda-Sceppa C, Binder EF, Schroeder ET, Wang Y, Bhasin S, Kawakubo M, Stewart Y, Yarasheski KE, Ulloor J, et al. Testosterone and growth hormone improve body composition and muscle performance in older men. J Clin Endocrinol Metab. 2009; 94:1991–2001. PMID: 19293261.
Article5. Storer TW, Basaria S, Traustadottir T, Harman SM, Pencina K, Li Z, Travison TG, Miciek R, Tsitouras P, Hally K, et al. Effects of testosterone supplementation for 3 years on muscle performance and physical function in older men. J Clin Endocrinol Metab. 2017; 102:583–593. PMID: 27754805.6. Kumar P, Kumar N, Thakur DS, Patidar A. Male hypogonadism: symptoms and treatment. J Adv Pharm Technol Res. 2010; 1:297–301. PMID: 22247861.7. Leisegang K, Roychoudhury S, Slama P, Finelli R. The mechanisms and management of age-related oxidative stress in male hypogonadism associated with non-communicable chronic disease. Antioxidants. 2021; 10:1834. PMID: 34829704.
Article8. Chunchai T, Apaijai N, Keawtep P, Mantor D, Arinno A, Pratchayasakul W, Chattipakorn N, Chattipakorn SC. Testosterone deprivation intensifies cognitive decline in obese male rats via glial hyperactivity, increased oxidative stress, and apoptosis in both hippocampus and cortex. Acta Physiol (Oxf). 2019; 226:e13229. PMID: 30506942.9. Zhao X, Xu W, Wu J, Zhang D, Abou-Shakra A, Di L, Wang Z, Wang L, Yang F, Qiao Z. Nicotine induced autophagy of Leydig cells rather than apoptosis is the major reason of the decrease of serum testosterone. Int J Biochem Cell Biol. 2018; 100:30–41. PMID: 29753783.
Article10. Cho YH, Lee SY, Lee CH, Park JH, So YS. Effect of Schisandra chinensis Baillon extracts and regular low-intensity exercise on muscle strength and mass in older adults: a randomized, double-blind, placebo-controlled trial. Am J Clin Nutr. 2021; 113:1440–1446. PMID: 33710261.
Article11. Shen B. A new golden age of natural products drug discovery. Cell. 2015; 163:1297–1300. PMID: 26638061.12. Nowak A, Zakłos-Szyda M, Błasiak J, Nowak A, Zhang Z, Zhang B. Potential of Schisandra chinensis (Turcz.) Baill. in human health and nutrition: a review of current knowledge and therapeutic perspectives. Nutrients. 2019; 11:333. PMID: 30720717.
Article13. Szopa A, Ekiert R, Ekiert H. Current knowledge of Schisandra chinensis (Turcz.) Baill. (Chinese magnolia vine) as a medicinal plant species: a review on the bioactive components, pharmacological properties, analytical and biotechnological studies. Phytochem Rev. 2017; 16:195–218. PMID: 28424569.
Article14. Huang T, Shen P, Shen Y. Preparative separation and purification of deoxyschisandrin and gamma-schisandrin from Schisandra chinensis (Turcz.) Baill by high-speed counter-current chromatography. J Chromatogr A. 2005; 1066:239–242. PMID: 15794577.15. Huang H, Shen Z, Geng Q, Wu Z, Shi P, Miao X. Protective effect of Schisandra chinensis bee pollen extract on liver and kidney injury induced by cisplatin in rats. Biomed Pharmacother. 2017; 95:1765–1776. PMID: 28962082.
Article16. Kopustinskiene DM, Bernatoniene J. Antioxidant effects of Schisandra chinensis fruits and their active constituents. Antioxidants. 2021; 10:10.17. Choi YW, Takamatsu S, Khan SI, Srinivas PV, Ferreira D, Zhao J, Khan IA. Schisandrene, a dibenzocyclooctadiene lignan from Schisandra chinensis: structure-antioxidant activity relationships of dibenzocyclooctadiene lignans. J Nat Prod. 2006; 69:356–359. PMID: 16562834.
Article18. Panossian A, Wikman G. Pharmacology of Schisandra chinensis Bail.: an overview of Russian research and uses in medicine. J Ethnopharmacol. 2008; 118:183–212. PMID: 18515024.
Article19. Zhao J, Sun T, Wu JJ, Cao YF, Fang ZZ, Sun HZ, Zhu ZT, Yang K, Liu YZ, Gonzalez FJ, et al. Inhibition of human CYP3A4 and CYP3A5 enzymes by gomisin C and gomisin G, two lignan analogs derived from Schisandra chinensis . Fitoterapia. 2017; 119:26–31. PMID: 28344076.
Article20. Wan CK, Tse AK, Yu ZL, Zhu GY, Wang H, Fong DW. Inhibition of cytochrome P450 3A4 activity by schisandrol A and gomisin A isolated from Fructus Schisandrae chinensis. Phytomedicine. 2010; 17:702–705. PMID: 20089387.
Article21. Ward JF, Evans JW, Limoli CL, Calabro-Jones PM. Radiation and hydrogen peroxide induced free radical damage to DNA. Br J Cancer Suppl. 1987; 8:105–112. PMID: 2820457.22. Habu JB, Ibeh BO. In vitro antioxidant capacity and free radical scavenging evaluation of active metabolite constituents of Newbouldia laevis ethanolic leaf extract. Biol Res. 2015; 48:16. PMID: 25849161.23. Aktumsek A, Zengin G, Guler GO, Cakmak YS, Duran A. Antioxidant potentials and anticholinesterase activities of methanolic and aqueous extracts of three endemic Centaurea L. species. Food Chem Toxicol. 2013; 55:290–296. PMID: 23357566.
Article24. Dandona P, Rosenberg MT. A practical guide to male hypogonadism in the primary care setting. Int J Clin Pract. 2010; 64:682–696. PMID: 20518947.
Article25. Han DH, Lee JH, Kim H, Ko MK, Chae MR, Kim HK, So I, Jeon JH, Park JK, Lee SW. Effects of Schisandra chinensis extract on the contractility of corpus cavernosal smooth muscle (CSM) and Ca2+ homeostasis in CSM cells. BJU Int. 2012; 109:1404–1413. PMID: 21951618.
Article26. Martin LJ, Touaibia M. Improvement of testicular steroidogenesis using flavonoids and isoflavonoids for prevention of late-onset male hypogonadism. Antioxidants. 2020; 9:237. PMID: 32183155.
Article27. Smith SJ, Lopresti AL, Teo SY, Fairchild TJ. Examining the effects of herbs on testosterone concentrations in men: a systematic review. Adv Nutr. 2021; 12:744–765. PMID: 33150931.
Article28. Hong SH, Li M, Jeung EB, Lee GS, Hong EJ, Choi YW, An BS. Therapeutic effects of Schisandra chinensis on the hyperprolactinemia in rat. Int J Oncol. 2017; 50:1448–1454. PMID: 28259983.
Article29. Zirkin BR, Papadopoulos V. Leydig cells: formation, function, and regulation. Biol Reprod. 2018; 99:101–111. PMID: 29566165.
Article30. Wu FC, Tajar A, Pye SR, Silman AJ, Finn JD, O’Neill TW, Bartfai G, Casanueva F, Forti G, Giwercman A, et al. Hypothalamic-pituitary-testicular axis disruptions in older men are differentially linked to age and modifiable risk factors: the European Male Aging Study. J Clin Endocrinol Metab. 2008; 93:2737–2745. PMID: 18270261.
Article31. Hu W, Shi L, Li MY, Zhou PH, Qiu B, Yin K, Zhang HH, Gao Y, Kang R, Qin SL, et al. Adrenomedullin protects Leydig cells against lipopolysaccharide-induced oxidative stress and inflammatory reaction via MAPK/NF-κB signalling pathways. Sci Rep. 2017; 7:16479. PMID: 29184072.
Article32. Karna KK, Choi BR, You JH, Shin YS, Soni KK, Cui WS, Lee SW, Kim CY, Kim HK, Park JK. Cross-talk between ER stress and mitochondrial pathway mediated adriamycin-induced testicular toxicity and DA-9401 modulate adriamycin-induced apoptosis in Sprague-Dawley rats. Cancer Cell Int. 2019; 19:85. PMID: 30992692.
Article33. Flaherty RL, Owen M, Fagan-Murphy A, Intabli H, Healy D, Patel A, Allen MC, Patel BA, Flint MS. Glucocorticoids induce production of reactive oxygen species/reactive nitrogen species and DNA damage through an iNOS mediated pathway in breast cancer. Breast Cancer Res. 2017; 19:35. PMID: 28340615.
Article34. Bakunina N, Pariante CM, Zunszain PA. Immune mechanisms linked to depression via oxidative stress and neuroprogression. Immunology. 2015; 144:365–373. PMID: 25580634.35. Pérez-Jiménez J, Díaz-Rubio ME, Saura-Calixto F. Contribution of macromolecular antioxidants to dietary antioxidant capacity: a study in the Spanish Mediterranean diet. Plant Foods Hum Nutr. 2015; 70:365–370. PMID: 26482738.
Article36. He JL, Zhou ZW, Yin JJ, He CQ, Zhou SF, Yu Y. Schisandra chinensis regulates drug metabolizing enzymes and drug transporters via activation of Nrf2-mediated signaling pathway. Drug Des Devel Ther. 2014; 9:127–146.37. Karna KK, Choi BR, Kim MJ, Kim HK, Park JK. The effect of Schisandra chinensis Baillon on cross-talk between oxidative stress, endoplasmic reticulum stress, and mitochondrial signaling pathway in testes of varicocele-induced SD rat. Int J Mol Sci. 2019; 20:5785. PMID: 31744253.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Lignans with NADPH Oxidase 2 (NOX2)-inhibitory Activity from the Fruits of Schisandra chinensis
- NO Production Inhibition of Lignans from Vietnamese Schisandra sphenanthera Rehd. et Wils. Fruits
- Improvement of andropause symptoms by dandelion and rooibos extract complex CRS-10 in aging male
- Suppressive effects of Schizandra chinensis Baillon water extract on allergy-related cytokine generation and degranulation in IgE-antigen complex-stimulated RBL-2H3 cells
- Fancd2os Reduces Testosterone Production by Inhibiting Steroidogenic Enzymes and Promoting Cellular Apoptosis in Murine Testicular Leydig Cells