Nutr Res Pract.
2012 Dec;6(6):505-512.
Improvement of andropause symptoms by dandelion and rooibos extract complex CRS-10 in aging male
- Affiliations
-
- 1Department of Anatomy and Cell Biology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea.
- 2BioGrand Biomedical Research Center, BioGrand Inc., Gyeonggi 427-800, Korea.
- 3Korea Food Research Institute, Gyeonggi 463-746, Korea.
- 4Department of Microbiology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea.
- 5Department of Physiology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea.
- 6Department of Food Science and Nutrition, College of Natural Science, Dankook University, 152 Jukjeon-ro, Suji-gu, Yongin, Gyeonggi 448-701, Korea. yjeong@dankook.ac.kr
- 7Korea Health Supplements Institute, Gyeonggi 463-400, Korea.
- 8Department of Urology, College of Medicine, Chung-Ang University, 221 Heukseok-dong, Dongjak-gu, Seoul 156-756, Korea. yjeong@dankook.ac.kr
Abstract
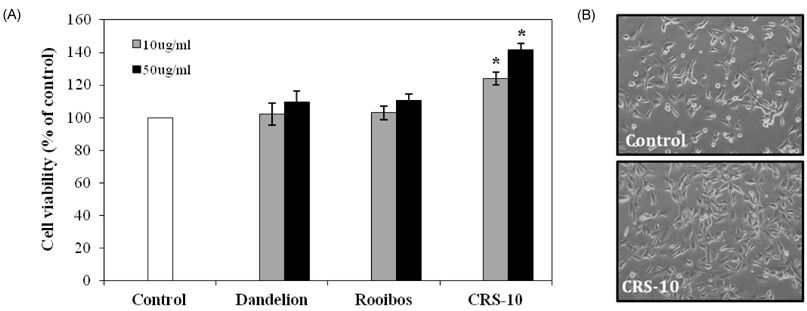
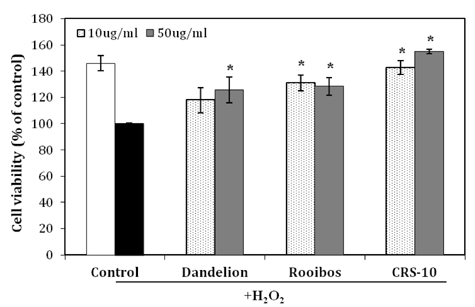
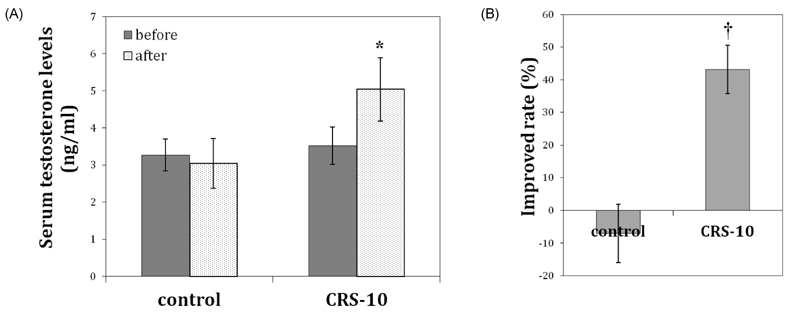
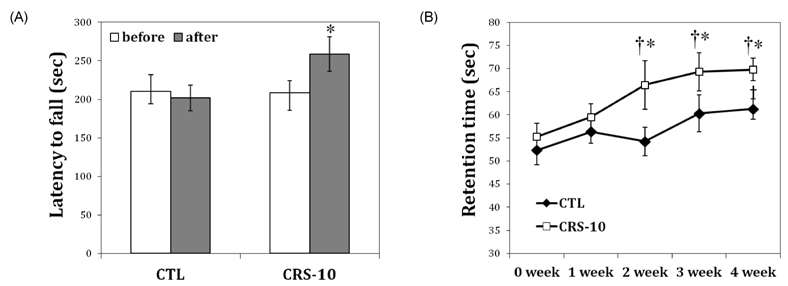
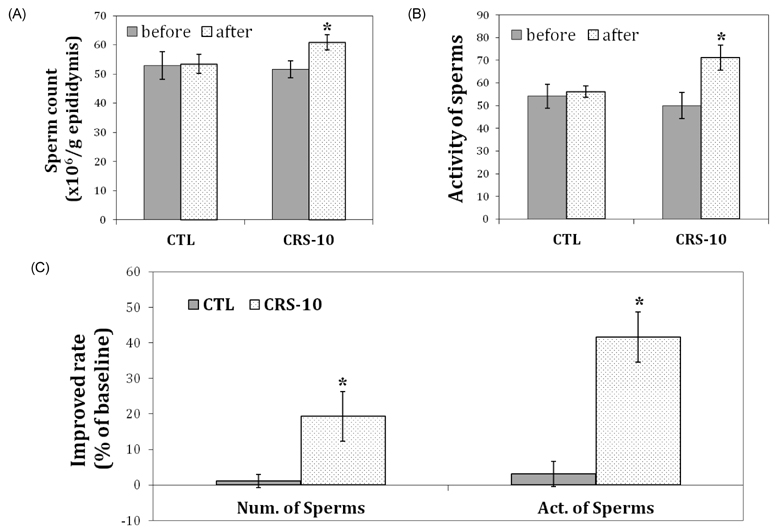
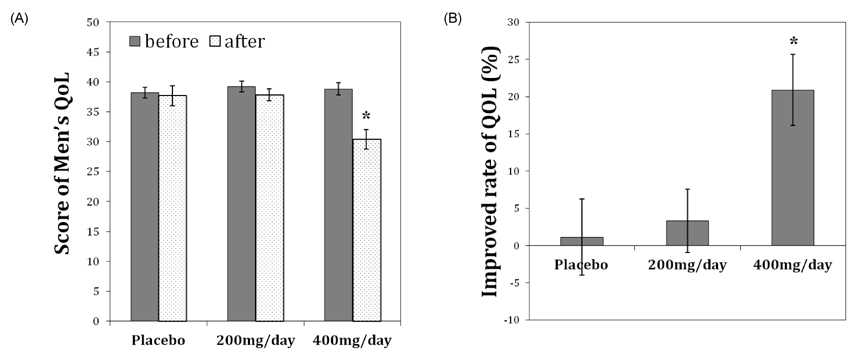
- Many aging male suffer various andropause symptoms including loss of physical and mental activities. This study evaluated the putative alleviative effects of CRS-10 dandelion and rooibos extract complex (CRS-10) on the symptoms of andropause. The survival rate of TM3 Leydig cells (TM3 cells) treated with CRS-10 was measured based on typical physiological stress. After daily intake of CRS-10 for 4 weeks, the level of testosterone, physical activity and both the number and activity of sperm in older rats (18 weeks) were measured. Furthermore, thirty males were surveyed with AMS (Aging Males' Symptoms) questionnaire after intake of 400 mg of CRS-10. Overall, CRS-10 protected TM3 cells from serum restriction and oxidative stress via activation of ERK and Akt pathways. The level of testosterone and activation of spermatogenesis in rats were significantly enhanced. In addition, physical locomotion was markedly improved. Daily intake of 400 mg of CRS-10 improved the quality of life among agingmale respondents, according to a clinical survey using the AMS. The results indicate the potential of CRS-10 as a safe and efficacious natural substance for reducing or alleviating andropause symptoms.
Keyword
MeSH Terms
Figure
Reference
-
1. Zhang Y, Ge R, Hardy MP. Androgen-forming stem Leydig cells: identification, function and therapeutic potential. Dis Markers. 2008. 24:277–286.
Article2. Chen L, Zhao Y, Zhang Y. Progress on the research of stem Leydig cell line in the testis of rodents. Wei Sheng Yan Jiu. 2012. 41:158–162.3. Wang C, Hikim AS, Ferrini M, Bonavera JJ, Vernet D, Leung A, Lue YH, Gonzalez-Cadavid NF, Swerdloff RS. Male reproductive ageing: using the brown Norway rat as a model for man. Novartis Found Symp. 2002. 242:82–95. discussion 95-7.
Article4. Luo L, Chen H, Zirkin BR. Are Leydig cell steroidogenic enzymes differentially regulated with aging? J Androl. 1996. 17:509–515.5. Wu CY, Yu TJ, Chen MJ. Age related testosterone level changes and male andropause syndrome. Chang Gung Med J. 2000. 23:348–353.6. Travison TG, Basaria S, Storer TW, Jette AM, Miciek R, Farwell WR, Choong K, Lakshman K, Mazer NA, Coviello AD, Knapp PE, Ulloor J, Zhang A, Brooks B, Nguyen AH, Eder R, LeBrasseur N, Elmi A, Appleman E, Hede-Brierley L, Bhasin G, Bhatia A, Lazzari A, Davis S, Ni P, Collins L, Bhasin S. Clinical meaningfulness of the changes in muscle performance and physical function associated with testosterone administration in older men with mobility limitation. J Gerontol A Biol Sci Med Sci. 2011. 66:1090–1099.
Article7. Sattler F, Bhasin S, He J, Chou CP, Castaneda-Sceppa C, Yarasheski K, Binder E, Schroeder ET, Kawakubo M, Zhang A, Roubenoff R, Azen S. Testosterone threshold levels and lean tissue mass targets needed to enhance skeletal muscle strength and function: the HORMA trial. J Gerontol A Biol Sci Med Sci. 2011. 66:122–129.
Article8. Chen RY, Ng KK. Self-referred older Asian males in a men's health clinic: the inter-relationships between androgens, metabolic parameters and quality of life measures. Aging Male. 2010. 13:233–241.
Article9. Ibebunjo C, Eash JK, Li C, Ma Q, Glass DJ. Voluntary running, skeletal muscle gene expression, and signaling inversely regulated by orchidectomy and testosterone replacement. Am J Physiol Endocrinol Metab. 2011. 300:E327–E340.
Article10. Nigro N, Christ-Crain M. Testosterone treatment in the aging male: myth or reality? Swiss Med Wkly. 2012. 142:w13539.
Article11. Barqawi A, Crawford ED. Testosterone replacement therapy and the risk of prostate cancer. Is there a link? Int J Impot Res. 2006. 18:323–328.
Article12. Tenover JL. Male hormone replacement therapy including "andropause". Endocrinol Metab Clin North Am. 1998. 27:969–987. x
Article13. Hu C, Kitts DD. Dandelion (Taraxacum officinale) flower extract suppresses both reactive oxygen species and nitric oxide and prevents lipid oxidation in vitro. Phytomedicine. 2005. 12:588–597.
Article14. He W, Han H, Wang W, Gao B. Anti-influenza virus effect of aqueous extracts from dandelion. Virol J. 2011. 8:538.
Article15. Huang Y, Wu T, Zeng L, Li S. Chinese medicinal herbs for sore throat. Cochrane Database Syst Rev. 2012. 3:CD004877.
Article16. Ovadje P, Chatterjee S, Griffin C, Tran C, Hamm C, Pandey S. Selective induction of apoptosis through activation of caspase-8 in human leukemia cells (Jurkat) by dandelion root extract. J Ethnopharmacol. 2011. 133:86–91.
Article17. Utter AC, Quindry JC, Emerenziani GP, Valiente JS. Effects of rooibos tea, bottled water, and a carbohydrate beverage on blood and urinary measures of hydration after acute dehydration. Res Sports Med. 2010. 18:85–96.
Article18. Darvesh AS, Carroll RT, Bishayee A, Geldenhuys WJ, Van der Schyf CJ. Oxidative stress and Alzheimer's disease: dietary polyphenols as potential therapeutic agents. Expert Rev Neurother. 2010. 10:729–745.
Article19. Bramati L, Aquilano F, Pietta P. Unfermented rooibos tea: quantitative characterization of flavonoids by HPLC-UV and determination of the total antioxidant activity. J Agric Food Chem. 2003. 51:7472–7474.
Article20. Awoniyi DO, Aboua YG, Marnewick J, Brooks N. The effects of rooibos (Aspalathus linearis), green tea (Camellia sinensis) and commercial rooibos and green tea supplements on epididymal sperm in oxidative stress-induced rats. Phytother Res. 2012. 26:1231–1239.
Article21. Marnewick JL, Rautenbach F, Venter I, Neethling H, Blackhurst DM, Wolmarans P, Macharia M. Effects of rooibos (Aspalathus linearis) on oxidative stress and biochemical parameters in adults at risk for cardiovascular disease. J Ethnopharmacol. 2011. 133:46–52.
Article22. Baba H, Ohtsuka Y, Haruna H, Lee T, Nagata S, Maeda M, Yamashiro Y, Shimizu T. Studies of anti-inflammatory effects of rooibos tea in rats. Pediatr Int. 2009. 51:700–704.
Article23. Tan RS, Pu SJ. Impact of obesity on hypogonadism in the andropause. Int J Androl. 2002. 25:195–201.
Article24. Vermeulen A, Kaufman JM. Ageing of the hypothalamo-pituitary-testicular axis in men. Horm Res. 1995. 43:25–28.
Article25. Vermeulen A. Environment, human reproduction, menopause, and andropause. Environ Health Perspect. 1993. 101:Suppl 2. 91–100.
Article26. Bornman MS, Reif S. Serum testosterone levels in South African men and the onset of androgen decline in ageing males. S Afr J Surg. 2007. 45:62. 64.27. Moon C, Kim JS, Jang H, Lee HJ, Kim SH, Kang SS, Bae CS, Kim JC, Kim S, Lee Y, Shin T. Activation of Akt/protein kinase B and extracellular signal-regulated kinase in rats with acute experimental testicular torsion. J Vet Med Sci. 2008. 70:337–341.
Article28. Kim JY, Han EH, Kim HG, Oh KN, Kim SK, Lee KY, Jeong HG. Bisphenol A-induced aromatase activation is mediated by cyclooxygenase-2 up-regulation in rat testicular Leydig cells. Toxicol Lett. 2010. 193:200–208.
Article29. Yamashita S, Tai P, Charron J, Ko C, Ascoli M. The Leydig cell MEK/ERK pathway is critical for maintaining a functional population of adult Leydig cells and for fertility. Mol Endocrinol. 2011. 25:1211–1222.
Article30. Walsh PC, Swerdloff RS. Biphasic effect of testosterone on spermatogenesis in the rat. Invest Urol. 1973. 11:190–193.31. Chen H, Liu J, Luo L, Zirkin BR. Dibutyryl cyclic adenosine monophosphate restores the ability of aged Leydig cells to produce testosterone at the high levels characteristic of young cells. Endocrinology. 2004. 145:4441–4446.
Article32. Horstman AM, Dillon EL, Urban RJ, Sheffield-Moore M. The role of androgens and estrogens on healthy aging and longevity. J Gerontol A Biol Sci Med Sci. 2012. 67:1140–1152.
Article33. Bormann CL, Smith GD, Padmanabhan V, Lee TM. Prenatal testosterone and dihydrotestosterone exposure disrupts ovine testicular development. Reproduction. 2011. 142:167–173.
Article34. Sofikitis N, Giotitsas N, Tsounapi P, Baltogiannis D, Giannakis D, Pardalidis N. Hormonal regulation of spermatogenesis and spermiogenesis. J Steroid Biochem Mol Biol. 2008. 109:323–330.
Article35. Schwartz E, Holtorf K. Hormone replacement therapy in the geriatric patient: current state of the evidence and questions for the future. Estrogen, progesterone, testosterone, and thyroid hormone augmentation in geriatric clinical practice: part 1. Clin Geriatr Med. 2011. 27:541–559.
Article36. Schwartz E, Morelli V, Holtorf K. Hormone replacement therapy in the geriatric patient: current state of the evidence and questions for the future--estrogen, progesterone, testosterone, and thyroid hormone augmentation in geriatric clinical practice: part 2. Clin Geriatr Med. 2011. 27:561–575.
Article37. Heinemann LA. Aging Males' Symptoms scale: a standardized instrument for the practice. J Endocrinol Invest. 2005. 28:34–38.38. Miwa Y, Kaneda T, Yokoyama O. Correlation between the Aging Males' Symptoms scale and sex steroids, gonadotropins, dehydroepiandrosterone sulfate, and growth hormone levels in ambulatory men. J Sex Med. 2006. 3:723–726.
Article39. Kawa G, Taniguchi H, Kinoshita H, Matsuda T, Urakami M, Sawaragi I. Aging Male Symptoms and serum testosterone levels in healthy Japanese middle-aged men. Nihon Hinyokika Gakkai Zasshi. 2008. 99:645–651.
Article