Nutr Res Pract.
2018 Apr;12(2):101-109. 10.4162/nrp.2018.12.2.101.
Effect of vitamin C on azoxymethane (AOM)/dextran sulfate sodium (DSS)-induced colitis-associated early colon cancer in mice
- Affiliations
-
- 1Department of Nutritional Science and Food Management, Ewha Womans University, 52 Ewhayeodae-gil, Seodaemun-gu, Seoul 03760, Korea. yuri.kim@ewha.ac.kr
- 2Department of Biomedical Laboratory Science, Eulji University, Gyeonggi 13135, Korea.
- 3Kwang-Dong Pharmaceutical Co., Ltd., Seoul 06650, Korea.
- KMID: 2407806
- DOI: http://doi.org/10.4162/nrp.2018.12.2.101
Abstract
- BACKGROUND/OBJECTIVES
The objective of this study was to investigate the effects of vitamin C on inflammation, tumor development, and dysbiosis of intestinal microbiota in an azoxymethane (AOM)/dextran sulfate sodium (DSS)-induced inflammation-associated early colon cancer mouse model.
MATERIALS/METHODS
Male BALB/c mice were injected intraperitoneally with AOM [10 mg/kg body weight (b.w)] and given two 7-d cycles of 2% DSS drinking water with a 14 d inter-cycle interval. Vitamin C (60 mg/kg b.w. and 120 mg/kg b.w.) was supplemented by gavage for 5 weeks starting 2 d after the AOM injection.
RESULTS
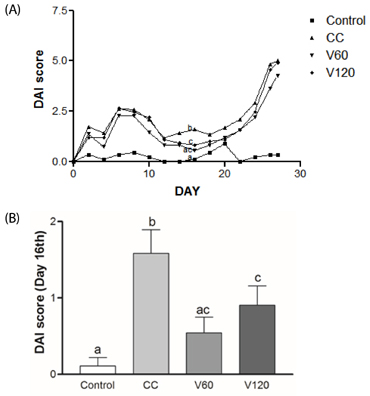
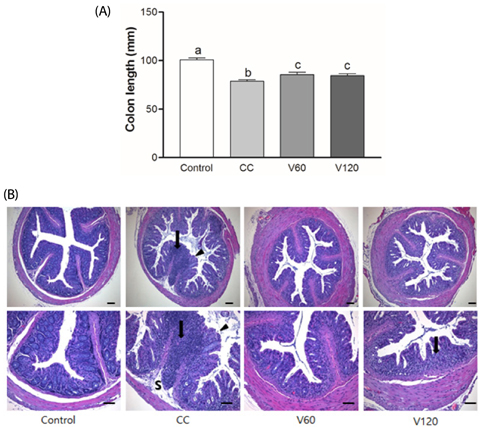
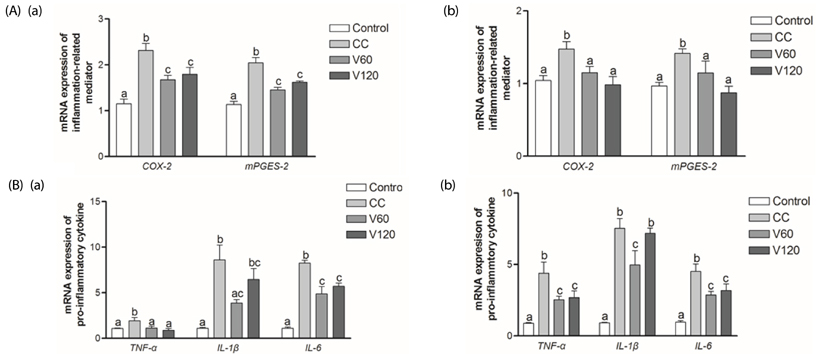
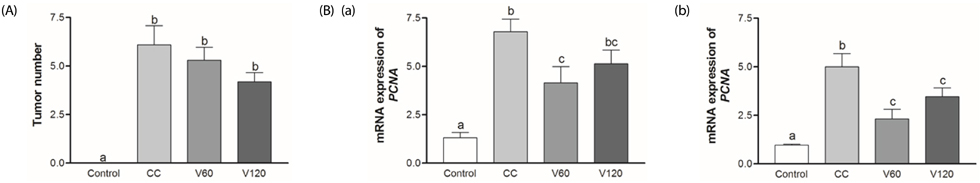
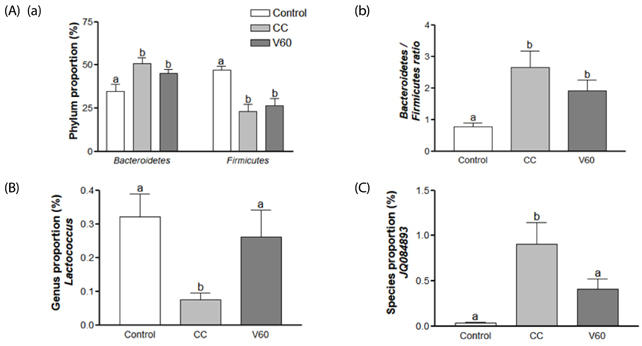
The vitamin C treatment suppressed inflammatory morbidity, as reflected by disease activity index (DAI) in recovery phase and inhibited shortening of the colon, and reduced histological damage. In addition, vitamin C supplementation suppressed mRNA levels of pro-inflammatory mediators and cytokines, including cyclooxygenase-2, microsomal prostaglandin E synthase-2, tumor necrosis factor-α, Interleukin (IL)-1β, and IL-6, and reduced expression of the proliferation marker, proliferating cell nuclear antigen, compared to observations of AOM/DSS animals. Although the microbial composition did not differ significantly between the groups, administration of vitamin C improved the level of inflammation-related Lactococcus and JQ084893 to control levels.
CONCLUSION
Vitamin C treatment provided moderate suppression of inflammation, proliferation, and certain inflammation-related dysbiosis in a murine model of colitis associated-early colon cancer. These findings support that vitamin C supplementation can benefit colonic health. Long-term clinical studies with various doses of vitamin C are warranted.
Keyword
MeSH Terms
-
Animals
Ascorbic Acid*
Azoxymethane*
Body Weight
Colitis
Colon*
Colonic Neoplasms*
Cyclooxygenase 2
Cytokines
Drinking Water
Dysbiosis
Gastrointestinal Microbiome
Humans
Inflammation
Interleukin-6
Interleukins
Lactococcus
Male
Mice*
Microbiota
Necrosis
Proliferating Cell Nuclear Antigen
RNA, Messenger
Sodium*
Vitamins*
Ascorbic Acid
Azoxymethane
Cyclooxygenase 2
Cytokines
Drinking Water
Interleukin-6
Interleukins
Proliferating Cell Nuclear Antigen
RNA, Messenger
Sodium
Vitamins
Figure
Reference
-
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66:7–30.
Article2. Boyle P, Langman JS. ABC of colorectal cancer: Epidemiology. BMJ. 2000; 321:805–808.
Article3. Bernstein CN, Blanchard JF, Kliewer E, Wajda A. Cancer risk in patients with inflammatory bowel disease: a population-based study. Cancer. 2001; 91:854–862.
Article4. Itzkowitz SH, Yio X. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004; 287:G7–G17.
Article5. Tanaka T. Development of an inflammation-associated colorectal cancer model and its application for research on carcinogenesis and chemoprevention. Int J Inflam. 2012; 2012:658786.
Article6. Meira LB, Bugni JM, Green SL, Lee CW, Pang B, Borenshtein D, Rickman BH, Rogers AB, Moroski-Erkul CA, McFaline JL, Schauer DB, Dedon PC, Fox JG, Samson LD. DNA damage induced by chronic inflammation contributes to colon carcinogenesis in mice. J Clin Invest. 2008; 118:2516–2525.
Article7. Strober W, Fuss IJ. Proinflammatory cytokines in the pathogenesis of inflammatory bowel diseases. Gastroenterology. 2011; 140:1756–1767.
Article8. Szkaradkiewicz A, Marciniak R, Chudzicka-Strugala I, Wasilewska A, Drews M, Majewski P, Karpiński T, Zwoździak B. Proinflammatory cytokines and IL-10 in inflammatory bowel disease and colorectal cancer patients. Arch Immunol Ther Exp (Warsz). 2009; 57:291–294.
Article9. Turnbaugh PJ, Ley RE, Hamady M, Fraser-Liggett CM, Knight R, Gordon JI. The human microbiome project. Nature. 2007; 449:804–810.
Article10. Laukens D, Brinkman BM, Raes J, De Vos M, Vandenabeele P. Heterogeneity of the gut microbiome in mice: guidelines for optimizing experimental design. FEMS Microbiol Rev. 2016; 40:117–132.
Article11. Kamada N, Seo SU, Chen GY, Nunez G. Role of the gut microbiota in immunity and inflammatory disease. Nat Rev Immunol. 2013; 13:321–335.
Article12. Holmes E, Li JV, Athanasiou T, Ashrafian H, Nicholson JK. Understanding the role of gut microbiome-host metabolic signal disruption in health and disease. Trends Microbiol. 2011; 19:349–359.
Article13. Mai V, Morris JG Jr. Colonic bacterial flora: changing understandings in the molecular age. J Nutr. 2004; 134:459–464.
Article14. Dueñas M, Muñoz-González I, Cueva C, Jiménez-Girón A, Sánchez-Patán F, Santos-Buelga C, Moreno-Arribas MV, Bartolomé B. A survey of modulation of gut microbiota by dietary polyphenols. Biomed Res Int. 2015; 2015:850902.
Article15. Biesalski HK. Nutrition meets the microbiome: micronutrients and the microbiota. Ann N Y Acad Sci. 2016; 1372:53–64.
Article16. Nishikimi M, Yagi K. Molecular basis for the deficiency in humans of gulonolactone oxidase, a key enzyme for ascorbic acid biosynthesis. Am J Clin Nutr. 1991; 54:1203S–1208S.
Article17. Jacob RA, Sotoudeh G. Vitamin C function and status in chronic disease. Nutr Clin Care. 2002; 5:66–74.
Article18. Block G. Vitamin C and cancer prevention: the epidemiologic evidence. Am J Clin Nutr. 1991; 53:270S–282S.
Article19. Deicher R, Ziai F, Bieglmayer C, Schillinger M, Horl WH. Low total vitamin C plasma level is a risk factor for cardiovascular morbidity and mortality in hemodialysis patients. J Am Soc Nephrol. 2005; 16:1811–1818.
Article20. Wang X, Willen R, Wadstrom T. Astaxanthin-rich algal meal and vitamin C inhibit Helicobacter pylori infection in BALB/cA mice. Antimicrob Agents Chemother. 2000; 44:2452–2457.
Article21. Korantzopoulos P, Kolettis TM, Kountouris E, Dimitroula V, Karanikis P, Pappa E, Siogas K, Goudevenos JA. Oral vitamin C administration reduces early recurrence rates after electrical cardioversion of persistent atrial fibrillation and attenuates associated inflammation. Int J Cardiol. 2005; 102:321–326.
Article22. Kasetsuwan N, Wu FM, Hsieh F, Sanchez D, McDonnell PJ. Effect of topical ascorbic acid on free radical tissue damage and inflammatory cell influx in the cornea after excimer laser corneal surgery. Arch Ophthalmol. 1999; 117:649–652.
Article23. Yan H, Wang H, Zhang X, Li X, Yu J. Ascorbic acid ameliorates oxidative stress and inflammation in dextran sulfate sodium-induced ulcerative colitis in mice. Int J Clin Exp Med. 2015; 8:20245–20253.24. Remely M, Ferk F, Sterneder S, Setayesh T, Roth S, Kepcija T, Noorizadeh R, Rebhan I, Greunz M, Beckmann J, Wagner KH, Knasmüller S, Haslberger AG. EGCG prevents high fat diet-induced changes in gut microbiota, decreases of DNA strand breaks, and changes in expression and DNA methylation of Dnmt1 and MLH1 in C57BL/6J male mice. Oxid Med Cell Longev. 2017; 2017:3079148.25. Xu J, Xu C, Chen X, Cai X, Yang S, Sheng Y, Wang T. Regulation of an antioxidant blend on intestinal redox status and major microbiota in early weaned piglets. Nutrition. 2014; 30:584–589.
Article26. Yu C, Wen XD, Zhang Z, Zhang CF, Wu XH, Martin A, Du W, He TC, Wang CZ, Yuan CS. American ginseng attenuates azoxymethane/dextran sodium sulfate-induced colon carcinogenesis in mice. J Ginseng Res. 2015; 39:14–21.
Article27. Kim KM, Kim YS, Lim JY, Min SJ, Shin JH, Ko HC, Kim SJ, Lim Y, Kim Y. Sasa quelpaertensis leaf extract suppresses dextran sulfate sodium-induced colitis in mice by inhibiting the proinflammatory mediators and mitogen-activated protein kinase phosphorylation. Nutr Res. 2014; 34:894–905.
Article28. Okayasu I, Hatakeyama S, Yamada M, Ohkusa T, Inagaki Y, Nakaya R. A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology. 1990; 98:694–702.
Article29. Dieleman LA, Palmen MJ, Akol H, Bloemena E, Peña AS, Meuwissen SG, Van Rees EP. Chronic experimental colitis induced by dextran sulphate sodium (DSS) is characterized by Th1 and Th2 cytokines. Clin Exp Immunol. 1998; 114:385–391.
Article30. Araki A, Kanai T, Ishikura T, Makita S, Uraushihara K, Iiyama R, Totsuka T, Takeda K, Akira S, Watanabe M. MyD88-deficient mice develop severe intestinal inflammation in dextran sodium sulfate colitis. J Gastroenterol. 2005; 40:16–23.
Article31. Wen XD, Wang CZ, Yu C, Zhao L, Zhang Z, Matin A, Wang Y, Li P, Xiao SY, Du W, He TC, Yuan CS. Panax notoginseng attenuates experimental colitis in the azoxymethane/dextran sulfate sodium mouse model. Phytother Res. 2014; 28:892–898.
Article32. Sánchez-Fidalgo S, Cárdeno A, Sánchez-Hidalgo M, Aparicio-Soto M, de la Lastra CA. Dietary extra virgin olive oil polyphenols supplementation modulates DSS-induced chronic colitis in mice. J Nutr Biochem. 2013; 24:1401–1413.
Article33. Tanaka T, Kohno H, Suzuki R, Yamada Y, Sugie S, Mori H. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci. 2003; 94:965–973.
Article34. Wang D, Dubois RN. The role of COX-2 in intestinal inflammation and colorectal cancer. Oncogene. 2010; 29:781–788.
Article35. Sheibanie AF, Yen JH, Khayrullina T, Emig F, Zhang M, Tuma R, Ganea D. The proinflammatory effect of prostaglandin E2 in experimental inflammatory bowel disease is mediated through the IL-23--〉IL-17 axis. J Immunol. 2007; 178:8138–8147.
Article36. Wang D, Dubois RN. Prostaglandins and cancer. Gut. 2006; 55:115–122.
Article37. Gudis K, Tatsuguchi A, Wada K, Futagami S, Nagata K, Hiratsuka T, Shinji Y, Miyake K, Tsukui T, Fukuda Y, Sakamoto C. Microsomal prostaglandin E synthase (mPGES)-1, mPGES-2 and cytosolic PGES expression in human gastritis and gastric ulcer tissue. Lab Invest. 2005; 85:225–236.
Article38. Murakami M, Nakashima K, Kamei D, Masuda S, Ishikawa Y, Ishii T, Ohmiya Y, Watanabe K, Kudo I. Cellular prostaglandin E2 production by membrane-bound prostaglandin E synthase-2 via both cyclooxygenases-1 and -2. J Biol Chem. 2003; 278:37937–37947.
Article39. Meduri GU, Headley S, Kohler G, Stentz F, Tolley E, Umberger R, Leeper K. Persistent elevation of inflammatory cytokines predicts a poor outcome in ARDS. Plasma IL-1 beta and IL-6 levels are consistent and efficient predictors of outcome over time. Chest. 1995; 107:1062–1073.
Article40. Härtel C, Strunk T, Bucsky P, Schultz C. Effects of vitamin C on intracytoplasmic cytokine production in human whole blood monocytes and lymphocytes. Cytokine. 2004; 27:101–106.
Article41. Jung IK, Choi HJ, Kim SS, Hong SH. Prognostic significance of proliferating cell nuclear antigen (PCNA) expression in patients with colorectal carcinoma. J Korean Cancer Assoc. 1995; 27:550–558.42. Suzuki R, Kohno H, Sugie S, Tanaka T. Dose-dependent promoting effect of dextran sodium sulfate on mouse colon carcinogenesis initiated with azoxymethane. Histol Histopathol. 2005; 20:483–492.43. De Robertis M, Massi E, Poeta ML, Carotti S, Morini S, Cecchetelli L, Signori E, Fazio VM. The AOM/DSS murine model for the study of colon carcinogenesis: From pathways to diagnosis and therapy studies. J Carcinog. 2011; 10:9.
Article44. Onodera H, Maetani S, Kawamoto K, Kan S, Kondo S, Imamura M. Pathologic significance of tumor progression in locally recurrent rectal cancer: different nature from primary cancer. Dis Colon Rectum. 2000; 43:775–781.
Article45. Jackson PE, O'Connor PJ, Cooper DP, Margison GP, Povey AC. Associations between tissue-specific DNA alkylation, DNA repair and cell proliferation in the colon and colon tumour yield in mice treated with 1,2-dimethylhydrazine. Carcinogenesis. 2003; 24:527–533.
Article46. Bussey HJ, DeCosse JJ, Deschner EE, Eyers AA, Lesser ML, Morson BC, Ritchie SM, Thomson JP, Wadsworth J. A randomized trial of ascorbic acid in polyposis coli. Cancer. 1982; 50:1434–1439.
Article47. Jacobs EJ, Connell CJ, Patel AV, Chao A, Rodriguez C, Seymour J, McCullough ML, Calle EE, Thun MJ. Vitamin C and vitamin E supplement use and colorectal cancer mortality in a large American Cancer Society cohort. Cancer Epidemiol Biomarkers Prev. 2001; 10:17–23.48. Cahill RJ, O'Sullivan KR, Mathias PM, Beattie S, Hamilton H, O'Morain C. Effects of vitamin antioxidant supplementation on cell kinetics of patients with adenomatous polyps. Gut. 1993; 34:963–967.
Article49. Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J. 2008; 22:659–661.
Article50. Levine M, Conry-Cantilena C, Wang Y, Welch RW, Washko PW, Dhariwal KR, Park JB, Lazarev A, Graumlich JF, King J, Cantilena LR. Vitamin C pharmacokinetics in healthy volunteers: evidence for a recommended dietary allowance. Proc Natl Acad Sci U S A. 1996; 93:3704–3709.
Article51. Carr AC, Frei B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am J Clin Nutr. 1999; 69:1086–1107.
Article52. Jenab M, Riboli E, Ferrari P, Sabate J, Slimani N, Norat T, Friesen M, Tjønneland A, Olsen A, Overvad K, Boutron-Ruault MC, Clavel-Chapelon F, Touvier M, Boeing H, Schulz M, Linseisen J, Nagel G, Trichopoulou A, Naska A, Oikonomou E, Krogh V, Panico S, Masala G, Sacerdote C, Tumino R, Peeters PH, Numans ME, Bueno-de-Mesquita HB, Büchner FL, Lund E, Pera G, Sanchez CN, Sánchez MJ, Arriola L, Barricarte A, Quirós JR, Hallmans G, Stenling R, Berglund G, Bingham S, Khaw KT, Key T, Allen N, Carneiro F, Mahlke U, Del Giudice G, Palli D, Kaaks R, Gonzalez CA. Plasma and dietary vitamin C levels and risk of gastric cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC-EURGAST). Carcinogenesis. 2006; 27:2250–2257.
Article53. Strnadová E, Prokopic J. Changes in ascorbic acid content in various organs and serum of mice experimentally infected with Taenia crassiceps (Zeder, 1800) cysticerci. Folia Parasitol (Praha). 1985; 32:185–188.54. Mosca A, Leclerc M, Hugot JP. Gut microbiota diversity and human diseases: should we reintroduce key predators in our ecosystem? Front Microbiol. 2016; 7:455.
Article55. Louis P, Hold GL, Flint HJ. The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol. 2014; 12:661–672.
Article56. Ott SJ, Musfeldt M, Wenderoth DF, Hampe J, Brant O, Fölsch UR, Timmis KN, Schreiber S. Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut. 2004; 53:685–693.
Article57. Claesson MJ, Jeffery IB, Conde S, Power SE, O'Connor EM, Cusack S, Harris HM, Coakley M, Lakshminarayanan B, O'Sullivan O, Fitzgerald GF, Deane J, O'Connor M, Harnedy N, O'Connor K, O'Mahony D, van Sinderen D, Wallace M, Brennan L, Stanton C, Marchesi JR, Fitzgerald AP, Shanahan F, Hill C, Ross RP, O'Toole PW. Gut microbiota composition correlates with diet and health in the elderly. Nature. 2012; 488:178–184.
Article58. Ahn J, Sinha R, Pei Z, Dominianni C, Wu J, Shi J, Goedert JJ, Hayes RB, Yang L. Human gut microbiome and risk for colorectal cancer. J Natl Cancer Inst. 2013; 105:1907–1911.
Article59. Zackular JP, Baxter NT, Iverson KD, Sadler WD, Petrosino JF, Chen GY, Schloss PD. The gut microbiome modulates colon tumorigenesis. MBio. 2013; 4:e00692–e00613.
Article60. Berry D, Kuzyk O, Rauch I, Heider S, Schwab C, Hainzl E, Decker T, Müller M, Strobl B, Schleper C, Urich T, Wagner M, Kenner L, Loy A. Intestinal microbiota signatures associated with inflammation history in mice experiencing recurring colitis. Front Microbiol. 2015; 6:1408.
Article61. Yeom Y, Kim BS, Kim SJ, Kim Y. Sasa quelpaertensis leaf extract regulates microbial dysbiosis by modulating the composition and diversity of the microbiota in dextran sulfate sodium-induced colitis mice. BMC Complement Altern Med. 2016; 16:481.
Article62. Chen HM, Yu YN, Wang JL, Lin YW, Kong X, Yang CQ, Yang L, Liu ZJ, Yuan YZ, Liu F, Wu JX, Zhong L, Fang DC, Zou W, Fang JY. Decreased dietary fiber intake and structural alteration of gut microbiota in patients with advanced colorectal adenoma. Am J Clin Nutr. 2013; 97:1044–1052.
Article63. Morrison DJ, Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes. 2016; 7:189–200.
Article64. Hemarajata P, Versalovic J. Effects of probiotics on gut microbiota: mechanisms of intestinal immunomodulation and neuromodulation. Therap Adv Gastroenterol. 2013; 6:39–51.
Article65. de Moreno de LeBlanc A, LeBlanc JG, Perdigón G, Miyoshi A, Langella P, Azevedo V, Sesma F. Oral administration of a catalase-producing Lactococcus lactis can prevent a chemically induced colon cancer in mice. J Med Microbiol. 2008; 57:100–105.
Article66. David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE, Ling AV, Devlin AS, Varma Y, Fischbach MA, Biddinger SB, Dutton RJ, Turnbaugh PJ. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014; 505:559–563.
Article67. Willing BP, Dicksved J, Halfvarson J, Andersson AF, Lucio M, Zheng Z, Järnerot G, Tysk C, Jansson JK, Engstrand L. A pyrosequencing study in twins shows that gastrointestinal microbial profiles vary with inflammatory bowel disease phenotypes. Gastroenterology. 2010; 139:1844–1854.e1.
Article68. Lozupone CA, Stombaugh JI, Gordon JI, Jansson JK, Knight R. Diversity, stability and resilience of the human gut microbiota. Nature. 2012; 489:220–230.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Selenium on Colon Carcinogenesis Induced by Azoxymethane and Dextran Sodium Sulfate in Mouse Model with High-Iron Diet
- Nuclear Factor Erythroid 2-related Factor 2 Knockout Suppresses the Development of Aggressive Colorectal Cancer Formation Induced by Azoxymethane/Dextran Sulfate Sodium-Treatment in Female Mice
- The Effect of Sex on the Azoxymethane/Dextran Sulfate Sodium-treated Mice Model of Colon Cancer
- Induction of colorectal carcinogenesis in the C57BL/6J and A/J mouse strains with a reduced DSS dose in the AOM/DSS model
- Dietary Selenium Supplement Prevents Colon Carcinogenesis Induced by Azoxymethane and Dextran Sodium Sulfate in ICR Mice