J Bone Metab.
2017 Nov;24(4):207-215. 10.11005/jbm.2017.24.4.207.
Fexaramine Inhibits Receptor Activator of Nuclear Factor-κB Ligand-induced Osteoclast Formation via Nuclear Factor of Activated T Cells Signaling Pathways
- Affiliations
-
- 1College of Pharmacy, Sookmyung Women's University, Seoul, Korea. myim@sm.ac.kr
- KMID: 2397553
- DOI: http://doi.org/10.11005/jbm.2017.24.4.207
Abstract
- BACKGROUND
Osteoclasts are bone resorbing cells and are responsible for bone erosion in diseases as diverse as osteoporosis, periodontitis, and rheumatoid arthritis. Fexaramine has been developed as an agonist for the farnesoid X receptor (FXR). This study investigated the effects of fexaramine on receptor activator of nuclear factor (NF)-κB ligand (RANKL)-induced osteoclast formation and signaling pathways.
METHODS
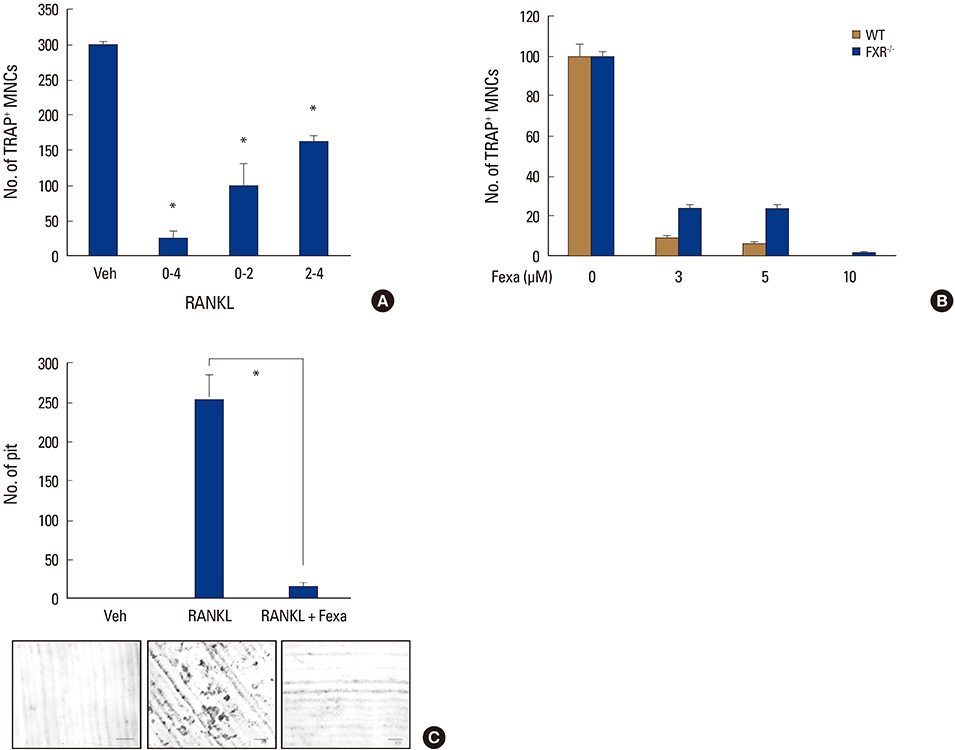
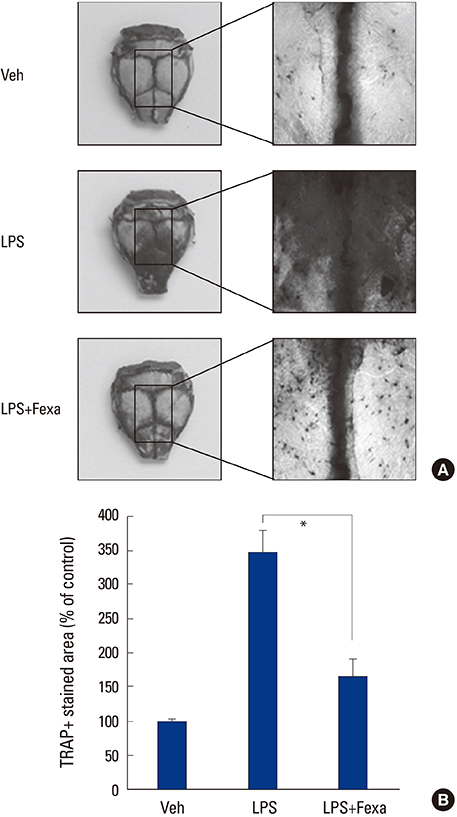
Osteoclasts were formed by culturing mouse bone marrow-derived macrophages (BMMs) with macrophage colony-stimulating factor (M-CSF) and RANKL. Bone resorption assays were performed using dentine slices. The mRNA expression level was analyzed by real-time polymerase chain reaction. Western blotting assays were conducted to detect the expression or activation level of proteins. Lipopolysaccharide-induced osteoclast formation was performed using a mouse calvarial model.
RESULTS
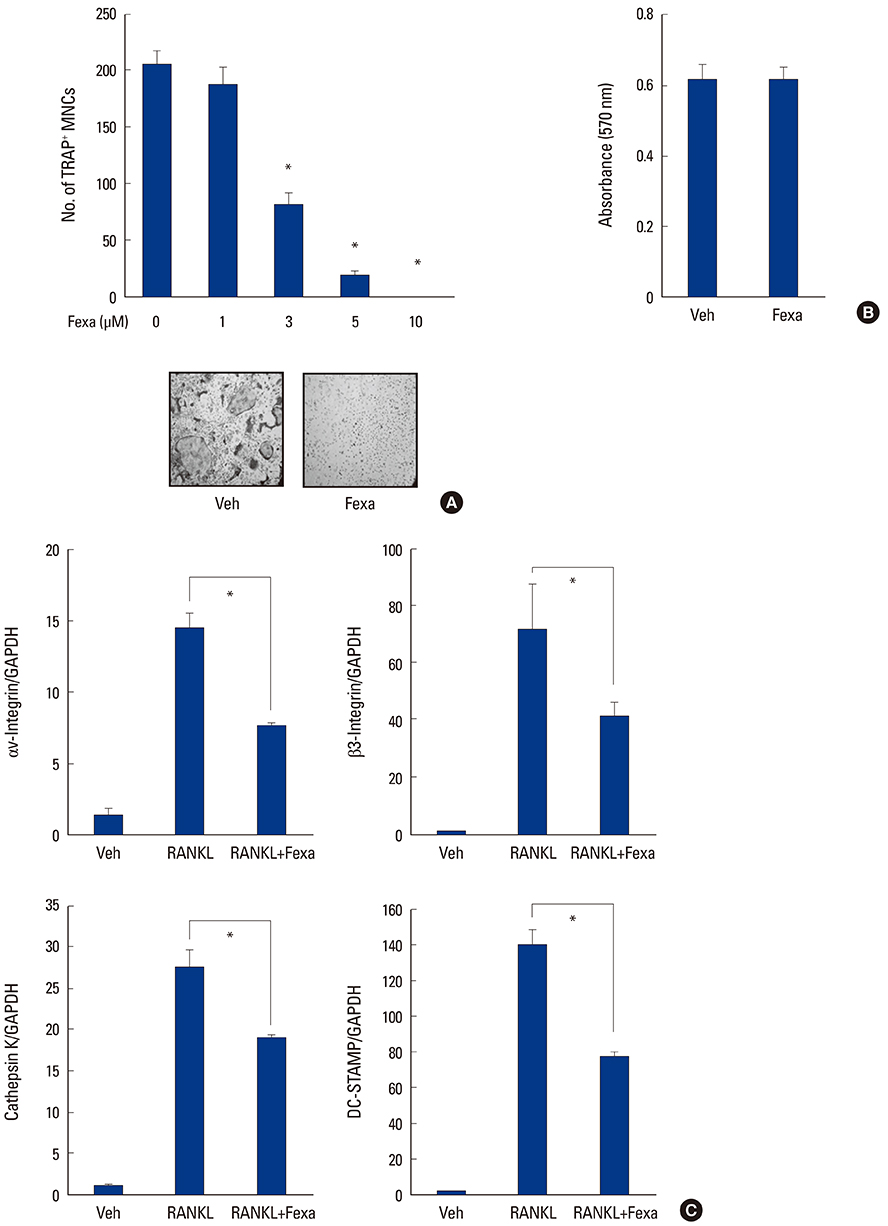
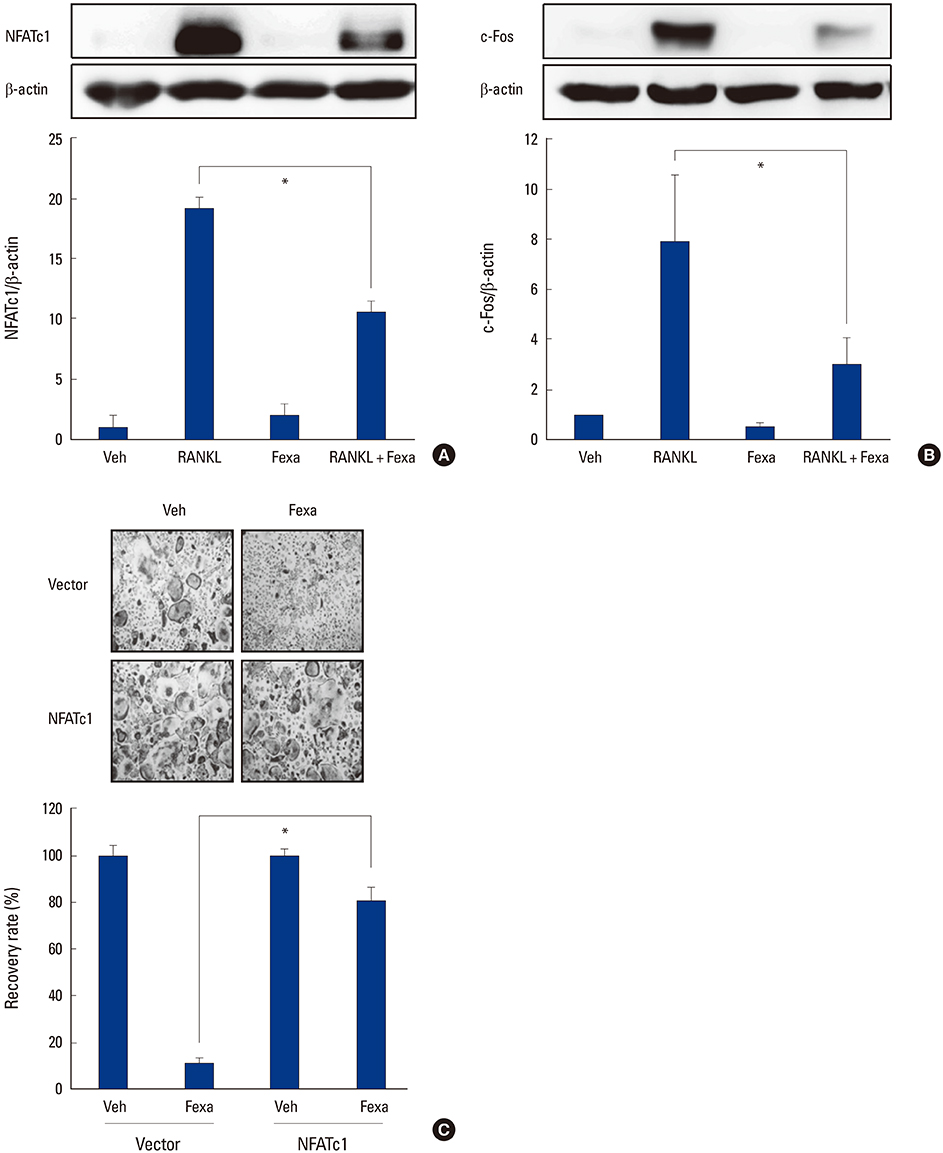
Fexaramine inhibited RANKL-induced osteoclast formation, without cytotoxicity. Furthermore, fexaramine diminished the RANKL-stimulated bone resorption. Mechanistically, fexaramine blocked the RANKL-triggered p38, extracellular signal-regulated kinase, and glycogen synthase kinase 3β phosphorylation, resulting in suppressed expression of c-Fos and NF of activated T cells (NFATc1). Consistent with the in vitro anti-osteoclastogenic effect, fexaramine suppressed lipopolysaccharide-induced osteoclast formation in the calvarial model.
CONCLUSIONS
The present data suggest that fexaramine has an inhibitory effect on osteoclast differentiation and function, via downregulation of NFATc1 signaling pathways. Thus, fexaramine could be useful for the treatment of bone diseases associated with excessive bone resorption.
Keyword
MeSH Terms
-
Animals
Arthritis, Rheumatoid
Blotting, Western
Bone Diseases
Bone Resorption
Dentin
Down-Regulation
Glycogen Synthase Kinases
In Vitro Techniques
Macrophage Colony-Stimulating Factor
Macrophages
Mice
NF-kappa B
Osteoclasts*
Osteoporosis
Periodontitis
Phosphorylation
Phosphotransferases
RANK Ligand
Real-Time Polymerase Chain Reaction
RNA, Messenger
T-Lymphocytes*
Glycogen Synthase Kinases
Macrophage Colony-Stimulating Factor
NF-kappa B
Phosphotransferases
RANK Ligand
RNA, Messenger
Figure
Reference
-
1. Zaidi M. Skeletal remodeling in health and disease. Nat Med. 2007; 13:791–801.
Article2. Rodan GA, Martin TJ. Therapeutic approaches to bone diseases. Science. 2000; 289:1508–1514.
Article3. Suda T, Takahashi N, Udagawa N, et al. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr Rev. 1999; 20:345–357.
Article4. Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003; 423:337–342.
Article5. Wong BR, Rho J, Arron J, et al. TRANCE is a novel ligand of the tumor necrosis factor receptor family that activates c-Jun N-terminal kinase in T cells. J Biol Chem. 1997; 272:25190–25194.
Article6. Theill LE, Boyle WJ, Penninger JM. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annu Rev Immunol. 2002; 20:795–823.
Article7. Lee ZH, Kim HH. Signal transduction by receptor activator of nuclear factor kappa B in osteoclasts. Biochem Biophys Res Commun. 2003; 305:211–214.
Article8. Jang HD, Shin JH, Park DR, et al. Inactivation of glycogen synthase kinase-3beta is required for osteoclast differentiation. J Biol Chem. 2011; 286:39043–39050.
Article9. Matsuo K, Galson DL, Zhao C, et al. Nuclear factor of activated T-cells (NFAT) rescues osteoclastogenesis in precursors lacking c-Fos. J Biol Chem. 2004; 279:26475–26480.
Article10. Wagner EF, Eferl R. Fos/AP-1 proteins in bone and the immune system. Immunol Rev. 2005; 208:126–140.
Article11. Asagiri M, Sato K, Usami T, et al. Autoamplification of NFATc1 expression determines its essential role in bone homeostasis. J Exp Med. 2005; 202:1261–1269.
Article12. Takayanagi H, Kim S, Koga T, et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell. 2002; 3:889–901.
Article13. Downes M, Verdecia MA, Roecker AJ, et al. A chemical, genetic, and structural analysis of the nuclear bile acid receptor FXR. Mol Cell. 2003; 11:1079–1092.
Article14. Wang YD, Chen WD, Moore DD, et al. FXR: a metabolic regulator and cell protector. Cell Res. 2008; 18:1087–1095.
Article15. Li T, Chiang JY. Bile acids as metabolic regulators. Curr Opin Gastroenterol. 2015; 31:159–165.
Article16. Cho SW, An JH, Park H, et al. Positive regulation of osteogenesis by bile acid through FXR. J Bone Miner Res. 2013; 28:2109–2121.
Article17. Ha H, Lee JH, Kim HN, et al. Alpha-Lipoic acid inhibits inflammatory bone resorption by suppressing prostaglandin E2 synthesis. J Immunol. 2006; 176:111–117.
Article18. Id Boufker H, Lagneaux L, Fayyad-Kazan H, et al. Role of farnesoid X receptor (FXR) in the process of differentiation of bone marrow stromal cells into osteoblasts. Bone. 2011; 49:1219–1231.
Article19. Kim HJ, Ohk B, Kang WY, et al. Deficiency of lipocalin-2 promotes proliferation and differentiation of osteoclast precursors via regulation of c-fms expression and nuclear factor-kappa B activation. J Bone Metab. 2016; 23:8–15.
Article20. Lim HS, Park YH, Kim SK. Relationship between serum inflammatory marker and bone mineral density in healthy adults. J Bone Metab. 2016; 23:27–33.
Article21. Gennari L, Rotatori S, Bianciardi S, et al. Treatment needs and current options for postmenopausal osteoporosis. Expert Opin Pharmacother. 2016; 17:1141–1152.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Humanin suppresses receptor activator of nuclear factor-κB ligand-induced osteoclast differentiation via AMP-activated protein kinase activation
- NF-kappaB-Mediated Regulation of Osteoclastogenesis
- Iris Koreana NAKAI Inhibits Osteoclast Formation via p38-Mediated Nuclear Factor of Activated T Cells 1 Signaling Pathway
- Signaling Pathways in Osteoclast Differentiation
- Effect of Cornus Officinalis on Receptor Activator of Nuclear Factor-kappaB Ligand (RANKL)-induced Osteoclast Differentiation