Nutr Res Pract.
2016 Jun;10(3):265-273. 10.4162/nrp.2016.10.3.265.
Effect of Hijikia fusiforme extracts on degenerative osteoarthritis in vitro and in vivo models
- Affiliations
-
- 1Department of Medical Nutrition, Kyung Hee University, 1732, Deogyeong-daero, Giheung-gu, Yongin 17104, Korea. Jlee2007@khu.ac.kr
- 2Division of Food and Nutritional Science, Chonnam National University, Gwangju 61186, Korea.
- KMID: 2342128
- DOI: http://doi.org/10.4162/nrp.2016.10.3.265
Abstract
- BACKGROUND/OBJECTIVES
The inhibitory effect of Hijikia fusiforme (HF) extracts on degenerative osteoarthritis was examined in primary cultured rat cartilage cells and a monosodium iodoacetate (MIA)-induced osteoarthritis rat model.
MATERIALS/METHODS
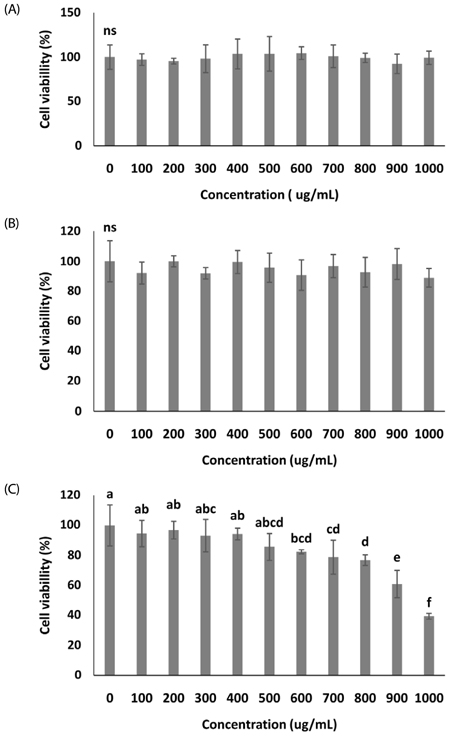
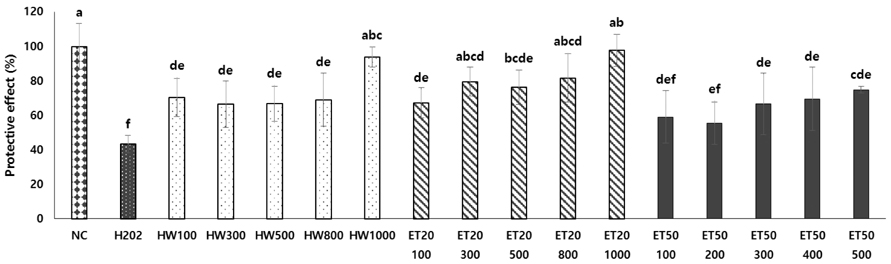
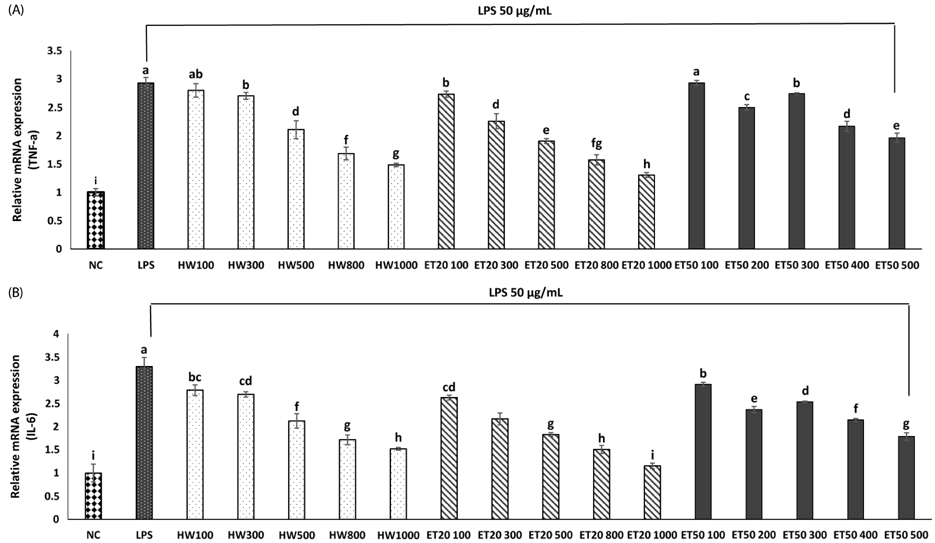
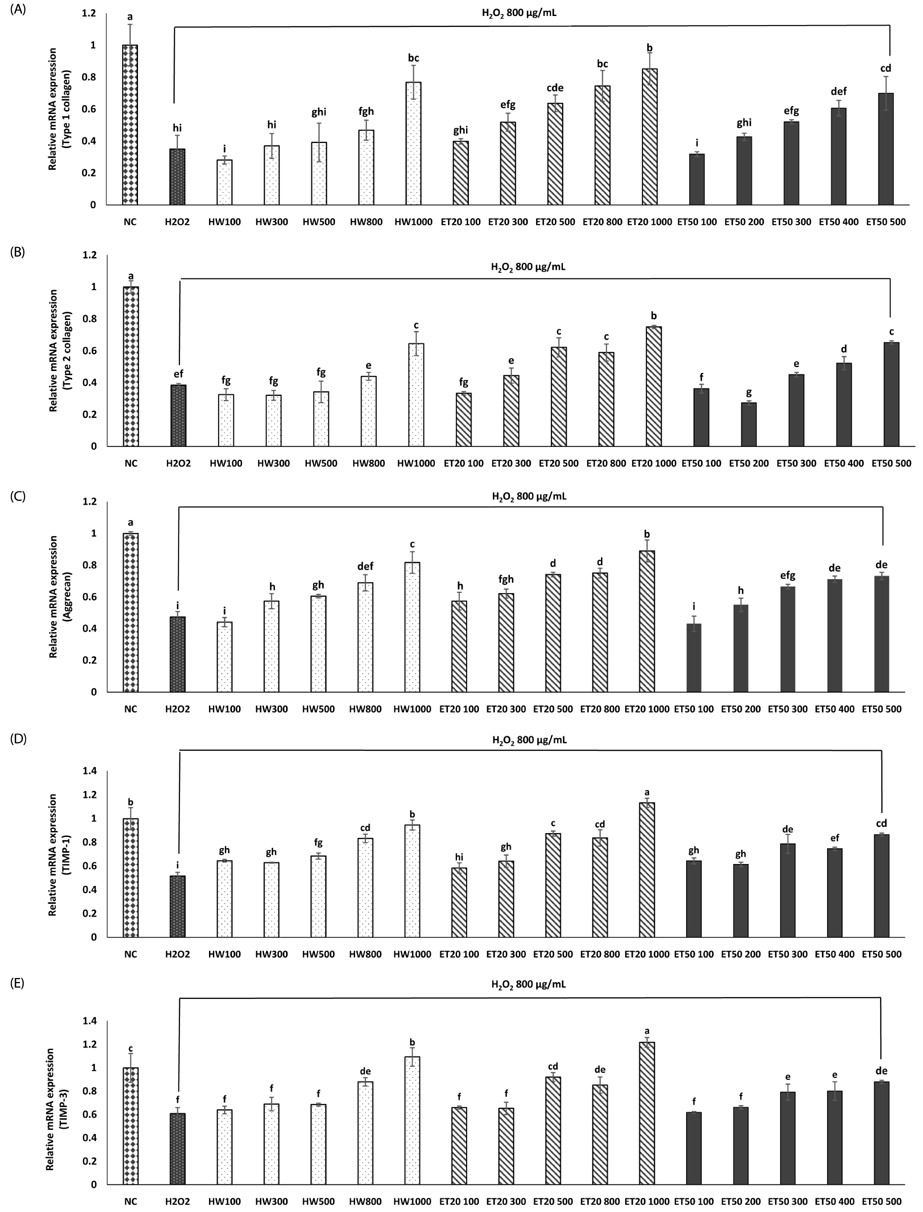
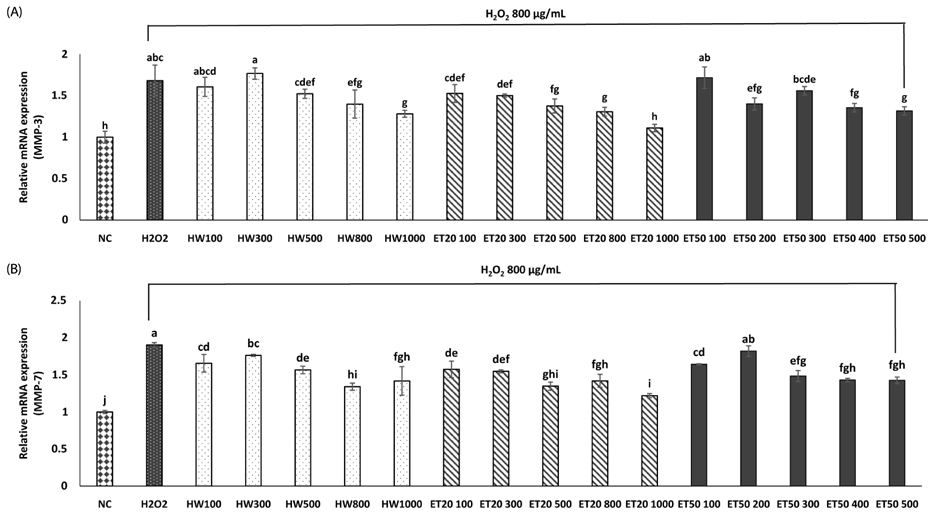
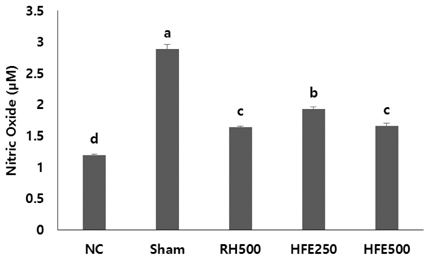
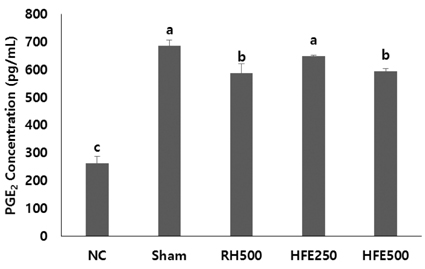
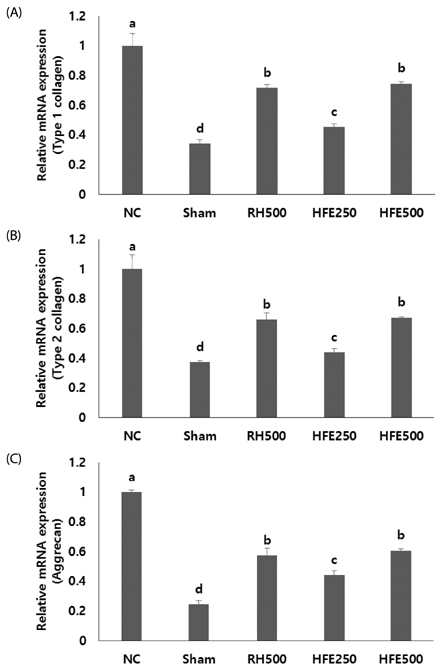
In vitro, cell survival and the expression of matrix metalloproteinases (MMPs), collagen type I, collagen type II, aggrecan, and tissue inhibitor of metalloproteinases (TIMPs) was measured after H2O2 (800 µM, 2 hr) treatment in primary chondrocytes. In vivo animal study, osteoarthritis was induced by intra-articular injection of MIA into knee joints of rats, and then RH500, HFE250 and HFE500 were administered orally once a day for 28 days. To determine the anti-inflammatory effects of HFE, nitric oxide (NO), prostaglandin E2 (PGE2) expression were measured. In addition, real-time PCR was performed to measure the genetic expression of MMPs, collagen type I, collagen type II, aggrecan, and TIMPs.
RESULTS
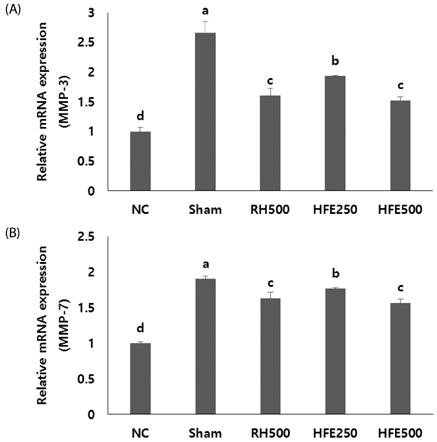
In the in vitro assay, cell survival after H2O2 treatment was increased by HFE extract (20% EtOH). In addition, anabolic factors (genetic expression of collagen type I, II, and aggrecan) were increased by HFE extract (20% EtOH). However, the genetic expression of MMP-3 and 7, known as catabolic factors were significantly inhibited by treatment with HFE extract (20% EtOH). In the in vivo assay, anabolic factors (genetic expression of collagen type I, II, aggrecan, and TIMPs) were increased by oral administration of HFE extract. However, the genetic expression of MMP-3 and 7, known as catabolic factors, and production of NO and PGE2 were significantly inhibited by treatment with oral administration of HFE extract.
CONCLUSIONS
HFE extract inhibited articular cartilage degeneration through preventing extracellular matrix degradation and chondrocyte injury.
Keyword
MeSH Terms
-
Administration, Oral
Aggrecans
Animals
Cartilage
Cartilage, Articular
Cell Survival
Chondrocytes
Collagen
Collagen Type I
Collagen Type II
Dinoprostone
Extracellular Matrix
In Vitro Techniques*
Injections, Intra-Articular
Knee Joint
Matrix Metalloproteinases
Models, Animal
Nitric Oxide
Osteoarthritis*
Rats
Real-Time Polymerase Chain Reaction
Tissue Inhibitor of Metalloproteinases
Aggrecans
Collagen
Collagen Type I
Collagen Type II
Dinoprostone
Matrix Metalloproteinases
Nitric Oxide
Tissue Inhibitor of Metalloproteinases
Figure
Reference
-
1. Ministry of Health and Welfare, Korea Centers for Disease Control and Prevention. Korea Health Statistics 2012: Korea National Health and Nutrition Examination Survey (KNHANES V-3). Cheongwon: Korea Centers for Disease Control and Prevention;2013.2. Statistics Korea. Senior Statistical Reports. Daejeon: Statistics Korea;2010.3. Garner BC, Stoker AM, Kuroki K, Evans R, Cook CR, Cook JL. Using animal models in osteoarthritis biomarker research. J Knee Surg. 2011; 24:251–264.
Article4. Nam DE, Kim OK, Shim TJ, Kim JH, Lee J. Effect of Boswellia serrata extracts on degenerative osteoarthritis in vitro and in vivo models. J Korean Soc Food Sci Nutr. 2014; 43:631–640.
Article5. Lee JH, Fitzgerald JB, Dimicco MA, Grodzinsky AJ. Mechanical injury of cartilage explants causes specific time-dependent changes in chondrocyte gene expression. Arthritis Rheum. 2005; 52:2386–2395.
Article6. Roy S, Khanna S, Krishnaraju AV, Subbaraju GV, Yasmin T, Bagchi D, Sen CK. Regulation of vascular responses to inflammation: inducible matrix metalloproteinase-3 expression in human microvascular endothelial cells is sensitive to antiinflammatory Boswellia. Antioxid Redox Signal. 2006; 8:653–660.
Article7. Lee V, Cao L, Zhang Y, Kiani C, Adams ME, Yang BB. The roles of matrix molecules in mediating chondrocyte aggregation, attachment, and spreading. J Cell Biochem. 2000; 79:322–333.
Article8. Korea Health Industry Development Institute. Health Functional Food Industry Development Assistance Report 2011. Cheongwon: Korea Health Industry Development Institute;2011.9. Jeon MH, Kim M. Effect of Hijikia fusiforme Fractions on Proliferation and Differentiation in Osteoblastic MC3T3-E1 Cells. J Life Sci. 2011; 21:300–308.
Article10. Kim JA, Lee JM. The change of biologically functional compounds and antioxidant activities in Hizikia fusiformis with drying methods. Korean J Food Cult. 2004; 19:200–208.11. Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier JP, Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011; 7:33–42.
Article12. Ministry for Food, Agriculture, Forestry and Fisheries. Korea Food Research Institute. Development of Instant Health Foods Using Hijiki. Gwacheon: Ministry for Food, Agriculture, Forestry and Fisheries;1997.13. Watanabe T, Hirayama T, Takahashi T, Kokubo T, Ikeda M. Toxicological evaluation of arsenic in edible seaweed, Hizikia species. Toxicology. 1979; 14:1–22.
Article14. Dobashi K, Nishino T, Fujihara M, Nagumo T. Isolation and preliminary characterization of fucose-containing sulfated polysaccharides with blood-anticoagulant activity from the brown seaweed Hizikia fusiforme. Carbohydr Res. 1989; 194:315–320.
Article15. Holan ZR, Volesky B. Biosorption of lead and nickel by biomass of marine algae. Biotechnol Bioeng. 1994; 43:1001–1009.
Article16. Jeong SC, Jeong YT, Lee SM, Kim JH. Immune-modulating activities of polysaccharides extracted from brown algae Hizikia fusiforme. Biosci Biotechnol Biochem. 2015; 79:1362–1365.
Article17. Mathy-Hartert M, Martin G, Devel P, Deby-Dupont G, Pujol JP, Reginster JY, Henrotin Y. Reactive oxygen species downregulate the expression of pro-inflammatory genes by human chondrocytes. Inflamm Res. 2003; 52:111–118.
Article18. Asada S, Fukuda K, Oh M, Hamanishi C, Tanaka S. Effect of hydrogen peroxide on the metabolism of articular chondrocytes. Inflamm Res. 1999; 48:399–403.
Article19. Khan IM, Gilbert SJ, Caterson B, Sandell LJ, Archer CW. Oxidative stress induces expression of osteoarthritis markers procollagen IIA and 3B3(-) in adult bovine articular cartilage. Osteoarthritis Cartilage. 2008; 16:698–707.
Article20. Catrina AI, Lampa J, Ernestam S, af Klint E, Bratt J, Klareskog L, Ulfgren AK. Anti-tumour necrosis factor (TNF)-alpha therapy (etanercept) down-regulates serum matrix metalloproteinase (MMP)-3 and MMP-1 in rheumatoid arthritis. Rheumatology (Oxford). 2002; 41:484–489.
Article21. Houssiau FA, Devogelaer JP, Van Damme J, de Deuxchaisnes CN, Van Snick J. Interleukin-6 in synovial fluid and serum of patients with rheumatoid arthritis and other inflammatory arthritides. Arthritis Rheum. 1988; 31:784–788.
Article22. Tchetverikov I, Lohmander LS, Verzijl N, Huizinga TW, TeKoppele JM, Hanemaaijer R, DeGroot J. MMP protein and activity levels in synovial fluid from patients with joint injury, inflammatory arthritis, and osteoarthritis. Ann Rheum Dis. 2005; 64:694–698.
Article23. Wink DA, Hines HB, Cheng RY, Switzer CH, Flores-Santana W, Vitek MP, Ridnour LA, Colton CA. Nitric oxide and redox mechanisms in the immune response. J Leukoc Biol. 2011; 89:873–891.
Article24. Thomas CM, Fuller CJ, Whittles CE, Sharif M. Chondrocyte death by apoptosis is associated with the initiation and severity of articular cartilage degradation. Int J Rheum Dis. 2011; 14:191–198.
Article25. Vane JR, Bakhle YS, Botting RM. Cyclooxygenases 1 and 2. Annu Rev Pharmacol Toxicol. 1998; 38:97–120.
Article26. Bensen WG, Fiechtner JJ, McMillen JI, Zhao WW, Yu SS, Woods EM, Hubbard RC, Isakson PC, Verburg KM, Geis GS. Treatment of osteoarthritis with celecoxib, a cyclooxygenase-2 inhibitor: a randomized controlled trial. Mayo Clin Proc. 1999; 74:1095–1105.
Article27. Hardy MM, Seibert K, Manning PT, Currie MG, Woerner BM, Edwards D, Koki A, Tripp CS. Cyclooxygenase 2-dependent prostaglandin E2 modulates cartilage proteoglycan degradation in human osteoarthritis explants. Arthritis Rheum. 2002; 46:1789–1803.
Article28. Combe R, Bramwell S, Field MJ. The monosodium iodoacetate model of osteoarthritis: a model of chronic nociceptive pain in rats? Neurosci Lett. 2004; 370:236–240.
Article29. Kim MJ, Lee HH, Jeong JW, Seo MJ, Kang BW, Park JU, Kim KS, Cho YS, Seo KI, Kim GY, Kim JI, Choi YH, Jeong YK. Anti-inflammatory effects of 5-hydroxy-3,6,7,8,3',4'-hexamethoxyflavone via NF-κB inactivation in lipopolysaccharide-stimulated RAW 264.7. Mol Med Rep. 2014; 9:1197–1203.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Dietary Calcium Level and Hijikia fusiforme Supplementation on Bone Indices and Serum Lipid Levels in Ovariectomized Rats
- Effect of Hizikia Fusiforme Water Extracts on Mouse Immune Cell Activation
- Effects of Cudrania tricuspidata and Sargassum fusiforme extracts on hair growth in C57BL/6 mice
- Promotional effects of Sargassum fusiforme fractions on hair growth via in vitro and in vivo models
- Anti-Inflammatory Herbal Extracts and Their Drug Discovery Perspective in Atopic Dermatitis