Nutr Res Pract.
2015 Dec;9(6):569-578. 10.4162/nrp.2015.9.6.569.
Antioxidant activity and anti-inflammatory activity of ethanol extract and fractions of Doenjang in LPS-stimulated RAW 264.7 macrophages
- Affiliations
-
- 1Institute on Aging, Seoul National University, #304 Biomedical Science building, 103 Daehak-ro, Jongno-gu, Seoul 110-799, Korea. kwakcs@snu.ac.kr
- 2Department of Biotechnology, Hoseo University, Chungnam 336-795, Korea.
- 3Department of Food and Nutrition, Seoul National University, Seoul 151-742, Korea.
- KMID: 2313879
- DOI: http://doi.org/10.4162/nrp.2015.9.6.569
Abstract
- BACKGROUND/OBJECTIVES
Fermentation can increase functional compounds in fermented soybean products, thereby improving antioxidant and/or anti-inflammatory activities. We investigated the changes in the contents of phenolics and isoflavones, antioxidant activity and anti-inflammatory activity of Doenjang during fermentation and aging.
MATERIALS/METHODS
Doenjang was made by inoculating Aspergillus oryzae and Bacillus licheniformis in soybeans, fermenting and aging for 1, 3, 6, 8, and 12 months (D1, D3, D6, D8, and D12). Doenjang was extracted using ethanol, and sequentially fractioned by hexane, dichloromethane (DM), ethylacetate (EA), n-butanol, and water. The contents of total phenolics, flavonoids and isoflavones, 2,2-diphenyl-1 picryl hydrazyl (DPPH) radical scavenging activity, and ferric reducing antioxidant power (FRAP) were measured. Anti-inflammatory effects in terms of nitric oxide (NO), prostaglandin (PG) E2 and pro-inflammatory cytokine production and inducible nitric oxide synthase (iNOS) and cyclooxygenase (COX)-2 expressions were also measured using LPS-treated RAW 264.7 macrophages.
RESULTS
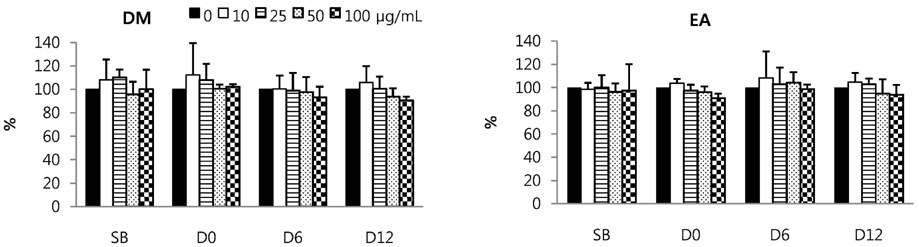
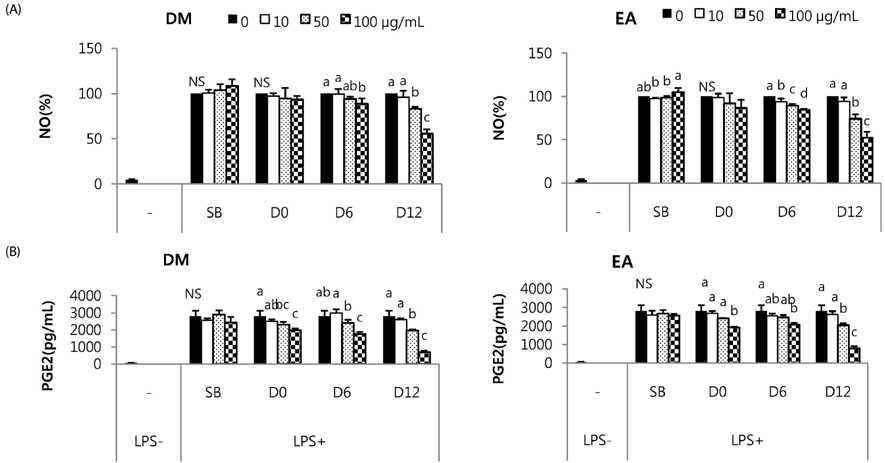
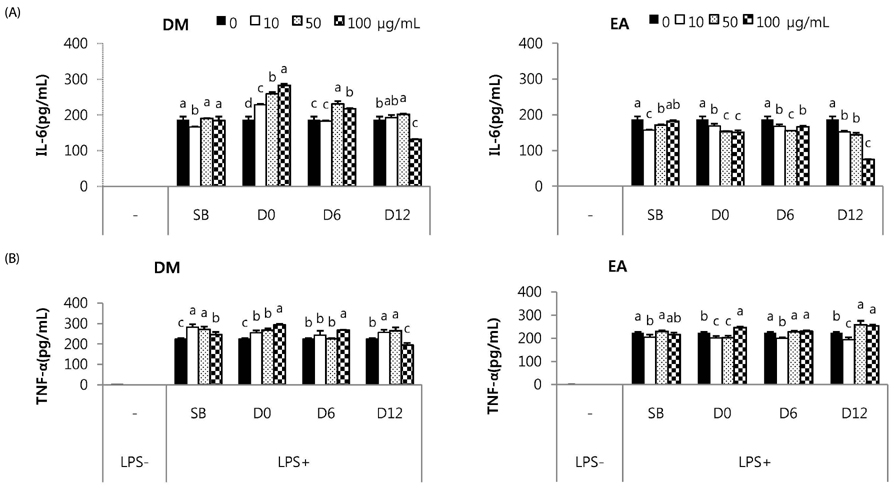
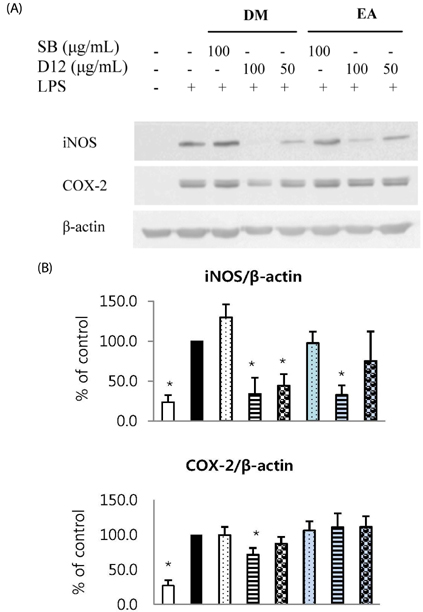
Total phenolic and flavonoid contents showed a gradual increase during fermentation and 6 months of aging and were sustained thereafter. DPPH radical scavenging activity and FRAP were increased by fermentation. FRAP was further increased by aging, but DPPH radical scavenging activity was not. Total isoflavone and glycoside contents decreased during fermentation and the aging process, while aglycone content and its proportion increased up to 3 or 6 months of aging and then showed a slow decrease. DM and EA fractions of Doenjang showed much higher total phenolic and flavonoid contents, and DPPH radical scavenging activity than the others. At 100 microg/mL, DM and EA fractions of D12 showed strongly suppressed NO production to 55.6% and 52.5% of control, respectively, and PGE2 production to 25.0% and 28.3% of control with inhibition of iNOS or COX-2 protein expression in macrophages.
CONCLUSIONS
Twelve month-aged Doenjang has potent antioxidant and anti-inflammatory activities with high levels of phenolics and isoflavone aglycones, and can be used as a beneficial food for human health.
MeSH Terms
-
1-Butanol
Aging
Aspergillus oryzae
Bacillus
Dinoprostone
Ethanol*
Fermentation
Flavonoids
Humans
Inflammation
Isoflavones
Macrophages*
Methylene Chloride
Nitric Oxide
Nitric Oxide Synthase Type II
Phenol
Prostaglandin-Endoperoxide Synthases
Soybeans
Water
1-Butanol
Dinoprostone
Ethanol
Flavonoids
Isoflavones
Methylene Chloride
Nitric Oxide
Nitric Oxide Synthase Type II
Phenol
Prostaglandin-Endoperoxide Synthases
Water
Figure
Reference
-
1. Lee TH, Do MH, Oh YL, Cho DW, Kim SH, Kim SY. Dietary fermented soybean suppresses UVB-induced skin inflammation in hairless mice via regulation of the MAPK signaling pathway. J Agric Food Chem. 2014; 62:8962–8972.
Article2. Kwon DY, Daily JW 3rd, Kim HJ, Park S. Antidiabetic effects of fermented soybean products on type 2 diabetes. Nutr Res. 2010; 30:1–13.
Article3. Chai C, Ju HK, Kim SC, Park JH, Lim J, Kwon SW, Lee J. Determination of bioactive compounds in fermented soybean products using GC/MS and further investigation of correlation of their bioactivities. J Chromatogr B Analyt Technol Biomed Life Sci. 2012; 880:42–49.
Article4. Chung SI, Rico CW, Kang MY. Comparative study on the hypoglycemic and antioxidative effects of fermented paste (doenjang) prepared from soybean and brown rice mixed with rice bran or red ginseng marc in mice fed with high fat diet. Nutrients. 2014; 6:4610–4624.
Article5. Hu CC, Hsiao CH, Huang SY, Fu SH, Lai CC, Hong TM, Chen HH, Lu FJ. Antioxidant activity of fermented soybean extract. J Agric Food Chem. 2004; 52:5735–5739.
Article6. Chen YC, Sugiyama Y, Abe N, Kuruto-Niwa R, Nozawa R, Hirota A. DPPH radical-scavenging compounds from Dou-chi, a soybean fermented food. Biosci Biotechnol Biochem. 2005; 69:999–1006.
Article7. Kwak CS, Lee MS, Park SC. Higher antioxidant properties of Chungkookjang, a fermented soybean paste, may be due to increased aglycone and malonylglycoside isoflavone during fermentation. Nutr Res. 2007; 27:719–727.
Article8. Jung KO, Park SY, Park KY. Longer aging time increases the anticancer and antimetastatic properties of doenjang. Nutrition. 2006; 22:539–545.
Article9. Park KY, Jung KO, Rhee SH, Choi YH. Antimutagenic effects of doenjang (Korean fermented soypaste) and its active compounds. Mutat Res. 2003; 523-524:43–53.
Article10. Park KJ, Kim YM, Lee BH, Lee BK. Fungal microflora on Korean home-made Meju. Korean J Mycol. 1977; 5:7–12.11. Lee SS. Meju fermentation for a raw material of Korean traditional soy products. Korean J Mycol. 1995; 23:161–175.12. Kwak CS, Park SC, Song KY. Doenjang, a fermented soybean paste, decreased visceral fat accumulation and adipocyte size in rats fed with high fat diet more effectively than nonfermented soybeans. J Med Food. 2012; 15:1–9.
Article13. Park NY, Rico CW, Lee SC, Kang MY. Comparative effects of doenjang prepared from soybean and brown rice on the body weight and lipid metabolism in high fat-fed mice. J Clin Biochem Nutr. 2012; 51:235–240.
Article14. Cha YS, Yang JA, Back HI, Kim SR, Kim MG, Jung SJ, Song WO, Chae SW. Visceral fat and body weight are reduced in overweight adults by the supplementation of Doenjang, a fermented soybean paste. Nutr Res Pract. 2012; 6:520–526.
Article15. Blay M, Espinel AE, Delgado MA, Baiges I, Bladé C, Arola L, Salvadó J. Isoflavone effect on gene expression profile and biomarkers of inflammation. J Pharm Biomed Anal. 2010; 51:382–390.
Article16. Scott A, Khan KM, Cook JL, Duronio V. What is "inflammation"? Are we ready to move beyond Celsus? Br J Sports Med. 2004; 38:248–249.
Article17. Prasad S, Phromnoi K, Yadav VR, Chaturvedi MM, Aggarwal BB. Targeting inflammatory pathways by flavonoids for prevention and treatment of cancer. Planta Med. 2010; 76:1044–1063.
Article18. Witkamp R, Monshouwer M. Signal transduction in inflammatory processes, current and future therapeutic targets: a mini review. Vet Q. 2000; 22:11–16.
Article19. Bak MJ, Jeong JH, Kang HS, Jin KS, Ok S, Jeong WS. Cedrela sinensis leaves suppress oxidative stress and expressions of iNOS and COX-2 via MAPK signaling pathways in RAW 264.7 cells. J Food Sci Nutr. 2009; 14:269–276.
Article20. Kwon SH, Lee KB, Im KS, Kim SO, Park KY. Weight reduction and lipid lowering effects of Korean traditional soybean fermented products. J Korean Soc Food Sci Nutr. 2006; 35:1194–1199.
Article21. Jang CH, Lim JK, Kim JH, Park CS, Kwon DY, Kim YS, Shin DH, Kim JS. Change of isoflavone content during manufacturing of Cheonggukjang, a traditional Korean fermented soyfood. Food Sci Biotechnol. 2006; 15:643–646.22. Nakajima N, Nozaki N, Ishihara K, Ishikawa A, Tsuji H. Analysis of isoflavone content in tempeh, a fermented soybean, and preparation of a new isoflavone-enriched tempeh. J Biosci Bioeng. 2005; 100:685–687.
Article23. Shon MY, Seo KI, Lee SW, Choi SH, Sung NJ. Biological activities of Chungkugjang prepared with black bean and changes in phytoestrogen content during fermentation. Korean J Food Sci Technol. 2000; 32:936–941.24. Danciu C, Soica C, Csanyi E, Ambrus R, Feflea S, Peev C, Dehelean C. Changes in the anti-inflammatory activity of soy isoflavonoid genistein versus genistein incorporated in two types of cyclodextrin derivatives. Chem Cent J. 2012; 6:58.
Article25. Liang YC, Huang YT, Tsai SH, Lin-Shiau SY, Chen CF, Lin JK. Suppression of inducible cyclooxygenase and inducible nitric oxide synthase by apigenin and related flavonoids in mouse macrophages. Carcinogenesis. 1999; 20:1945–1952.
Article26. Ji G, Zhang Y, Yang Q, Cheng S, Hao J, Zhao X, Jiang Z. Genistein suppresses LPS-induced inflammatory response through inhibiting NF-κB following AMP kinase activation in RAW 264.7 macrophages. PLoS One. 2012; 7:e53101.
Article27. Singleton VL, Orthofer R, Lamuela-Raventós RM. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999; 299:152–178.28. Chae SK, Kang GS, Ma SJ, Bang KW, Oh MW, Oh SH. Standard Food Analysis. Seoul: Jigu Publishing Co.;1997.29. Senba Y, Nishishita T, Saito K, Yoshioka H, Yoshioka H. Stopped-flow and spectrophotometric study on radical scavenging by tea catechins and the model compounds. Chem Pharm Bull (Tokyo). 1999; 47:1369–1374.
Article30. Yildirim A, Mavi A, Kara AA. Determination of antioxidant and antimicrobial activities of Rumex crispus L. extracts. J Agric Food Chem. 2001; 49:4083–4089.
Article31. Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983; 65:55–63.
Article32. Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem. 1982; 126:131–138.
Article33. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976; 72:248–254.
Article34. Zhang R, Brennan ML, Shen Z, MacPherson JC, Schmitt D, Molenda CE, Hazen SL. Myeloperoxidase functions as a major enzymatic catalyst for initiation of lipid peroxidation at sites of inflammation. J Biol Chem. 2002; 277:46116–46122.
Article35. Coulibaly AY, Kiendrebeogo M, Kehoe PG, Sombie PA, Lamien CE, Millogo JF, Nacoulma OG. Antioxidant and anti-inflammatory effects of Scoparia dulcis L. J Med Food. 2011; 14:1576–1582.
Article36. Chang ST, Wu JH, Wang SY, Kang PL, Yang NS, Shyur LF. Antioxidant activity of extracts from Acacia confusa bark and heartwood. J Agric Food Chem. 2001; 49:3420–3424.
Article37. Michel P, Dobrowolska A, Kicel A, Owczarek A, Bazylko A, Granica S, Piwowarski JP, Olszewska MA. Polyphenolic profile, antioxidant and anti-inflammatory activity of eastern teaberry (Gaultheria procumbens L.) leaf extracts. Molecules. 2014; 19:20498–20520.
Article38. Noh KH, Jang JH, Min KH, Chinzorig R, Lee MO, Song YS. Suppressive effect of green tea seed coat ethyl acetate fraction on inflammation and its mechanism in RAW264.7 macrophage cell. J Korean Soc Food Sci Nutr. 2011; 40:625–634.
Article39. Kim YS, Lee SJ, Hwang JW, Kim EH, Park PJ, Jeong JH. Anti-inflammatory effects of extracts from Ligustrum ovalifolium H. leaves on RAW264.7 macrophages. J Korean Soc Food Sci Nutr. 2012; 41:1205–1210.
Article40. Velioglu YS, Mazza G, Gao L, Oomah BD. Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J Agric Food Chem. 1998; 46:4113–4117.
Article41. Zainol MK, Abd-Hamid A, Yusof S, Muse R. Antioxidative activity and total phenolic compounds of leaf, root and petiole of four accessions of Centella asiatica (L.) urban. Food Chem. 2003; 81:575–581.
Article42. Kwak CS, Kim SA, Lee MS. The correlation of antioxidative effects of 5 Korean common edible seaweeds and total polyphenol content. J Korean Soc Food Sci Nutr. 2005; 34:1143–1150.
Article43. Setchell KD, Brown NM, Zimmer-Nechemias L, Brashear WT, Wolfe BE, Kirschner AS, Heubi JE. Evidence for lack of absorption of soy isoflavone glycosides in humans, supporting the crucial role of intestinal metabolism for bioavailability. Am J Clin Nutr. 2002; 76:447–453.
Article44. Izumi T, Piskula MK, Osawa S, Obata A, Tobe K, Saito M, Kataoka S, Kubota Y, Kikuchi M. Soy isoflavone aglycones are absorbed faster and in higher amounts than their glucosides in humans. J Nutr. 2000; 130:1695–1699.
Article45. Pratt DE. Natural antioxidants of soybeans and other oil-seeds. In : Simic MG, Karel M, editors. Autoxidation in Food and Biological Systems. New York (NY): Plenum Press;1980. p. 283–293.46. Sheih IC, Fang TJ, Wu TK, Chen RY. Effects of fermentation on antioxidant properties and phytochemical composition of soy germ. J Sci Food Agric. 2014; 94:3163–3170.
Article47. Dziedzic SZ, Hudson BJ. Hydroxy isoflavones as antioxidants for edible oils. Food Chem. 1983; 11:161–166.
Article48. Kwon SH, Shon MY. Antioxidant and anticarcinogenic effects of traditional Doenjang during maturation periods. Korean J Food Preserv. 2004; 11:461–467.49. Kim MH, Im SS, Kim SH, Kim GE, Lee JH. Antioxidative materials in domestic Meju and Doenjang 2. Seperation of lipophilic brown pigment and their antioxidative activity. J Korean Soc Food Sci Nutr. 1994; 23:251–260.50. Jeong JK, Zheng Y, Choi HS, Han GJ, Park KY. Catabolic enzyme activities and physiological functionalities of lactic acid bacteria isolated from Korean traditional Meju. J Korean Soc Food Sci Nutr. 2010; 39:1854–1859.
Article51. Roh C, Jung U, Jo SK. 6,7,4'-Trihydroxyisoflavone from Doenjang inhibits lipid accumulation. Food Chem. 2011; 129:183–187.
Article52. Park JS, Park HY, Kim DH, Kim DH, Kim HK. Ortho-dihydroxyisoflavone derivatives from aged Doenjang (Korean fermented soypaste) and its radical scavenging activity. Bioorg Med Chem Lett. 2008; 18:5006–5009.
Article53. Wang HJ, Murphy PA. Mass balance study of isoflavones during soybean processing. J Agric Food Chem. 1996; 44:2377–2383.
Article54. Wu CH, Chou CC. Enhancement of aglycone, vitamin K2 and superoxide dismutase activity of black soybean through fermentation with Bacillus subtilis BCRC 14715 at different temperatures. J Agric Food Chem. 2009; 57:10695–10700.
Article55. Lee S, Seo MH, Oh DK, Lee CH. Targeted metabolomics for Aspergillus oryzae-mediated biotransformation of soybean isoflavones, showing variations in primary metabolites. Biosci Biotechnol Biochem. 2014; 78:167–174.
Article56. Jeong JW, Lee HH, Han MH, Kim GY, Kim WJ, Choi YH. Anti-inflammatory effects of genistein via suppression of the toll-like receptor 4-mediated signaling pathway in lipopolysaccharide-stimulated BV2 microglia. Chem Biol Interact. 2014; 212:30–39.
Article57. Sung YH, Chang HK, Kim SE, Kim YM, Seo JH, Shin MC, Shin MS, Yi JW, Shin DH, Kim H, Kim CJ. Anti-inflammatory and analgesic effects of the aqueous extract of Corni fructus in murine RAW 264.7 macrophage cells. J Med Food. 2009; 12:788–795.
Article58. Bredt DS, Snyder SH. Nitric oxide: a physiologic messenger molecule. Annu Rev Biochem. 1994; 63:175–195.
Article59. Lee AK, Sung SH, Kim YC, Kim SG. Inhibition of lipopolysaccharide-inducible nitric oxide synthase, TNF-α and COX-2 expression by sauchinone effects on IκBα phosphorylation, C/EBP and AP-1 activation. Br J Pharmacol. 2003; 139(1):11–20.
Article60. Lee EJ, Kim C, Kim JY, Kim SM, Nam D, Jang HJ, Kim SH, Shim BS, Ahn KS, Choi SH, Jung SH, Ahn KS. Inhibition of LPS-induced inflammatory biomarkers by ethyl acetate fraction of Patrinia scabiosaefolia through suppression of NF-κB activation in RAW 264.7 cells. Immunopharmacol Immunotoxicol. 2012; 34:282–291.
Article61. Koeberle A, Northoff H, Werz O. Curcumin blocks prostaglandin E2 biosynthesis through direct inhibition of the microsomal prostaglandin E2 synthase-1. Mol Cancer Ther. 2009; 8:2348–2355.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inhibitory Activity of Cordyceps bassiana Extract on LPS-induced Inflammation in RAW 264.7 Cells by Suppressing NF-κB Activation
- Antioxidant and Anti-inflammatory Activities of Rumex acetosa
- Inhibitory Effect of Ginsenosides Rh1 and Rg2 on Oxidative Stress in LPS-Stimulated RAW 264.7 Cells
- Anti-inflammatory Effect of Mangosteen (Garcinia mangostana L.) Peel Extract and its Compounds in LPS-induced RAW264.7 Cells
- YJI-7 Suppresses ROS Production and Expression of Inflammatory Mediators via Modulation of p38MAPK and JNK Signaling in RAW 264.7 Macrophages