Nutr Res Pract.
2010 Dec;4(6):462-469.
Effect of dietary protamine on lipid metabolism in rats
- Affiliations
-
- 1Department of Life Science and Biotechnology, Faculty of Chemistry, Materials and Bioengineering, Kansai University, Suita, Osaka, 564-8680, Japan. fukunagk@kansai-u.ac.jp
- 2Department of Biotechnology and Environmental Chemistry, Kitami Institute of Technology, Kitami, Hokkaido 090-8507, Japan.
- 3Department of Public Health, Kansai Medical University, Moriguchi, Osaka, 570-8506, Japan.
Abstract
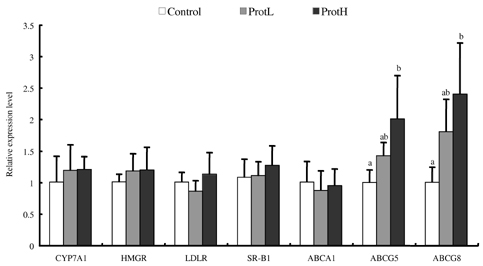
- Protamine has been widely used as a pharmaceutical product and natural food preservative. However, few studies have been conducted to assess the beneficial function of dietary protamine. This study examined the effects of dietary salmon protamine on serum and liver lipid levels and the expression levels of genes encoding proteins involved in lipid homeostasis in the liver of rats. Groups of male Wistar rats were fed AIN93G diet containing 2% or 5% protamine. After 4 weeks of feeding these diets, markedly decreased serum and liver cholesterol (CHOL) and triacylglycerol levels were noted. Increased activity of liver carnitine palmitoyltransferase-2 and acyl-CoA oxidase, which are key enzymes of fatty acid beta-oxidation in the mitochondria and peroxisomes, was found in rats fed on protamine. Furthermore, rats fed protamine showed enhanced fecal excretion of CHOL and bile acid and increased liver mRNA expression levels of ATP-binding cassette (ABC) G5 and ABCG8, which form heterodimers and play a major role in the secretion of CHOL into bile. The decrease in triacylglycerol levels in protamine-fed rats was due to the enhancement of liver beta-oxidation. Furthermore, rats fed protamine exhibited decreased CHOL levels through the suppression of CHOL and bile acid absorption and the enhancement of CHOL secretion into bile. These results suggest that dietary protamine has beneficial effects that may aid in the prevention of lifestyle-related diseases such as hyperlipidemia and atherosclerosis.
MeSH Terms
Figure
Reference
-
1. Formiguera X, Cantón A. Obesity: epidemiology and clinical aspects. Best Pract Res Clin Gastroenterol. 2004. 18:1125–1146.
Article2. Dyerberg J, Bang HO, Hjorne N. Fatty acid composition of the plasma lipids in Greenland Eskimos. Am J Clin Nutr. 1975. 28:958–966.
Article3. Iso H, Kobayashi M, Ishihara J, Sasaki S, Okada K, Kita Y, Kokubo Y, Tsugane S. JPHC Study Group. Intake of fish and n3 fatty acids and risk of coronary heart disease among Japanese: the Japan Public Health Center-Based (JPHC) Study Cohort I. Circulation. 2006. 113:195–202.
Article4. He K, Song Y, Daviglus ML, Liu K, Van Horn L, Dyer AR, Greenland P. Accumulated evidence on fish consumption and coronary heart disease mortality: a meta-analysis of cohort studies. Circulation. 2004. 109:2705–2711.
Article5. Simopoulos AP. Omega-3 fatty acids in inflammation and autoimmune diseases. J Am Coll Nutr. 2002. 21:495–505.
Article6. Simopoulos AP. Omega-3 fatty acids in health and disease and in growth and development. Am J Clin Nutr. 1991. 54:438–463.
Article7. Dahl L, Bjørkkjaer T, Graff IE, Malde MK, Klementsen B. Fish--more than just omega 3. Tidsskr Nor Laegeforen. 2006. 126:309–311.8. Zhang X, Beynen AC. Influence of dietary fish proteins on plasma and liver cholesterol concentrations in rats. Br J Nutr. 1993. 69:767–777.
Article9. Murata M, Sano Y, Bannai S, Ishihara K, Matsushima R, Uchida M. Fish protein stimulated the fibrinolysis in rats. Ann Nutr Metab. 2004. 48:348–356.
Article10. Hosomi R, Fukunaga K, Arai H, Nishiyama T, Yoshida M. Effects of dietary fish protein on serum and liver lipid concentrations in rats and the expression of hepatic genes involved in lipid metabolism. J Agric Food Chem. 2009. 57:9256–9262.
Article11. Jaques LB. Protamine--antagonist to heparin. Can Med Assoc J. 1973. 108:1291–1293. 1295–1297.12. Aspedon A, Groisman EA. The antibacterial action of protamine: evidence for disruption of cytoplasmic membrane energization in Salmonella typhimurium. Microbiology. 1996. 142:3389–3397.
Article13. Tsujita T, Matsuura Y, Okuda H. Studies on the inhibition of pancreatic and carboxylester lipases by protamine. J Lipid Res. 1996. 37:1481–1487.
Article14. Duarte-Vázquez MA, García-Padilla S, Olvera-Ochoa L, González-Romero KE, Acosta-Iñiguez J, De la Cruz-Cordero R, Rosado JL. Effect of protamine in obesity induced by high-fat diets in rats. Int J Obes (Lond). 2009. 33:687–692.
Article15. Reeves PG, Nielsen FH, Fahey GC Jr. AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr. 1993. 123:1939–1951.
Article16. Horton JD, Bashmakov Y, Shimomura I, Shimano H. Regulation of sterol regulatory element binding proteins in livers of fasted and refed mice. Proc Natl Acad Sci U S A. 1998. 95:5987–5992.
Article17. Hatch FT. Practical methods for plasma lipoprotein analysis. Adv Lipid Res. 1968. 6:1–68.18. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951. 193:265–275.
Article19. Ide T, Watanabe M, Sugano M, Yamamoto I. Activities of liver mitochondrial and peroxisomal fatty acid oxidation enzymes in rats fed trans fat. Lipids. 1987. 22:6–10.
Article20. Markwell MA, McGroarty EJ, Bieber LL, Tolbert NE. The subcellular distribution of carnitine acyltransferases in mammalian liver and kidney. A new peroxisomal enzyme. J Biol Chem. 1973. 248:3426–3432.
Article21. Kelley DS, Nelson GJ, Hunt JE. Effect of prior nutritional status on the activity of lipogenic enzymes in primary monolayer cultures of rat hepatocytes. Biochem J. 1986. 235:87–90.
Article22. Tanabe T, Nakanishi S, Hashimoto T, Ogiwara H, Nikawa J, Numa S. Acetyl-CoA carboxylase from rat liver : EC 6.4.1.2 Acetyl-CoA: carbon-dioxide ligase (ADP-forming). Lipids Part C. Methods Enzymol. 1981. New York: Academic Press;5–16.23. Kelley DS, Kletzien RF. Ethanol modulation of the hormonal and nutritional regulation of glucose 6-phosphate dehydrogenase activity in primary cultures of rat hepatocytes. Biochem J. 1984. 217:543–549.
Article24. Ichihara K, Norikura S, Fujii S. Microsomal phosphatidate phosphatase in maturing safflower seeds. Plant Physiol. 1989. 90:413–419.
Article25. van de Kamer JH, ten Bokkel Huinink H, Weyers HA. Rapid method for the determination of fat in feces. J Biol Chem. 1949. 177:347–355.
Article26. Moriyama T, Kishimoto K, Ngai K, Urade R, Ogawa T, Uthumi S, Maruyama N, Maebuchi M. Soybean beta-conglycinin diet suppresses serum triglyceride levels in normal and genetically obese mice by induction of beta-oxidation, downregulation of fatty acid synthase, and inhibition of triglyceride absorption. Biosci Biotechnol Biochem. 2004. 68:352–359.
Article27. Higaki N, Sato K, Suda H, Suzuka T, Komori T, Saeki T, Nakamura Y, Ohtsuki K, Iwami K, Kanamoto R. Evidence for the existence of a soybean resistant protein that captures bile acid and stimulates its fecal excretion. Biosci Biotechnol Biochem. 2006. 70:2844–2852.
Article28. Nagata Y, Tanaka K, Sugano M. Serum and liver cholesterol levels of rats and mice fed soy-bean protein or casein. J Nutr Sci Vitaminol (Tokyo). 1981. 27:583–593.
Article29. Tsujita T, Sumiyoshi M, Takaku T, Momsen WE, Lowe ME, Brockman HL. Inhibition of lipases by epsilon-polylysine. J Lipid Res. 2003. 44:2278–2286.30. Fielding BA, Frayn KN. Lipoprotein lipase and the disposition of dietary fatty acids. Br J Nutr. 1998. 80:495–502.
Article31. Iwami K, Kitagawa M, Ibuki F. Effect of dietary proteins and/or their digestive products on intestinal taurocholate absorption. J Nutr Sci Vitaminol (Tokyo). 1990. 36:S141–S146.
Article32. Kayashita J, Shimaoka I, Nakajoh M, Yamazaki M, Kato N. Consumption of buckwheat protein lowers plasma cholesterol and raises fecal neutral sterols in cholesterol-Fed rats because of its low digestibility. J Nutr. 1997. 127:1395–1400.
Article33. Tamehiro N, Shigemoto-Mogami Y, Kakeya T, Okuhira K, Suzuki K, Sato R, Nagao T, Nishimaki-Mogami T. Sterol regulatory element-binding protein-2- and liver X receptor-driven dual promoter regulation of hepatic ABC transporter A1 gene expression: mechanism underlying the unique response to cellular cholesterol status. J Biol Chem. 2007. 282:21090–21099.
Article34. García-Villafranca J, Guillén A, Castro J. Involvement of nitric oxide/cyclic GMP signaling pathway in the regulation of fatty acid metabolism in rat hepatocytes. Biochem Pharmacol. 2003. 65:807–812.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Modulation of lipid metabolism by mixtures of protamine and chitooligosaccharide through pancreatic lipase inhibitory activity in a rat model
- Effect of Dietary Iron Levels on Lipid Metabolism, Antioxidative and Antithrombogenic Capacities in 16-month-old Rats
- Effects of Legume Supplementation on the Glucose and Lipid Metabolism and Lipid Peroxidation in Streptozotocin-Induced Diabetic Rats
- Effect of Dietary Grape Pomace on Lipid Oxidation and Related Enzyme Activities in Rats Fed High Fat Diet
- Effect of Fish Oil Diet on Activities of Lipogenic Enzymes and Glucose -6 -phosphatase in Rat Liver and Adipose