Korean J Physiol Pharmacol.
2014 Aug;18(4):307-311. 10.4196/kjpp.2014.18.4.307.
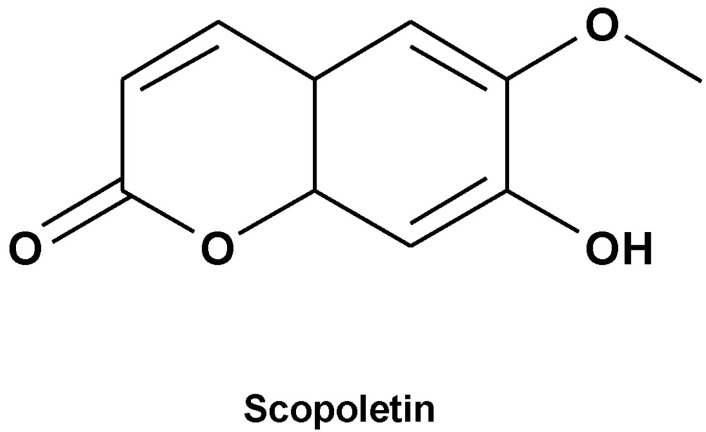
Scopoletin from Cirsium setidens Increases Melanin Synthesis via CREB Phosphorylation in B16F10 Cells
- Affiliations
-
- 1Department of Pharmacognosy, Chung-Ang University College of Pharmacy, Seoul 156-756, Korea.
- 2Department of Biochemistry, Chung-Ang University College of Medicine, Seoul 156-756, Korea. ds_kim@cau.ac.kr
- 3Thomas J. Long School of Pharmacy, University of the Pacific, CA 95211, USA.
- KMID: 2285524
- DOI: http://doi.org/10.4196/kjpp.2014.18.4.307
Abstract
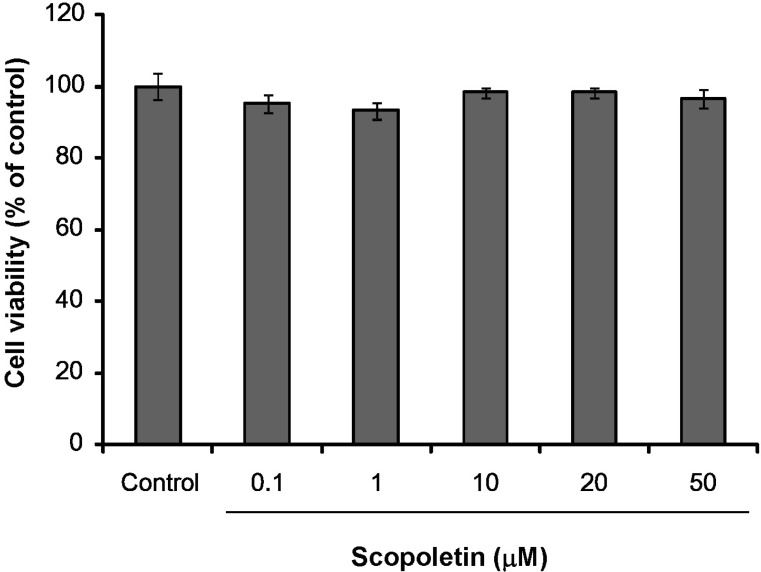
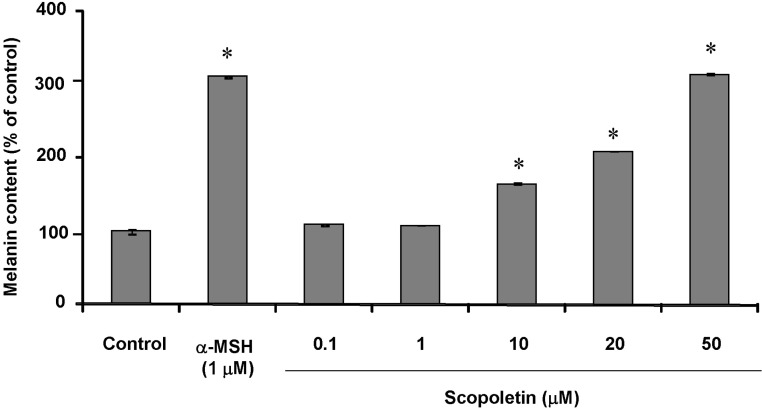
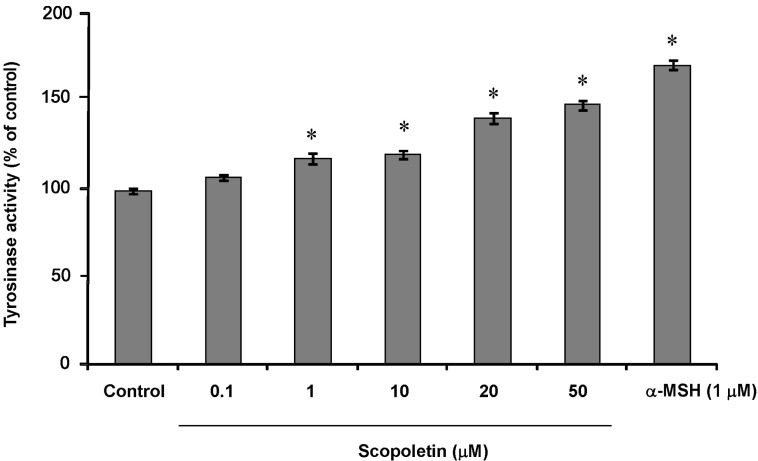
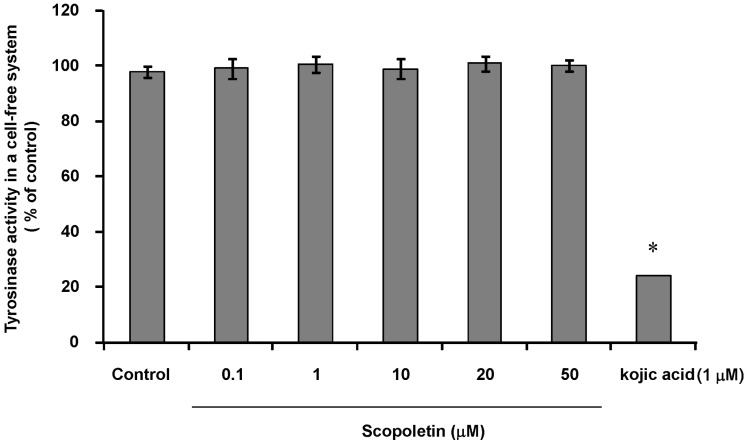
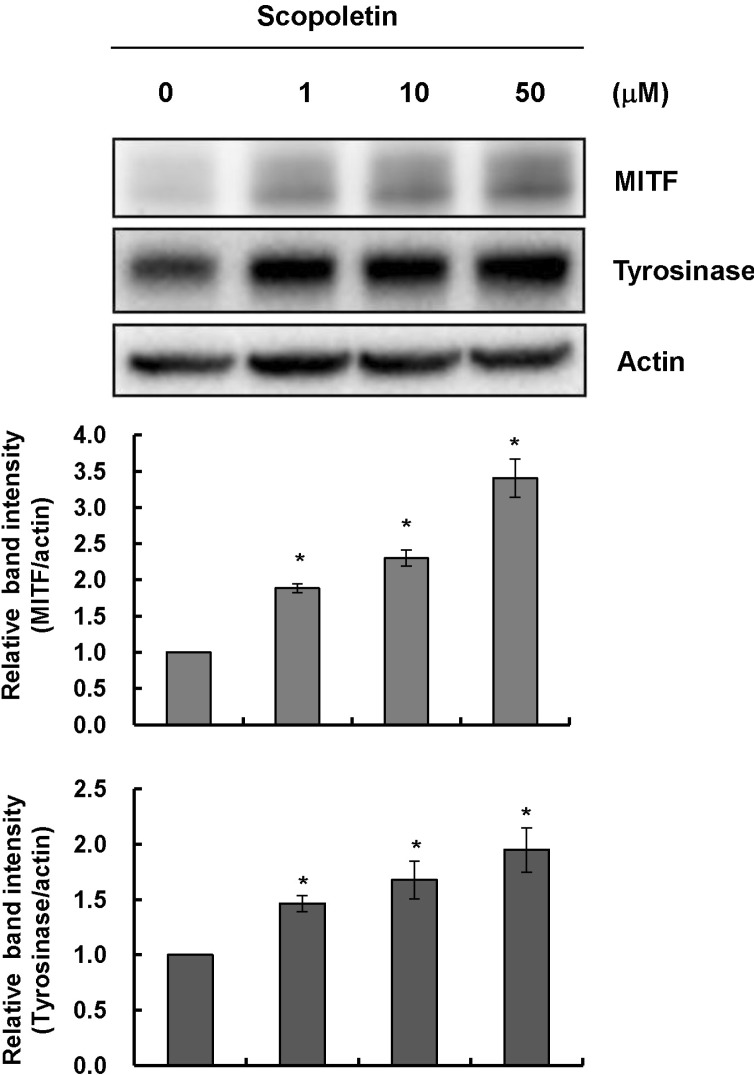
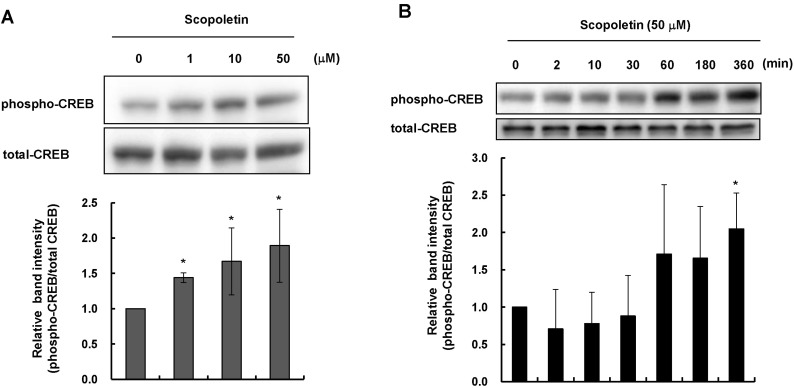
- In this study, we isolated scopoletin from Cirsium setidens Nakai (Compositae) and tested its effects on melanogenesis. Scopoletin was not toxic to cells at concentrations less than 50 microM and increased melanin synthesis in a dose-dependent manner. As melanin synthesis increased, scopoletin stimulated the total tyrosinase activity, the rate-limiting enzyme of melanogenesis. In a cell-free system, however, scopoletin did not increase tyrosinase activity, indicating that scopoletin is not a direct activator of tyrosinase. Furthermore, Western blot analysis showed that scopoletin stimulated the production of microphthalmia-associated transcription factor (MITF) and tyrosinase expression via cAMP response element-binding protein (CREB) phosphorylation in a dose-dependent manner. Based on these results, preclinical and clinical studies are needed to assess the use of scopoletin for the treatment of vitiligo.
Keyword
MeSH Terms
-
Blotting, Western
Cell-Free System
Cirsium*
Cyclic AMP Response Element-Binding Protein
Melanins*
Microphthalmia-Associated Transcription Factor
Monophenol Monooxygenase
Phosphorylation*
Scopoletin*
Vitiligo
Cyclic AMP Response Element-Binding Protein
Melanins
Microphthalmia-Associated Transcription Factor
Monophenol Monooxygenase
Scopoletin
Figure
Reference
-
1. Hearing VJ, Tsukamoto K. Enzymatic control of pigmentation in mammals. FASEB J. 1991; 5:2902–2909. PMID: 1752358.
Article2. Cho YS, Lee KH, Park JW. Pyrithione-zinc Prevents UVB-induced Epidermal Hyperplasia by Inducing HIF-1alpha. Korean J Physiol Pharmacol. 2010; 14:91–97. PMID: 20473380.3. Pichler R, Sfetsos K, Badics B, Gutenbrunner S, Auböck J. Vitiligo patients present lower plasma levels of alpha-melanotropin immunoreactivities. Neuropeptides. 2006; 40:177–183. PMID: 16677711.4. Lee WB, Kwon HC, Cho OR, Lee KC, Choi SU, Baek NI, Lee KR. Phytochemical constituents of Cirsium setidens Nakai and their cytotoxicity against human cancer cell lines. Arch Pharm Res. 2002; 25:628–635. PMID: 12433195.5. Tal B, Robeson DJ. The metabolism of sunflower phytoalexins ayapin and scopoletin: plant-fungus interactions. Plant Physiol. 1986; 82:167–172. PMID: 16664986.6. Gnonlonfin BG, Gbaguidi F, Gbenou JD, Sanni A, Brimer L. Changes in scopoletin concentration in cassava chips from four varieties during storage. J Sci Food Agric. 2011; 91:2344–2347. PMID: 21604276.
Article7. Rollinger JM, Hornick A, Langer T, Stuppner H, Prast H. Acetylcholinesterase inhibitory activity of scopolin and scopoletin discovered by virtual screening of natural products. J Med Chem. 2004; 47:6248–6254. PMID: 15566295.
Article8. Lee SH, Ding Y, Yan XT, Kim YH, Jang HD. Scopoletin and scopolin isolated from Artemisia iwayomogi suppress differentiation of osteoclastic macrophage RAW 264.7 cells by scavenging reactive oxygen species. J Nat Prod. 2013; 76:615–620. PMID: 23510022.
Article9. Yao X, Ding Z, Xia Y, Wei Z, Luo Y, Feleder C, Dai Y. Inhibition of monosodium urate crystal-induced inflammation by scopoletin and underlying mechanisms. Int Immunopharmacol. 2012; 14:454–462. PMID: 22914669.
Article10. Zhang WY, Lee JJ, Kim Y, Kim IS, Park JS, Myung CS. Amelioration of insulin resistance by scopoletin in high-glucose-induced, insulin-resistant HepG2 cells. Horm Metab Res. 2010; 42:930–935. PMID: 20886413.
Article11. Costin GE, Hearing VJ. Human skin pigmentation: melanocytes modulate skin color in response to stress. FASEB J. 2007; 21:976–994. PMID: 17242160.
Article12. Yasumoto K, Yokoyama K, Takahashi K, Tomita Y, Shibahara S. Functional analysis of microphthalmia-associated transcription factor in pigment cell-specific transcription of the human tyrosinase family genes. J Biol Chem. 1997; 272:503–509. PMID: 8995290.
Article13. Lee HE, Kim EH, Choi HR, Sohn UD, Yun HY, Baek KJ, Kwon NS, Park KC, Kim DS. Dipeptides Inhibit Melanin Synthesis in Mel-Ab Cells through Down-Regulation of Tyrosinase. Korean J Physiol Pharmacol. 2012; 16:287–291. PMID: 22915995.
Article14. Buscà R, Ballotti R. Cyclic AMP a key messenger in the regulation of skin pigmentation. Pigment Cell Res. 2000; 13:60–69. PMID: 10841026.15. Wu Q, Zou L, Yang XW, Fu DX. Novel sesquiterpene and coumarin constituents from the whole herbs of Crossostephium chinense. J Asian Nat Prod Res. 2009; 11:85–90. PMID: 19177244.
Article16. Smalley K, Eisen T. The involvement of p38 mitogen-activated protein kinase in the alpha-melanocyte stimulating hormone (alpha-MSH)-induced melanogenic and anti-proliferative effects in B16 murine melanoma cells. FEBS Lett. 2000; 476:198–202. PMID: 10913613.17. Noh JM, Kwak SY, Kim DH, Lee YS. Kojic acid-tripeptide amide as a new tyrosinase inhibitor. Biopolymers. 2007; 88:300–307. PMID: 17211869.
Article18. Kim DS, Park SH, Kwon SB, Park ES, Huh CH, Youn SW, Park KC. Sphingosylphosphorylcholine-induced ERK activation inhibits melanin synthesis in human melanocytes. Pigment Cell Res. 2006; 19:146–153. PMID: 16524430.
Article19. Price ER, Horstmann MA, Wells AG, Weilbaecher KN, Takemoto CM, Landis MW, Fisher DE. alpha-Melanocyte-stimulating hormone signaling regulates expression of microphthalmia, a gene deficient in Waardenburg syndrome. J Biol Chem. 1998; 273:33042–33047. PMID: 9830058.20. Dou Y, Tong B, Wei Z, Li Y, Xia Y, Dai Y. Scopoletin suppresses IL-6 production from fibroblast-like synoviocytes of adjuvant arthritis rats induced by IL-1β stimulation. Int Immunopharmacol. 2013; 17:1037–1043. PMID: 24455774.
Article21. Grimes PE, Hamzavi I, Lebwohl M, Ortonne JP, Lim HW. The efficacy of afamelanotide and narrowband UV-B phototherapy for repigmentation of vitiligo. JAMA Dermatol. 2013; 149:68–73. PMID: 23407924.
Article22. Fabrikant J, Touloei K, Brown SM. A review and update on melanocyte stimulating hormone therapy: afamelanotide. J Drugs Dermatol. 2013; 12:775–779. PMID: 23884489.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diarylpropionitrile inhibits melanogenesis via protein kinase A/cAMP-response element-binding protein/microphthalmiaassociated transcription factor signaling pathway in α-MSHstimulated B16F10 melanoma cells
- Protective effects of Cirsium setidens ethanolic extracts against alcoholic fatty liver injury in rats
- The Evaluation of Skin Safety and Skin Cell Toxicity for Scutellaria baicalensis Georgi Extract according to Extraction Conditions
- Glyceollins, a novel class of soybean phytoalexins, inhibit SCF-induced melanogenesis through attenuation of SCF/c-kit downstream signaling pathways
- Anti-melanogenesis and Anti-inflammatory Effects of Cinnamomi Cortex Extract In Vitro