J Periodontal Implant Sci.
2010 Jun;40(3):119-124. 10.5051/jpis.2010.40.3.119.
Leptin potentiates Prevotella intermedia lipopolysaccharide-induced production of TNF-alpha in monocyte-derived macrophages
- Affiliations
-
- 1Department of Periodontology, Pusan National University College of Dentistry, Yangsan, Korea. sungjokim@pusan.ac.kr
- KMID: 2212138
- DOI: http://doi.org/10.5051/jpis.2010.40.3.119
Abstract
- PURPOSE
In addition to regulating body weight, leptin is also recognized for its role in the regulation of immune function and inflammation. The purpose of this study was to investigate the effect of leptin on Prevotella (P.) intermedia lipopolysaccharide (LPS)-induced tumor necrosis factor (TNF)-alpha production in differentiated THP-1 cells, a human monocytic cell line.
METHODS
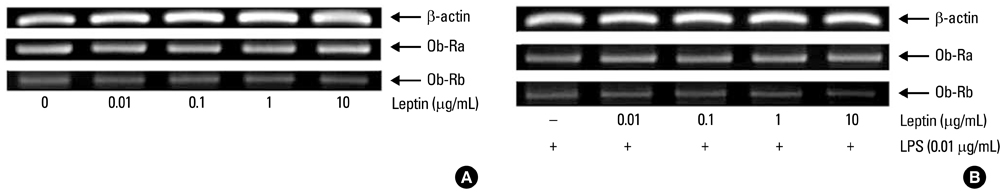
LPS from P. intermedia ATCC 25611 was prepared by the standard hot phenol-water method. THP-1 cells were incubated in the medium supplemented with phorbol myristate acetate to induce differentiation into macrophage-like cells. The amount of TNF-alpha and interleukin-8 secreted into the culture medium was determined by enzyme-linked immunosorbent assay (ELISA). TNF-alpha and Ob-R mRNA expression levels were determined by semi-quantitative reverse transcription-polymerase chain reaction analysis.
RESULTS
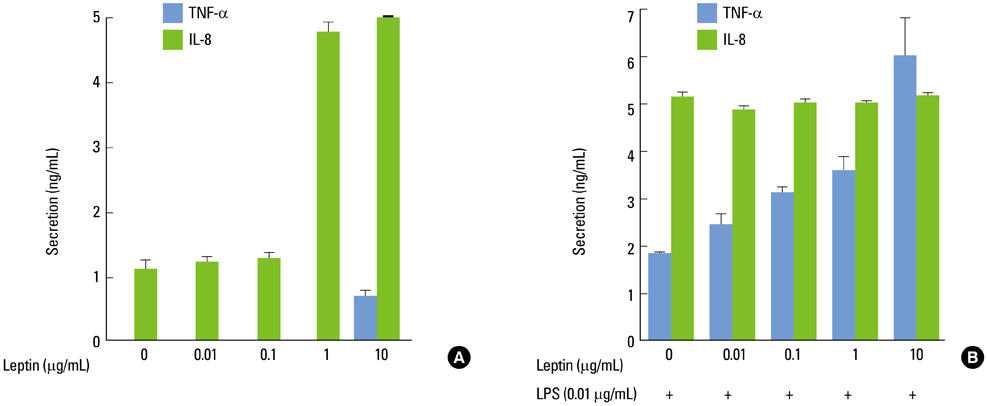
Leptin enhanced P. intermedia LPS-induced TNF-alpha production in a dose-dependent manner. Leptin modulated P. intermedia LPS-induced TNF-alpha expression predominantly at the transcriptional level. Effect of leptin on P. intermedia LPS-induced TNF-alpha production was not mediated by the leptin receptor.
CONCLUSIONS
The ability of leptin to enhance P. intermedia LPS-induced TNF-alpha production may be important in the establishment of chronic lesion accompanied by osseous tissue destruction observed in inflammatory periodontal disease.
MeSH Terms
-
Body Weight
Cell Line
Enzyme-Linked Immunosorbent Assay
Humans
Inflammation
Interleukin-8
Leptin
Macrophages
Periodontal Diseases
Phorbols
Prevotella
Prevotella intermedia
Receptors, Leptin
RNA, Messenger
Tetradecanoylphorbol Acetate
Tumor Necrosis Factor-alpha
Interleukin-8
Leptin
Phorbols
RNA, Messenger
Receptors, Leptin
Tetradecanoylphorbol Acetate
Tumor Necrosis Factor-alpha
Figure
Reference
-
1. Williams RC. Periodontal disease. N Engl J Med. 1990. 322:373–382.
Article2. Slots J, Bragd L, Wikstrom M, Dahlen G. The occurrence of Actinobacillus actinomycetemcomitans, Bacteroides gingivalis and Bacteroides intermedius in destructive periodontal disease in adults. J Clin Periodontol. 1986. 13:570–577.
Article3. Chung CP, Nisengard RJ, Slots J, Genco RJ. Bacterial IgG and IgM antibody titers in acute necrotizing ulcerative gingivitis. J Periodontol. 1983. 54:557–562.
Article4. Kornman KS, Loesche WJ. The subgingival microbial flora during pregnancy. J Periodontal Res. 1980. 15:111–122.
Article5. Morrison DC, Ryan JL. Endotoxins and disease mechanisms. Annu Rev Med. 1987. 38:417–432.
Article6. Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994. 372:425–432.
Article7. Maffei M, Fei H, Lee GH, Dani C, Leroy P, Zhang Y, et al. Increased expression in adipocytes of ob RNA in mice with lesions of the hypothalamus and with mutations at the db locus. Proc Natl Acad Sci U S A. 1995. 92:6957–6960.
Article8. Flier JS. The adipocyte: storage depot or node on the energy information superhighway? Cell. 1995. 80:15–18.
Article9. Bouloumie A, Drexler HC, Lafontan M, Busse R. Leptin, the product of Ob gene, promotes angiogenesis. Circ Res. 1998. 83:1059–1066.
Article10. Martin-Romero C, Santos-Alvarez J, Goberna R, Sanchez-Margalet V. Human leptin enhances activation and proliferation of human circulating T lymphocytes. Cell Immunol. 2000. 199:15–24.
Article11. Masuzaki H, Ogawa Y, Sagawa N, Hosoda K, Matsumoto T, Mise H, et al. Nonadipose tissue production of leptin: leptin as a novel placenta-derived hormone in humans. Nat Med. 1997. 3:1029–1033.
Article12. Lord GM, Matarese G, Howard JK, Baker RJ, Bloom SR, Lechler RI. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature. 1998. 394:897–901.
Article13. Janik JE, Curti BD, Considine RV, Rager HC, Powers GC, Alvord WG, et al. Interleukin 1 alpha increases serum leptin concentrations in humans. J Clin Endocrinol Metab. 1997. 82:3084–3086.14. Sanchez-Margalet V, Martin-Romero C. Human leptin signaling in human peripheral blood mononuclear cells: activation of the JAK-STAT pathway. Cell Immunol. 2001. 211:30–36.
Article15. Halaas JL, Friedman JM. Leptin and its receptor. J Endocrinol. 1997. 155:215–216.
Article16. Hill RA, Margetic S, Pegg GG, Gazzola C. Leptin: its pharmacokinetics and tissue distribution. Int J Obes Relat Metab Disord. 1998. 22:765–770.
Article17. Breidert M, Miehlke S, Glasow A, Orban Z, Stolte M, Ehninger G, et al. Leptin and its receptor in normal human gastric mucosa and in Helicobacter pylori-associated gastritis. Scand J Gastroenterol. 1999. 34:954–961.18. Groschl M, Rauh M, Wagner R, Neuhuber W, Metzler M, Tamguney G, et al. Identification of leptin in human saliva. J Clin Endocrinol Metab. 2001. 86:5234–5239.
Article19. Westphal O, Jann K. Whistler RL, editor. Bacterial lipopolysaccharides: extraction with phenol-water and further applications of the procedure. Methods in carbohydrate chemistry. General polysaccharides. 1965. Vol. 5. New York: Academic Press;83–91.20. Markwell MA, Haas SM, Bieber LL, Tolbert NE. A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem. 1978. 87:206–210.
Article21. Birkedal-Hansen H. Role of cytokines and inflammatory mediators in tissue destruction. J Periodontal Res. 1993. 28:500–510.
Article22. Stoufi ED, Taubman MA, Ebersole JL, Smith DJ, Stashenko PP. Phenotypic analyses of mononuclear cells recovered from healthy and diseased human periodontal tissues. J Clin Immunol. 1987. 7:235–245.
Article23. Arnalich F, Lopez J, Codoceo R, Jim nez M, Madero R, Montiel C. Relationship of plasma leptin to plasma cytokines and human survivalin sepsis and septic shock. J Infect Dis. 1999. 180:908–911.
Article24. Finck BN, Johnson RW. Tumor necrosis factor (TNF)-alpha induces leptin production through the p55 TNF receptor. Am J Physiol Regul Integr Comp Physiol. 2000. 278:R537–R543.25. Faggioni R, Fantuzzi G, Fuller J, Dinarello CA, Feingold KR, Grunfeld C. IL-1 beta mediates leptin induction during inflammation. Am J Physiol. 1998. 274:R204–R208.26. Ahima RS, Flier JS. Leptin. Annu Rev Physiol. 2000. 62:413–437.
Article27. Karthikeyan BV, Pradeep AR. Gingival crevicular fluid and serum leptin: their relationship to periodontal health and disease. J Clin Periodontol. 2007. 34:467–472.
Article28. Karthikeyan BV, Pradeep AR. Leptin levels in gingival crevicular fluid in periodontal health and disease. J Periodontal Res. 2007. 42:300–304.
Article29. Johnson RB, Serio FG. Leptin within healthy and diseased human gingiva. J Periodontol. 2001. 72:1254–1257.
Article30. Rossomando EF, Kennedy JE, Hadjimichael J. Tumour necrosis factor alpha in gingival crevicular fluid as a possible indicator of periodontal disease in humans. Arch Oral Biol. 1990. 35:431–434.
Article31. Stashenko P, Jandinski JJ, Fujiyoshi P, Rynar J, Socransky SS. Tissue levels of bone resorptive cytokines in periodontal disease. J Periodontol. 1991. 62:504–509.
Article32. Abu-Amer Y, Ross FP, Edwards J, Teitelbaum SL. Lipopolysaccharide-stimulated osteoclastogenesis is mediated by tumor necrosis factor via its P55 receptor. J Clin Invest. 1997. 100:1557–1565.
Article33. Kobayashi K, Takahashi N, Jimi E, Udagawa N, Takami M, Kotake S, et al. Tumor necrosis factor alpha stimulates osteoclast differentiation by a mechanism independent of the ODF/RANKL-RANK interaction. J Exp Med. 2000. 191:275–286.
Article34. Assuma R, Oates T, Cochran D, Amar S, Graves DT. IL-1 and TNF antagonists inhibit the inflammatory response and bone loss in experimental periodontitis. J Immunol. 1998. 160:403–409.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Chemical and Immunobiological Characterization of Lipopolysaccharides from Prevotella intermedia and Prevotella nigrescens
- Interleukin-8 production and interleukin-8 mRNA expression induced by lipopolysaccharides from Prevotella intermedia and Prevotella nigrescens in monocyte-derived macrophages
- Effect of quercetin on the production of nitric oxide in murine macrophages stimulated with lipopolysaccharide from Prevotella intermedia
- Curcumin suppresses the production of interleukin-6 in Prevotella intermedia lipopolysaccharide-activated RAW 264.7 cells
- The effect of high concentration of glucose on the production of proinflammatory cytokines and nitric oxide induced by lipopolysaccharides from periodontopathic bacteria