Ann Surg Treat Res.
2015 May;88(5):241-245. 10.4174/astr.2015.88.5.241.
Protective effect of ischemic postconditioning against hepatic ischemic reperfusion injury in rat liver
- Affiliations
-
- 1Department of Surgery, Korea University Ansan Hospital, Korea University College of Medicine, Ansan, Korea. tjsong@korea.ac.kr
- 2Department of Surgery, DamSoYu Hospital, Seoul, Korea.
- KMID: 2166991
- DOI: http://doi.org/10.4174/astr.2015.88.5.241
Abstract
- PURPOSE
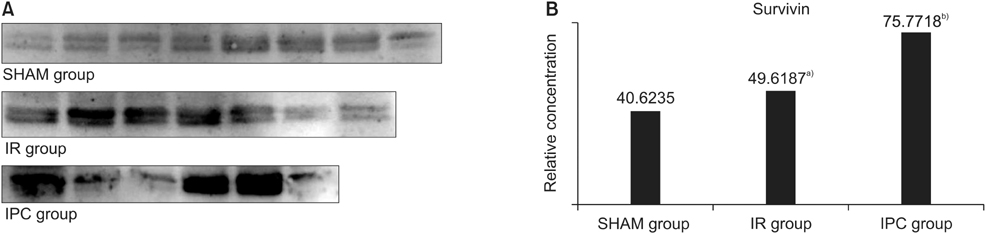
The efficiency of ischemic postconditioning (IPC) was evaluated in a rat model of ischemic liver. Concentration of survivin of liver tissue correlated with the degree of antiapoptosis, so survivin was estimated to evaluate the efficiency of IPC on ischemic reperfusion (IR) injury.
METHODS
Twenty-four healthy rats were divided to three groups (SHAM, IR, and IPC). Rats in the SHAM group displayed no change during 3 hours. Rats in the IR group were ischemic within 1 hour of clamping the left hepatic artery and left portal vein. Reperfusion for 2 hours was then done. IPC group, intermittent 2, 3, 5, and 7 minutes of reperfusion followed by 1 hour of warm ischemia. Two-minute reocclusion was done after each reperfusion. Rat sera were analyzed for AST and ALT, and Western blot analysis of rat liver tissue of rats evaluated malondialdehyde (MDA) and survivin.
RESULTS
MDA in the liver tissue of rats in the IR and IPC group were significantly high than in the liver tissue of the SHAM group (P = 0.003 and P = 0.008, respectively). Survivin was higher in the IPC group than in the SHAM and IR groups (P = 0.021 and P = 0.024, respectively).
CONCLUSION
IPC could not prevent lipid oxidation in liver cell mitochondria, but did aid in the regeneration of ischemic injured liver cells. The results indicate that IPC can suppress the apoptosis of liver cells and reduce reperfusion injury of liver tissue.
MeSH Terms
Figure
Reference
-
1. Gross GJ, Auchampach JA. Reperfusion injury: does it exist? J Mol Cell Cardiol. 2007; 42:12–18.2. Nauta RJ, Tsimoyiannis E, Uribe M, Walsh DB, Miller D, Butterfield A. Oxygen-derived free radicals in hepatic ischemia and reperfusion injury in the rat. Surg Gynecol Obstet. 1990; 171:120–125.3. Ryter SW, Kim HP, Hoetzel A, Park JW, Nakahira K, Wang X, et al. Mechanisms of cell death in oxidative stress. Antioxid Redox Signal. 2007; 9:49–89.4. Duffy MJ, O'Donovan N, Brennan DJ, Gallagher WM, Ryan BM. Survivin: a promising tumor biomarker. Cancer Lett. 2007; 249:49–60.5. Ouhtit A, Matrougui K, Bengrine A, Koochekpour S, Zerfaoui M, Yousief Z. Survivin is not only a death encounter but also a survival protein for invading tumor cells. Front Biosci. 2007; 12:1260–1270.6. Hausenloy DJ, Yellon DM. Survival kinases in ischemic preconditioning and postconditioning. Cardiovasc Res. 2006; 70:240–253.7. Lei DX, Peng CH, Peng SY, Jiang XC, Wu YL, Shen HW. Safe upper limit of intermittent hepatic inflow occlusion for liver resection in cirrhotic rats. World J Gastroenterol. 2001; 7:713–717.8. Wang KX, Hu SY, Jiang XS, Zhu M, Jin B, Zhang GY, et al. Protective effects of ischaemic postconditioning on warm/cold ischaemic reperfusion injury in rat liver: a comparative study with ischaemic preconditioning. Chin Med J (Engl). 2008; 121:2004–2009.9. Zhao ZQ, Corvera JS, Halkos ME, Kerendi F, Wang NP, Guyton RA, et al. Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol. 2003; 285:H579–H588.10. Galagudza M, Kurapeev D, Minasian S, Valen G, Vaage J. Ischemic postconditioning: brief ischemia during reperfusion converts persistent ventricular fibrillation into regular rhythm. Eur J Cardiothorac Surg. 2004; 25:1006–1010.11. Hausenloy DJ. Signalling pathways in ischaemic postconditioning. Thromb Haemost. 2009; 101:626–634.12. Minor T, Saad S, Nagelschmidt M, Kotting M, Fu Z, Paul A, et al. Successful transplantation of porcine livers after warm ischemic insult in situ and cold preservation including postconditioning with gaseous oxygen. Transplantation. 1998; 65:1262–1264.13. Mockford KA, Girn HR, Homer-Vanniasinkam S. Postconditioning: current controversies and clinical implications. Eur J Vasc Endovasc Surg. 2009; 37:437–442.14. Zhao ZQ, Vinten-Johansen J. Postconditioning: reduction of reperfusioninduced injury. Cardiovasc Res. 2006; 70:200–211.15. Zhu XM, Liu XH. Ischemic postconditioning, a new method to protect tissue from ischemia/reperfusion injury. Sheng Li Ke Xue Jin Zhan. 2007; 38:261–265.16. Ploeg RJ, D'Alessandro AM, Knechtle SJ, Stegall MD, Pirsch JD, Hoffmann RM, et al. Risk factors for primary dysfunction after liver transplantation: a multivariate analysis. Transplantation. 1993; 55:807–813.17. Wu BQ, Chu WW, Zhang LY, Wang P, Ma QY, Wang DH. Protection of preconditioning, postconditioning and combined therapy against hepatic ischemia/reperfusion injury. Chin J Traumatol. 2007; 10:223–227.18. Teixeira AR, Molan NT, Kubrusly MS, Bellodi-Privato M, Coelho AM, Leite KR, et al. Postconditioning ameliorates lipid peroxidation in liver ischemia-reperfusion injury in rats. Acta Cir Bras. 2009; 24:52–56.19. Wang N, Lu JG, He XL, Li N, Qiao Q, Yin JK, et al. Effects of ischemic postconditioning on reperfusion injury in rat liver grafts after orthotopic liver transplantation. Hepatol Res. 2009; 39:382–390.20. Sola S, Brito MA, Brites D, Moura JJ, Rodrigues CM. Membrane structural changes support the involvement of mitochondria in the bile salt-induced apoptosis of rat hepatocytes. Clin Sci (Lond). 2002; 103:475–485.21. Crompton M. The mitochondrial permeability transition pore and its role in cell death. Biochem J. 1999; 341(Pt 2):233–249.22. Li JY, Gu X, Yin HZ, Zhou Y, Zhang WH, Qin YM. Protective effect of ischemic preconditioning on hepatic ischemia-reperfusion injury by advancing the expressive phase of survivin in rats. Hepatobiliary Pancreat Dis Int. 2008; 7:615–620.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Protective effect of combination therapy with ischemic preconditioning and rapamycin in fibrotic rat livers
- Ischemic postconditioning may not influence early brain injury induced by focal cerebral ischemia/reperfusion in rats
- Effect of Ischemic Preconditioning on Catecholamine Release from the Isolated, Ischemic Reperfused Hearts of Rats
- Effects of postconditioning with N,N,N'N'-tetrakis-[2-pyridylmethyl]-ethylenediamine in isolated rat hearts
- CD44 Disruption Attenuates Murine Hepatic Ischemia/Reperfusion Injury