Immune Netw.
2010 Jun;10(3):99-103. 10.4110/in.2010.10.3.99.
GSK3beta Inhibitor Peptide Protects Mice from LPS-induced Endotoxin Shock
- Affiliations
-
- 1Division of Life and Pharmaceutical Sciences, Center for Cell Signaling & Drug Discovery Research, Ewha Womans University, Seoul 120-750, Korea. leesy@ewha.ac.kr
- 2Department of Life Science, College of Natural Sciences, Ewha Womans University, Seoul 120-750, Korea.
- KMID: 2150666
- DOI: http://doi.org/10.4110/in.2010.10.3.99
Abstract
- BACKGROUND
Glycogen synthase kinase 3beta (GSK3beta) is a ubiquitous serine/threonine kinase that is regulated by serine phosphorylation at 9. Recent studies have reported the beneficial effects of a number of the pharmacological GSK3beta inhibitors in rodent models of septic shock. Since most of the GSK3beta inhibitors are targeted at the ATP-binding site, which is highly conserved among diverse protein kinases, the development of novel non-ATP competitive GSK3beta inhibitors is needed.
METHODS
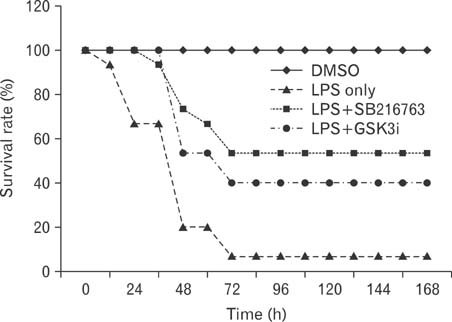
Based on the unique phosphorylation motif of GSK3beta, we designed and generated a novel class of GSK3beta inhibitor (GSK3i) peptides. In addition, we investigated the effects of a GSK3i peptide on lipopolysaccharide (LPS)-stimulated cytokine production and septic shock. Mice were intraperitoneally injected with GSK3i peptide and monitored over a 7-day period for survival.
RESULTS
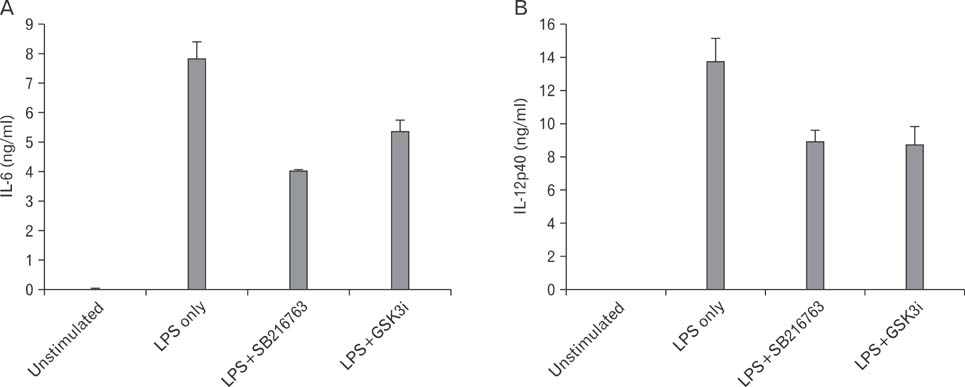
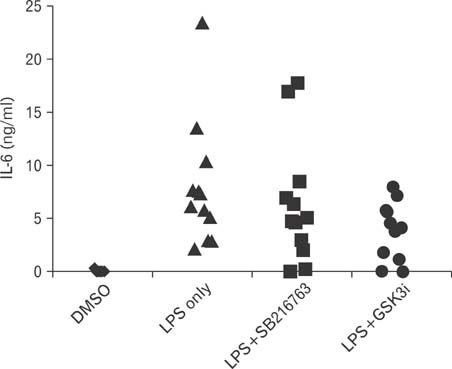
We first demonstrate its effects on LPS-stimulated pro-inflammatory cytokine production including interleukin (IL)-6 and IL-12p40. LPS-induced IL-6 and IL-12p40 production in macrophages was suppressed when macrophages were treated with the GSKi peptide. Administration of the GSK3i peptide potently suppressed LPS-mediated endotoxin shock.
CONCLUSION
Collectively, we present a rational strategy for the development of a therapeutic GSK3i peptide. This peptide may serve as a novel template for the design of non-ATP competitive GSK3 inhibitors.
Keyword
MeSH Terms
-
Animals
Cytokines
Glycogen Synthase Kinase 3
Glycogen Synthase Kinases
Interleukin-12 Subunit p40
Interleukin-6
Interleukins
Macrophages
Mice
Peptides
Phosphorylation
Phosphotransferases
Protein Kinases
Rodentia
Serine
Shock
Shock, Septic
Cytokines
Glycogen Synthase Kinase 3
Glycogen Synthase Kinases
Interleukin-12 Subunit p40
Interleukin-6
Interleukins
Peptides
Phosphotransferases
Protein Kinases
Serine
Figure
Reference
-
1. Cohen P, Frame S. The renaissance of GSK3. Nat Rev Mol Cell Biol. 2001. 2:769–776.
Article2. Frame S, Cohen P. GSK3 takes centre stage more than 20 years after its discovery. Biochem J. 2001. 359:1–16.
Article3. Woodgett JR. Judging a protein by more than its name: GSK-3. Sci STKE. 2001. 2001:re12.
Article4. Embi N, Rylatt DB, Cohen P. Glycogen synthase kinase-3 from rabbit skeletal muscle. Separation from cyclic-AMP-dependent protein kinase and phosphorylase kinase. Eur J Biochem. 1980. 107:519–527.5. Beals CR, Sheridan CM, Turck CW, Gardner P, Crabtree GR. Nuclear export of NF-ATc enhanced by glycogen synthase kinase-3. Science. 1997. 275:1930–1934.
Article6. El Jamali A, Freund C, Rechner C, Scheidereit C, Dietz R, Bergmann MW. Reoxygenation after severe hypoxia induces cardiomyocyte hypertrophy in vitro: activation of CREB downstream of GSK3 beta. FASEB J. 2004. 18:1096–1098.7. Wei W, Jin J, Schlisio S, Harper JW, Kaelin WG Jr. The v-Jun point mutation allows c-Jun to escape GSK3-dependent recognition and destruction by the Fbw7 ubiquitin ligase. Cancer Cell. 2005. 8:25–33.
Article8. Gregory MA, Qi Y, Hann SR. Phosphorylation by glycogen synthase kinase-3 controls c-myc proteolysis and subnuclear localization. J Biol Chem. 2003. 278:51606–51612.
Article9. Liang J, Slingerland JM. Multiple roles of the PI3K/PKB (Akt) pathway in cell cycle progression. Cell Cycle. 2003. 2:339–345.
Article10. Gum RJ, Gaede LL, Koterski SL, Heindel M, Clampit JE, Zinker BA, Trevillyan JM, Ulich RG, Jirousek MR, Rondinone CM. Reduction of protein tyrosine phosphatase 1B increases insulin-dependent signaling in ob/ob mice. Diabetes. 2003. 52:21–28.
Article11. Ring DB, Johnson KW, Henriksen EJ, Nuss JM, Goff D, Kinnick TR, Ma ST, Reeder JW, Samuels I, Slabiak T, Wagman AS, Hammond ME, Harrison SD. Selective glycogen synthase kinase 3 inhibitors potentiate insulin activation of glucose transport and utilization in vitro and in vivo. Diabetes. 2003. 52:588–595.
Article12. Martinez A, Perez DI. GSK-3 inhibitors: a ray of hope for the treatment of Alzheimer's disease? J Alzheimers Dis. 2008. 15:181–191.
Article13. Beaulieu JM, Gainetdinov RR, Caron MG. Akt/GSK3 signaling in the action of psychotropic drugs. Annu Rev Pharmacol Toxicol. 2009. 49:327–347.
Article14. Inoki K, Ouyang H, Zhu T, Lindvall C, Wang Y, Zhang X, Yang Q, Bennett C, Harada Y, Stankunas K, Wang CY, He X, MacDougald OA, You M, Williams BO, Guan KL. TSC2 integrates Wnt and energy signals via a coordinated phosphorylation by AMPK and GSK3 to regulate cell growth. Cell. 2006. 126:955–968.
Article15. Martin M, Rehani K, Jope RS, Michalek SM. Toll-like receptor-mediated cytokine production is differentially regulated by glycogen synthase kinase 3. Nat Immunol. 2005. 6:777–784.
Article16. Hu X, Paik PK, Chen J, Yarilina A, Kockeritz L, Lu TT, Woodgett JR, Ivashkiv LB. IFN-gamma suppresses IL-10 production and synergizes with TLR2 by regulating GSK3 and CREB/AP-1 proteins. Immunity. 2006. 24:563–574.
Article17. Rehani K, Wang H, Garcia CA, Kinane DF, Martin M. Toll-like receptor-mediated production of IL-1Ra is negatively regulated by GSK3 via the MAPK ERK1/2. J Immunol. 2009. 182:547–553.
Article18. Beurel E, Jope RS. Glycogen synthase kinase-3 promotes the synergistic action of interferon-gamma on lipopolysaccharide-induced IL-6 production in RAW264.7 cells. Cell Signal. 2009. 21:978–985.
Article19. Cohen P, Goedert M. GSK3 inhibitors: development and therapeutic potential. Nat Rev Drug Discov. 2004. 3:479–487.
Article20. Wada A. GSK-3 inhibitors and insulin receptor signaling in health, disease, and therapeutics. Front Biosci. 2009. 14:1558–1570.
Article21. Phukan S, Babu VS, Kannoji A, Hariharan R, Balaji VN. GSK3beta: role in therapeutic landscape and development of modulators. Br J Phamacol. 2010. 160:1–19.22. Plotkin B, Kaidanovich O, Talior I, Eldar-Finkelman H. Insulin mimetic action of synthetic phosphorylated peptide inhibitors of glycogen synthase kinase-3. J Pharmacol Exp Ther. 2003. 305:974–980.
Article23. Fischer PM. CDK versus GSK-3 inhibition: a purple haze no longer? Chem Biol. 2003. 10:1144–1146.24. Phiel CJ, Klein PS. Molecular targets of lithium action. Annu Rev Pharmacol Toxicol. 2001. 41:789–813.
Article25. Sawa M. Strategies for the design of selective protein kinase inhibitors. Mini Rev Med Chem. 2008. 8:1291–1297.
Article26. Dajani R, Fraser E, Roe SM, Young N, Good V, Dale TC, Pearl LH. Crystal structure of glycogen synthase kinase 3 beta: structural basis for phosphate-primed substrate specificity and autoinhibition. Cell. 2001. 105:721–732.27. Frame S, Cohen P, Biondi RM. A common phosphate binding site explains the unique substrate specificity of GSK3 and its inactivation by phosphorylation. Mol Cell. 2001. 7:1321–1327.
Article28. ter Haar E, Coll JT, Austen DA, Hsiao HM, Swenson L, Jain J. Structure of GSK3beta reveals a primed phosphorylation mechanism. Nat Struct Biol. 2001. 8:593–596.29. Choi JM, Ahn MH, Chae WJ, Jung YG, Park JC, Song HM, Kim YE, Shin JA, Park CS, Park JW, Park TK, Lee JH, Seo BF, Kim KD, Kim ES, Lee DH, Lee SK, Lee SK. Intranasal delivery of the cytoplasmic domain of CTLA-4 using a novel protein transduction domain prevents allergic inflammation. Nat Med. 2006. 12:574–579.
Article30. Kim H, Choi HK, Shin JH, Kim KH, Huh JY, Lee SA, Ko CY, Kim HS, Shin HI, Lee HJ, Jeong D, Kim N, Choi Y, Lee SY. Selective inhibition of RANK blocks osteoclast maturation and function and prevents bone loss in mice. J Clin Invest. 2009. 119:813–825.
Article31. Beurel E, Michalek SM, Jope RS. Innate and adaptive immune responses regulated by glycogen synthase kinase-3 (GSK3). Trends Immunol. 2010. 31:24–31.
Article32. Jope RS, Yuskaitis CJ, Beurel E. Glycogen synthase kinase-3 (GSK3): inflammation, diseases, and therapeutics. Neurochem Res. 2007. 32:577–595.
Article33. Rayasam GV, Tulasi VK, Sodhi R, Davis JA, Ray A. Glycogen synthase kinase 3: more than a namesake. Br J Pharmacol. 2009. 156:885–898.
Article34. Bax B, Carter PS, Lewis C, Guy AR, Bridges A, Tanner R, Pettman G, Mannix C, Culbert AA, Brown MJ, Smith DG, Reith AD. The structure of phosphorylated GSK-3beta complexed with a peptide, FRATtide, that inhibits beta-catenin phosphorylation. Structure. 2001. 9:1143–1152.
Article35. Rommel C, Bodine SC, Clarke BA, Rossman R, Nunez L, Stitt TN, Yancopoulos GD, Glass DJ. Mediation of IGF-1-induced skeletal myotube hypertrophy by PI(3)K/Akt/mTOR and PI(3)K/Akt/GSK3 pathways. Nat Cell Biol. 2001. 3:1009–1013.
Article36. Mottet D, Dumont V, Deccache Y, Demazy C, Ninane N, Raes M, Michiels C. Regulation of hypoxia-inducible factor-1alpha protein level during hypoxic conditions by the phosphatidylinositol 3-kinase/Akt/glycogen synthase kinase 3beta pathway in HepG2 cells. J Biol Chem. 2003. 278:31277–31285.
Article37. Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature. 1995. 378:785–789.
Article38. Doble BW, Woodgett JR. GSK-3: tricks of the trade for a multi-tasking kinase. J Cell Sci. 2003. 116:1175–1186.
Article39. Plotkin B, Kaidanovich O, Talior I, Eldar-Finkelman H. Insulin mimetic action of synthetic phosphorylated peptide inhibitors of glycogen synthase kinase-3. J Pharmacol Exp Ther. 2003. 305:974–980.
Article40. Sato AK, Viswanathan M, Kent RB, Wood CR. Therapeutic peptides: technological advances driving peptides into development. Curr Opin Biotechnol. 2006. 17:638–642.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Regulation of Endotoxin - Induced TNF-alpha Gene Expression
- The Extraction of Vibrio vulnificus Lipopolysaccharide by the Phenol-Chloroform-Petroleum Ether Method and Biological Activity of the LPS
- The Effect of Heat Shock Protein on Inflammatory Reaction and Cataractogenesis in Endotoxin-induced Uveitis
- Study of Pro-inflammatory Cytokines in Endotoxin Induced Uveitis
- Expression of Matrix Metalloproteinase-9 and Tissue Inhibitor of Metalloproteinase-1 after Administration of Endotoxin in Diabetic Rats